Which State Of Matter Has No Definite Shape Or Volume
Juapaving
Apr 02, 2025 · 6 min read
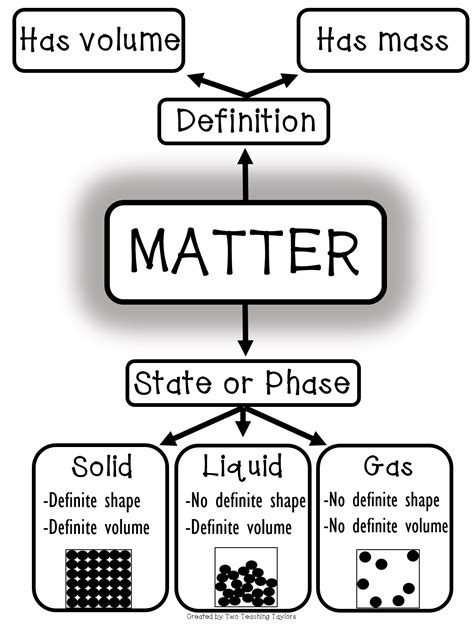
Table of Contents
Which State of Matter Has No Definite Shape or Volume? Understanding Gases
The answer is simple: gases. Unlike solids and liquids, gases do not possess a definite shape or volume. They readily conform to the shape and volume of their container. This defining characteristic stems from the unique properties of gas molecules and their interactions, a topic we'll explore in detail throughout this article. Understanding the behavior of gases is crucial in numerous fields, from meteorology and atmospheric science to chemistry, engineering, and even biology.
The Unique Properties of Gases
Gases are one of the four fundamental states of matter (the others being solids, liquids, and plasma). Their lack of definite shape and volume is a direct consequence of several key properties:
1. Weak Intermolecular Forces:
Gas molecules are characterized by weak intermolecular forces. This means that the attractive forces between individual gas molecules are significantly weaker than those found in solids or liquids. These weak forces allow gas molecules to move freely and independently, leading to their ability to expand and fill any available space.
2. High Kinetic Energy:
Gas molecules possess high kinetic energy. This means they are in constant, rapid, random motion. This constant movement allows them to overcome the weak intermolecular forces and spread out to occupy the entire volume of their container. The higher the temperature, the greater the kinetic energy, and the faster the gas molecules move.
3. Large Intermolecular Distances:
The distance between gas molecules is considerably larger than their size. This vast separation minimizes intermolecular interactions, further contributing to the lack of a definite shape or volume. The spacious arrangement allows gases to be easily compressed.
4. Compressibility and Expansibility:
Gases are highly compressible and expansible. This means their volume can be significantly reduced by applying pressure, or conversely, they can expand to fill a larger volume if the pressure is reduced or the container size is increased. This property is a direct result of the large intermolecular distances and weak intermolecular forces.
Understanding Gas Behavior: Key Concepts
Several fundamental concepts are essential for a thorough understanding of gas behavior:
1. Pressure (P):
Pressure is defined as the force exerted by gas molecules per unit area on the walls of their container. It's typically measured in atmospheres (atm), Pascals (Pa), or millimeters of mercury (mmHg). The pressure exerted by a gas is directly related to the number of gas molecules, their kinetic energy, and the size of the container. Higher temperature and more molecules mean higher pressure.
2. Volume (V):
Volume refers to the amount of space occupied by the gas. It is typically measured in liters (L) or cubic meters (m³). The volume of a gas is directly dependent on the size and shape of its container.
3. Temperature (T):
Temperature is a measure of the average kinetic energy of the gas molecules. It's typically measured in Kelvin (K), Celsius (°C), or Fahrenheit (°F). A higher temperature means higher kinetic energy, and therefore, faster-moving molecules and increased pressure.
4. Amount of Gas (n):
The amount of gas is usually expressed in moles (mol), which represents a specific number of gas molecules (Avogadro's number, approximately 6.022 x 10²³). More moles of gas mean more molecules and thus, higher pressure, assuming all other factors remain constant.
Ideal Gas Law: A Simplified Model
The ideal gas law provides a simplified model for describing the behavior of gases under certain conditions. It's expressed mathematically as:
PV = nRT
Where:
- P is pressure
- V is volume
- n is the amount of gas (in moles)
- R is the ideal gas constant (a proportionality constant)
- T is temperature (in Kelvin)
The ideal gas law works well for many gases under moderate conditions of temperature and pressure. However, it's important to remember that it's a simplification, and real gases may deviate from this behavior at extreme temperatures or pressures. Real gas behavior requires more complex equations to account for intermolecular forces and the finite volume occupied by the gas molecules themselves.
Real Gases vs. Ideal Gases: The Deviations
While the ideal gas law provides a good approximation for many gases under normal conditions, real gases deviate from ideal behavior under certain conditions:
-
High Pressure: At high pressures, gas molecules are closer together, and the intermolecular forces become more significant. This causes the gas to deviate from ideal behavior, as the forces are no longer negligible.
-
Low Temperature: At low temperatures, the kinetic energy of the gas molecules is lower. This makes the intermolecular forces more influential, causing the gas to deviate from ideal behavior. The molecules are more likely to stick together, reducing the overall volume and pressure.
-
Large Molecular Size: Gases with large molecules may deviate from ideal behavior, as the volume occupied by the molecules themselves becomes more significant compared to the total volume of the gas.
Equations like the van der Waals equation are used to describe the behavior of real gases, incorporating correction factors to account for intermolecular forces and molecular volume.
Applications of Understanding Gases
The principles governing gas behavior are vital in many fields:
-
Meteorology: Understanding atmospheric pressure, temperature, and humidity is crucial for weather forecasting. Atmospheric gases like nitrogen, oxygen, and carbon dioxide play a significant role in shaping weather patterns.
-
Aviation: The behavior of gases in aircraft engines and within the atmosphere significantly impacts flight dynamics and safety. Changes in atmospheric pressure with altitude are crucial considerations.
-
Chemistry: Gas laws are fundamental in chemical reactions involving gases, as they help predict the volume and pressure changes that occur during the reaction.
-
Medicine: Understanding the behavior of respiratory gases (oxygen and carbon dioxide) is essential for respiratory physiology and treatment. Anesthesia also relies on the properties of gases.
-
Engineering: Gas behavior is crucial in the design of various systems, including pipelines, combustion engines, and refrigeration systems. Understanding gas dynamics is critical for efficient and safe operations.
The Importance of Gas Laws in Daily Life
While the intricacies of gas laws may seem distant from daily life, they are fundamentally relevant. For example:
-
Balloons: The ability of a balloon to inflate is a direct result of the expansion of gases. The gas inside the balloon exerts pressure, pushing against the balloon's walls to maintain its shape.
-
Breathing: The process of breathing involves the exchange of gases between the lungs and the atmosphere. Understanding gas pressure differences allows for efficient gas exchange.
-
Cooking: Many cooking methods rely on the properties of gases. For example, baking relies on the expansion of gases trapped in dough, giving bread its characteristic texture.
Conclusion: A Deep Dive into the World of Gases
Gases, with their undefined shape and volume, are fascinating states of matter with behavior dictated by fundamental principles. Understanding the interplay of pressure, volume, temperature, and the amount of gas, encapsulated in concepts like the ideal gas law, unlocks a deeper understanding of how the world around us works. While the ideal gas law serves as a valuable tool, remembering its limitations and the intricacies of real gas behavior allows for a more comprehensive understanding of gaseous systems, impacting fields from everyday life to cutting-edge scientific research. The study of gases continues to be an active area of research, continually revealing new insights into their properties and applications.
Latest Posts
Latest Posts
-
Balanced Chemical Equation For Magnesium Oxide
Apr 03, 2025
-
What Is The Group Of Birds Called
Apr 03, 2025
-
Common Factors Of 28 And 42
Apr 03, 2025
-
Can The Quotient Of Two Irrational Numbers Be Rational
Apr 03, 2025
-
12 Cm Is What In Inches
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which State Of Matter Has No Definite Shape Or Volume . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
