What Are The Vertical Rows On The Periodic Table Called
Juapaving
Apr 02, 2025 · 6 min read
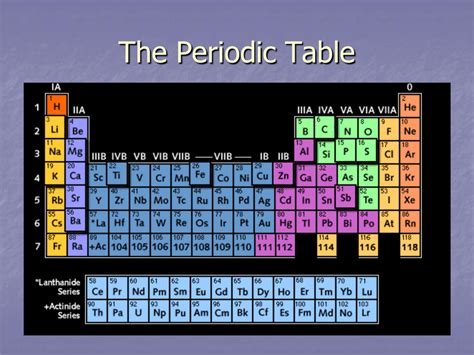
Table of Contents
What are the Vertical Rows on the Periodic Table Called? Understanding Groups and Families in Chemistry
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. While many are familiar with the horizontal rows, called periods, the vertical columns hold equally significant information and are known as groups or families. Understanding the organization and properties associated with these groups is fundamental to grasping chemical behavior and reactivity. This comprehensive guide delves into the intricacies of these vertical columns, exploring their naming conventions, characteristic properties, and the underlying reasons for their periodic arrangement.
Understanding the Vertical Organization: Groups and Families
The vertical columns of the periodic table are called groups or families. These terms are used interchangeably and represent elements with similar chemical properties. This similarity stems from the fact that elements within the same group have the same number of valence electrons. Valence electrons are the electrons in the outermost shell of an atom, and they are the primary players in chemical bonding. Since elements in a group share the same number of valence electrons, they tend to form similar types of chemical bonds and exhibit comparable chemical reactivity.
Why are they called Groups and Families?
The terms "group" and "family" are both descriptive of the similarities within the vertical columns. "Group" simply implies a collection of elements sharing common traits. "Family," on the other hand, evokes a sense of kinship, emphasizing the close relationship and shared characteristics among the elements. Both terms are widely accepted and used in chemistry literature.
The 18 Groups of the Periodic Table: A Detailed Look
The modern periodic table consists of 18 groups, numbered from 1 to 18. Historically, different numbering systems were used (IA-VIIIA, IB-VIIIB), leading to some confusion. However, the IUPAC (International Union of Pure and Applied Chemistry) officially adopted the 1-18 system to standardize the nomenclature. Let's examine some of the most important groups in detail:
Group 1: Alkali Metals
Alkali metals are highly reactive elements, characterized by having one valence electron. This single electron is easily lost, resulting in the formation of +1 ions. Their reactivity increases as you go down the group. Examples include:
- Lithium (Li): Used in batteries and certain alloys.
- Sodium (Na): Essential for human health and found abundantly in table salt (NaCl).
- Potassium (K): Also crucial for biological processes.
- Rubidium (Rb) and Cesium (Cs): Used in specialized applications, such as atomic clocks.
- Francium (Fr): A radioactive element, extremely rare.
Key Properties of Alkali Metals:
- Low density: They are relatively light.
- Low melting and boiling points: They are soft and easily melted.
- Highly reactive: They readily react with water and air.
- Good conductors of heat and electricity: Due to their loosely held valence electron.
Group 2: Alkaline Earth Metals
Alkaline earth metals have two valence electrons, making them less reactive than alkali metals but still capable of forming +2 ions. They are also more dense and have higher melting points than alkali metals. Examples include:
- Beryllium (Be): Used in alloys and aerospace applications.
- Magnesium (Mg): Crucial for plant and animal life, used in structural materials.
- Calcium (Ca): Essential for bone health and various biological processes.
- Strontium (Sr): Used in fireworks for its red color.
- Barium (Ba): Used in contrast agents for medical imaging.
- Radium (Ra): A radioactive element.
Key Properties of Alkaline Earth Metals:
- Higher density than alkali metals: They are heavier.
- Higher melting and boiling points than alkali metals.
- Reactive, but less so than alkali metals: React less vigorously with water.
- Good conductors of heat and electricity.
Group 17: Halogens
Halogens are highly reactive nonmetals with seven valence electrons. They readily gain one electron to achieve a stable octet, forming -1 ions. This strong electron affinity makes them excellent oxidizing agents. Examples include:
- Fluorine (F): The most reactive halogen, used in fluoridated water and Teflon.
- Chlorine (Cl): Used in disinfectants and PVC plastics.
- Bromine (Br): Used in flame retardants and photographic chemicals.
- Iodine (I): Used as an antiseptic and in iodized salt.
- Astatine (At): A radioactive element.
Key Properties of Halogens:
- Highly reactive nonmetals: They readily react with metals and other nonmetals.
- Exist as diatomic molecules (e.g., F₂, Cl₂): Two atoms bonded together.
- Strong oxidizing agents: They readily accept electrons.
- Varied physical states: Fluorine and chlorine are gases, bromine is a liquid, and iodine is a solid.
Group 18: Noble Gases
Noble gases are unique because they have a complete outer electron shell (eight valence electrons, except for helium with two). This stable configuration makes them extremely unreactive, hence their name "noble" gases. Examples include:
- Helium (He): Used in balloons and cryogenics.
- Neon (Ne): Used in neon lights.
- Argon (Ar): Used in incandescent light bulbs.
- Krypton (Kr): Used in some lasers.
- Xenon (Xe): Used in some specialized lighting.
- Radon (Rn): A radioactive element.
Key Properties of Noble Gases:
- Extremely unreactive: They rarely form chemical compounds.
- Colorless and odorless gases under normal conditions.
- Low boiling points: They exist as gases at room temperature.
- Used in various applications due to their inert nature.
The Transition Metals: A Bridge Between Groups
The transition metals occupy the central block of the periodic table. These elements are characterized by partially filled d orbitals in their atoms or ions. This results in a wide variety of oxidation states and complex ion formation. Their properties are less predictable than those of the main group elements.
Key Characteristics of Transition Metals:
- Variable oxidation states: They can form ions with different charges.
- Formation of colored compounds: Many of their compounds exhibit vivid colors.
- Catalytic activity: Many transition metals and their compounds act as catalysts in chemical reactions.
- Magnetic properties: Some transition metals exhibit ferromagnetism (e.g., iron, cobalt, nickel).
Inner Transition Metals: Lanthanides and Actinides
The lanthanides and actinides, also known as the inner transition metals, are placed separately at the bottom of the periodic table for aesthetic reasons. They are characterized by filling of the 4f and 5f orbitals, respectively. The lanthanides are relatively similar in their properties, making their separation and identification challenging. The actinides are all radioactive.
The Importance of Understanding Periodic Table Groups
Understanding the groups of the periodic table is essential for several reasons:
- Predicting Chemical Properties: The group number helps predict the number of valence electrons and thus the reactivity and bonding behavior of an element.
- Understanding Chemical Reactions: Knowing the group membership helps predict the products and outcome of chemical reactions.
- Material Science and Engineering: Understanding the properties of elements within a group is crucial in designing new materials with specific properties.
- Biological Applications: Understanding the roles of elements in biological systems relies on their chemical properties, which are related to their group placement.
Conclusion: Groups – the Key to Understanding Element Behavior
The vertical columns, or groups, of the periodic table provide a powerful organizational framework for understanding the chemical behavior and properties of elements. The similarities within each group, driven by the identical number of valence electrons, provide a predictive tool for chemists and material scientists. Whether investigating the reactivity of alkali metals, the versatility of transition metals, or the inertness of noble gases, a thorough understanding of the periodic table's groups is paramount in deciphering the intricacies of the chemical world. From simple chemical reactions to complex materials design, the groups remain an indispensable tool in the realm of chemistry and beyond.
Latest Posts
Latest Posts
-
Balanced Chemical Equation For Magnesium Oxide
Apr 03, 2025
-
What Is The Group Of Birds Called
Apr 03, 2025
-
Common Factors Of 28 And 42
Apr 03, 2025
-
Can The Quotient Of Two Irrational Numbers Be Rational
Apr 03, 2025
-
12 Cm Is What In Inches
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Are The Vertical Rows On The Periodic Table Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
