Melting Of Wax Is A Physical Change
Juapaving
Apr 01, 2025 · 6 min read
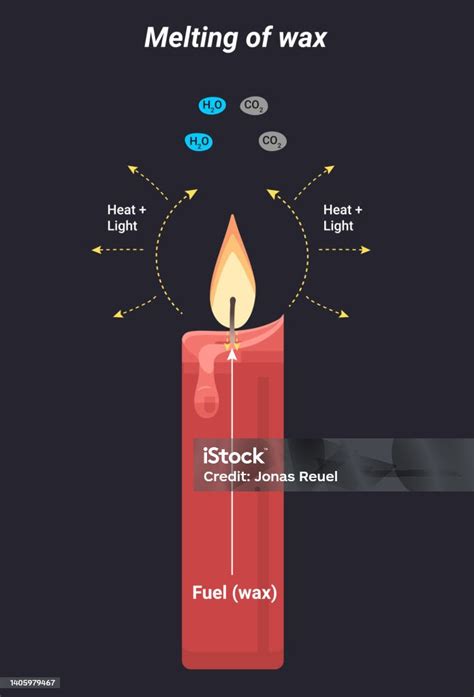
Table of Contents
Melting Wax: A Deep Dive into Physical Changes
The simple act of melting wax, a process we often take for granted, offers a fascinating window into the world of physical changes. Understanding this seemingly mundane phenomenon provides a solid foundation for grasping broader concepts in chemistry and physics. This comprehensive article will explore the melting of wax in detail, examining the scientific principles involved, differentiating it from chemical changes, and investigating various types of wax and their melting behaviors. We'll also delve into the practical applications of wax melting and explore some common misconceptions.
What is a Physical Change?
Before we dive into the specifics of melting wax, let's define what constitutes a physical change. A physical change is any change in the form or appearance of matter that does not alter its chemical composition. In other words, the molecules of the substance remain the same; only their arrangement or state of matter changes. Examples of physical changes include:
- Changes in state: Melting, freezing, boiling, condensation, sublimation (solid to gas), and deposition (gas to solid).
- Changes in shape: Cutting, bending, crushing, or breaking a solid.
- Changes in size: Dissolving a solid in a liquid (although the dissolved substance's properties might change somewhat).
Crucially, physical changes are reversible. You can usually return the substance to its original state. For instance, you can solidify melted wax back into a solid.
Why Melting Wax is a Physical Change
The melting of wax perfectly exemplifies a physical change. When you heat wax, you're supplying it with thermal energy. This energy increases the kinetic energy of the wax molecules, causing them to vibrate more vigorously. As the temperature reaches the wax's melting point, the molecules overcome the intermolecular forces holding them in a fixed, ordered structure (solid). They transition to a more disordered, fluid state (liquid), where they can move more freely past one another.
Crucially, the chemical composition of the wax remains unchanged. The long hydrocarbon chains that make up the wax molecules are still present; they simply have more freedom of movement in the liquid state. This is unlike a chemical change, where new molecules with different chemical bonds are formed.
Understanding Intermolecular Forces
The behavior of wax during melting is directly related to the intermolecular forces acting between its molecules. These forces are relatively weak compared to the strong covalent bonds within the wax molecules. These intermolecular forces, such as van der Waals forces and London dispersion forces, are responsible for holding the wax molecules together in the solid state. The addition of heat provides the energy needed to overcome these forces, allowing the molecules to transition to the liquid phase.
Differentiating Physical and Chemical Changes
It's vital to distinguish physical changes from chemical changes. A chemical change, or chemical reaction, involves the formation of new substances with different chemical compositions. This usually involves breaking and forming chemical bonds. Signs of a chemical change include:
- Formation of a gas: Bubbles or fumes.
- Formation of a precipitate: A solid forming from a solution.
- Color change: A significant, unexpected change in color.
- Temperature change: A significant increase or decrease in temperature without the addition or removal of heat.
- Light or sound emission: A chemical reaction producing light or sound.
Melting wax exhibits none of these characteristics. There's no new substance formed; the wax simply changes from solid to liquid and back again. This reversibility is a key indicator of a physical change.
Different Types of Wax and Their Melting Points
The melting point of wax is not a fixed value; it varies depending on the type of wax. Different waxes have different chemical compositions and, consequently, different intermolecular forces. Here are some common types of wax and their approximate melting points:
-
Paraffin Wax: This is the most common type of wax, derived from petroleum. It has a relatively low melting point, typically ranging from 46-68°C (115-154°F). Its melting behavior is relatively straightforward and easily observable.
-
Beeswax: A natural wax produced by honeybees, beeswax has a higher melting point than paraffin wax, typically around 62-72°C (144-162°F). Its composition is more complex, containing various esters and hydrocarbons.
-
Soy Wax: A vegetable-based wax derived from soybeans, soy wax has a lower melting point than beeswax, usually around 46-54°C (115-130°F). It's becoming increasingly popular due to its environmentally friendly nature.
-
Carnauba Wax: A very hard and high-melting-point wax derived from the leaves of the carnauba palm tree, carnauba wax melts at around 80-87°C (176-189°F). It's often used as a hardener in other wax blends.
These differences in melting points highlight the influence of molecular structure and intermolecular forces on the physical properties of waxes.
Practical Applications of Wax Melting
The melting of wax has numerous practical applications across various industries:
-
Candle Making: This is perhaps the most well-known application. Melting wax allows for the creation of candles with various shapes, sizes, and scents.
-
Cosmetics and Personal Care: Waxes are used in many cosmetic products, including lipsticks, creams, and ointments. Melting wax is crucial in the manufacturing process of these products.
-
Food Industry: Certain waxes are used as coatings for fruits and vegetables to extend their shelf life.
-
Industrial Applications: Waxes are used in various industrial processes, including waterproofing, coating, and polishing.
-
Art and Crafts: Wax is used in various art forms, including encaustic painting, where pigments are mixed with melted wax.
Common Misconceptions about Wax Melting
Several misconceptions surround the melting of wax:
-
Wax burning is a physical change: This is incorrect. Burning wax is a chemical change because it involves a reaction with oxygen, producing carbon dioxide and water. This is evident by the release of heat and light (combustion), the formation of new substances (carbon dioxide and water), and the irreversibility of the process. The wax is not simply changing state but undergoing a chemical transformation.
-
All waxes melt at the same temperature: As discussed earlier, different waxes have different melting points due to differences in their chemical compositions and intermolecular forces.
-
Melting wax is always a rapid process: The rate of melting depends on factors such as the type of wax, the amount of heat applied, and the surface area exposed to the heat source.
Conclusion
The melting of wax serves as a perfect example of a physical change, demonstrating the principles of intermolecular forces and the transition between states of matter. By understanding the scientific principles behind this seemingly simple process, we gain valuable insight into the nature of matter and its transformations. The practical applications of wax melting are extensive and showcase the importance of this physical change in various industries and artistic endeavors. Finally, clarifying common misconceptions ensures a more accurate understanding of this fundamental process in chemistry and physics. The exploration of wax melting, therefore, is not merely an examination of a simple phenomenon, but a gateway to understanding the intricacies of the physical world.
Latest Posts
Latest Posts
-
Do The Diagonals Of A Kite Bisect Bisect The Angles
Apr 03, 2025
-
Peasants And Workers Party Of India
Apr 03, 2025
-
Compressions And Rarefactions Are Characteristics Of
Apr 03, 2025
-
Explain The Process Of Making Yarn From Fibre Answer
Apr 03, 2025
-
Does Lemon Juice Dissolve In Water
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Melting Of Wax Is A Physical Change . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
