How Many Valence Electrons In Helium
Juapaving
Apr 02, 2025 · 5 min read
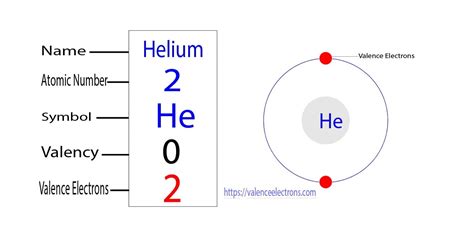
Table of Contents
How Many Valence Electrons Does Helium Have? Understanding Helium's Unique Electronic Structure
Helium, the second element on the periodic table, is a fascinating subject for scientific inquiry, particularly concerning its electronic structure. Understanding its valence electron count is crucial to grasping its unique chemical properties and its role in various applications. This in-depth article delves into the question: how many valence electrons does helium have? We will explore the concept of valence electrons, delve into helium's electronic configuration, and explain why helium’s inertness is directly linked to its valence electron number. Furthermore, we'll touch upon the implications of its unique electronic structure across diverse fields.
Understanding Valence Electrons
Before addressing helium specifically, let's clarify the fundamental concept of valence electrons. Valence electrons are the electrons located in the outermost shell, or energy level, of an atom. These electrons are the primary participants in chemical bonding. They determine an atom's reactivity and how it will interact with other atoms to form molecules or compounds. The number of valence electrons significantly influences an element's chemical properties and its position within the periodic table. Elements in the same group (vertical column) typically share the same number of valence electrons, hence exhibiting similar chemical behavior.
Determining Valence Electrons
There are several ways to determine the number of valence electrons in an atom. The simplest method is by looking at its group number in the periodic table. For the main group elements (groups 1, 2, and 13-18), the group number usually corresponds to the number of valence electrons (with some exceptions for transition metals). However, for a more precise and detailed understanding, we need to examine the element's electronic configuration.
Electronic configuration describes how electrons are distributed among the different energy levels and subshells within an atom. It follows specific rules and principles dictated by quantum mechanics. By knowing the electronic configuration, we can pinpoint the electrons residing in the outermost shell – the valence electrons.
Helium's Electronic Configuration and Valence Electrons
Helium's atomic number is 2, meaning it has two protons and, in a neutral atom, two electrons. Its electronic configuration is remarkably simple: 1s². This means both electrons occupy the lowest energy level (n=1), specifically the s subshell. The s subshell can hold a maximum of two electrons.
Because the 1s subshell is the outermost shell in helium, both electrons are valence electrons. This is a key feature that distinguishes helium from other elements.
Why Only Two Valence Electrons?
Helium's unique configuration stems from the principles governing electron filling in atomic orbitals. The first energy level (n=1) consists only of the 1s subshell, which can accommodate a maximum of two electrons. Once this shell is filled, helium achieves a stable, low-energy state, often referred to as a closed-shell configuration or a noble gas configuration.
This stable configuration explains helium's exceptional inertness. It has no tendency to gain, lose, or share electrons to form chemical bonds with other atoms. This is a characteristic shared by other noble gases, which also have completely filled outermost electron shells.
Helium's Inertness and Its Two Valence Electrons
Helium's chemical inertness is directly linked to its filled valence shell. The stability conferred by this closed-shell configuration makes it exceptionally unreactive. Helium rarely participates in chemical reactions under normal conditions. This unique property sets helium apart from most other elements.
Contrast with Other Elements
Compare helium's behavior to that of hydrogen, with one valence electron. Hydrogen readily reacts with other elements to form stable bonds by sharing or transferring its single valence electron. This reactivity stems from its incomplete valence shell.
The fundamental difference lies in the stability of the filled valence shell in helium versus the unfilled valence shell in hydrogen. This directly impacts their chemical behavior. Helium's reluctance to form chemical bonds is a hallmark of its noble gas nature.
Applications Leveraging Helium's Inertness
Helium's unique properties, primarily its inertness, have led to numerous applications across diverse scientific and technological fields. Its non-reactivity, low density, and low boiling point make it invaluable in various contexts.
Cryogenics and Superconductivity
Helium's exceptionally low boiling point (-268.93 °C) makes it essential in cryogenics. It’s used to cool superconducting magnets in MRI machines, particle accelerators, and other high-tech applications where extreme low temperatures are required. The inert nature prevents unwanted chemical reactions within the cooling systems.
Inert Atmosphere Protection
Helium's inertness also finds application in providing an inert atmosphere for sensitive materials or processes. It's used in welding, to prevent oxidation and improve the weld quality. It is also used to protect reactive materials during manufacturing or storage. The inertness prevents degradation or unwanted chemical changes in the materials.
Leak Detection
Helium's ability to leak through minute openings, combined with its inertness, allows it to be used as a tracer gas for leak detection in high-vacuum systems, pipelines, or any equipment where a small leak could have a significant impact.
Aerostatics and Lighter-Than-Air Applications
Helium's low density, being much lighter than air, renders it suitable for inflating weather balloons, airships, and other lighter-than-air applications. The inertness ensures safety, avoiding the flammability risks associated with using hydrogen.
Scientific Instrumentation
Helium is used as a carrier gas in gas chromatography, a crucial analytical technique used to separate and analyze complex mixtures. Its inertness ensures that the components being analyzed do not react with the carrier gas, preserving the integrity of the results.
Breathing Gas Mixtures
In certain specialized applications, such as deep-sea diving, helium is used in breathing gas mixtures due to its low solubility in blood, preventing the formation of bubbles in the blood (decompression sickness).
Conclusion: Helium's Significance
Helium, with its unique electronic configuration and only two valence electrons, demonstrates the significant influence of electron shell structure on an element's chemical behavior. Its remarkable inertness, stemming from its filled valence shell, is a cornerstone of its vast range of applications in various scientific, industrial, and technological fields. Understanding the number of valence electrons in helium and its resulting properties provides crucial insights into its important role in modern technology and scientific advancements. Its story underscores the fundamental relationship between atomic structure and the macroscopic properties of matter. The seemingly simple question of "how many valence electrons does helium have?" opens a window into the fascinating world of atomic structure and chemical behavior.
Latest Posts
Latest Posts
-
1 Out Of 10 As A Percentage
Apr 03, 2025
-
How Are Prokaryotes And Eukaryotes Alike
Apr 03, 2025
-
Which Of The Following Statements Is Always True
Apr 03, 2025
-
Balanced Chemical Equation For Magnesium Oxide
Apr 03, 2025
-
What Is The Group Of Birds Called
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Valence Electrons In Helium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
