Does Forming A Bond Require Energy
Juapaving
Apr 03, 2025 · 6 min read
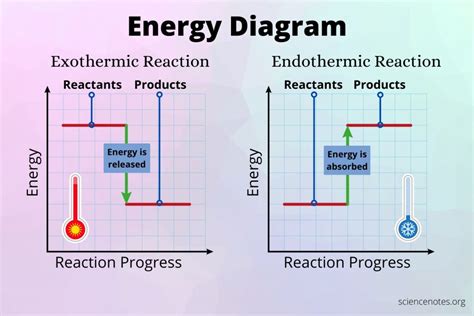
Table of Contents
Does Forming a Bond Require Energy? Exploring the Energetics of Chemical and Social Bonds
The question of whether forming a bond requires energy is deceptively complex. The answer depends heavily on the type of bond being considered. While the intuitive response might be "no, bonds release energy," a deeper dive reveals a more nuanced reality involving activation energies, enthalpy changes, and the subtle dance between energy input and energy output. This article will explore the energetics of both chemical bonds and, drawing parallels, the formation of social bonds, demonstrating the common thread of energy investment in the initial stages of relationship building.
Chemical Bonds: The Energetics of Attraction and Repulsion
In chemistry, bond formation is inherently linked to energy changes. The simplest picture involves the balance between attractive and repulsive forces between atoms. Initially, as atoms approach each other, their electron clouds begin to interact. This interaction can be attractive (leading to bond formation) or repulsive (preventing bond formation).
1. Activation Energy: The Initial Energy Barrier: Before a stable bond can form, the atoms must overcome an initial energy barrier known as the activation energy. This energy is required to bring the atoms close enough together that the attractive forces can overcome the repulsive forces between their electron clouds and nuclei. Think of it as pushing a boulder uphill – you need to expend energy to get it to the top before it can roll down the other side. This initial energy input is crucial for initiating the bond formation process. Without this activation energy, the atoms simply wouldn't get close enough to form a bond, even though the overall process is exothermic (energy releasing). Examples include the activation energy needed for many chemical reactions, such as the combustion of fuel.
2. Enthalpy Change (ΔH): The Overall Energy Balance: Once the activation energy barrier is overcome, the attractive forces between the atoms dominate. Electrons are shared or transferred, resulting in a more stable electronic configuration. This leads to a net release of energy, often in the form of heat, which is reflected in a negative enthalpy change (ΔH). This exothermic process is what often gives the impression that bond formation doesn't require energy. However, it's crucial to remember the crucial role of the preceding activation energy. The overall process is energetically favorable, but requires an initial investment. Consider the formation of a water molecule from hydrogen and oxygen: the reaction releases a significant amount of energy, but still requires an initial "spark" (activation energy) to start.
3. Bond Energy: A Measure of Strength: The strength of a chemical bond is directly related to the energy required to break that bond. A strong bond requires a large input of energy to break, indicating a large release of energy when the bond was initially formed. This is intimately linked to the enthalpy change; a more negative ΔH implies a stronger bond. The concept of bond energy is fundamental in understanding chemical stability and reactivity. Different types of chemical bonds (covalent, ionic, metallic) have widely varying bond energies, reflecting the differences in their electronic structures and attractive forces.
4. Beyond Simple Bonds: Complex Molecular Interactions: The energetics of bond formation become even more complex when considering the formation of larger molecules and the influence of intermolecular forces. These forces, such as hydrogen bonding and van der Waals forces, also involve energy changes, though often less significant than the energies associated with primary chemical bonds. Understanding these interactions is vital for comprehending the behavior of substances in different states of matter and their solubility properties.
Social Bonds: The Energetic Investment in Connection
The parallels between chemical bonding and the formation of social bonds are surprisingly strong. While the mechanisms differ drastically, the fundamental principle of initial energy investment holds true. Forming meaningful social connections requires significant effort and, metaphorically speaking, an "activation energy."
1. Vulnerability and Risk-Taking: Initiating a social bond requires vulnerability. Sharing personal information, expressing emotions, and putting yourself out there are inherently risky behaviors. This "risk-taking" represents the activation energy in social bonding. It demands emotional and psychological energy. Overcoming the fear of rejection or disappointment is a crucial first step. Think of the initial awkwardness of a new friendship or the bravery it takes to start a romantic relationship – these initial stages require significant emotional investment.
2. Communication and Effort: Maintaining a social bond also requires sustained effort. Open communication, active listening, resolving conflicts, and showing empathy all demand considerable energy. This ongoing investment contributes to the strength and longevity of the relationship. The more invested individuals are, the more resilient the bond is likely to be. This continuous effort represents a "maintenance energy" akin to the ongoing energy needed to maintain chemical bond integrity in various conditions.
3. Shared Experiences and Mutual Benefit: Just as strong chemical bonds are associated with a large release of energy, strong social bonds are frequently characterized by a sense of mutual benefit and shared experiences. These shared experiences act as a reinforcing mechanism, strengthening the bond over time and making the initial investment worthwhile. This mutual reinforcement mirrors the stability achieved through strong chemical bonds.
4. Emotional Energy and Resources: The emotional energy invested in social bonding can be considerable. Empathy, compassion, and emotional support all drain resources, yet are essential components of building and maintaining strong relationships. The payoff lies in the reciprocal benefits: companionship, support, love, and belonging. This emotional cost-benefit analysis reflects the overall energy balance observed in chemical bonding (although the metrics are vastly different).
5. The Role of Shared Values and Goals: Similar to how atoms with compatible electronic configurations form stable bonds, individuals with shared values and goals are more likely to form strong social bonds. This compatibility reduces the activation energy required to initiate a connection and promotes sustained interaction. Shared interests provide a common ground and a basis for ongoing engagement, mimicking the stability inherent in strong chemical bonds.
6. The Impact of Negative Interactions: Just as repulsive forces can prevent chemical bonds from forming, negative interactions (arguments, betrayal, etc.) can significantly weaken or even destroy social bonds. Repairing these damaged bonds often requires a substantial reinvestment of energy. This echoes the concept of bond dissociation energy in chemistry.
Conclusion: Energy is the Currency of Connection
Whether considering the formation of chemical bonds or social bonds, the investment of energy is fundamental. While chemical bond formation often results in a net release of energy, a crucial activation energy is always required to initiate the process. Similarly, the formation of social bonds demands substantial emotional, psychological, and communicative energy investment. The strength and stability of the resulting bond, whether chemical or social, are directly correlated to the energy invested in its formation and maintenance. Understanding this energetic perspective offers valuable insight into the dynamics of connection across diverse realms, highlighting the fundamental role of energy in shaping relationships, from the subatomic to the interpersonal. The initial investment, despite sometimes being challenging, ultimately underpins the rewarding strength of the bond itself.
Latest Posts
Latest Posts
-
Find The Prime Factorization Of 210
Apr 03, 2025
-
Biogeochemical Cycles Are Crucial To Ecosystem Function Because
Apr 03, 2025
-
Is Naoh A Base Or Acid
Apr 03, 2025
-
Is A Homogeneous Mixture A Solution
Apr 03, 2025
-
The Electrical Potential Energy Difference Between Two Points
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Does Forming A Bond Require Energy . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
