Is A Homogeneous Mixture A Solution
Juapaving
Apr 03, 2025 · 6 min read
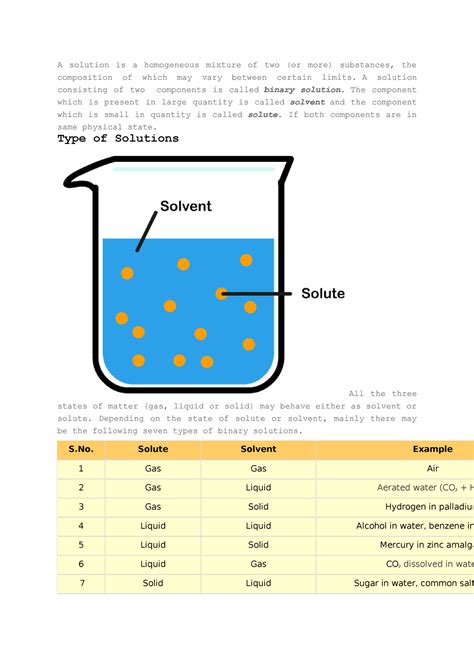
Table of Contents
Is a Homogeneous Mixture a Solution? A Deep Dive into Mixtures and Solutions
The terms "homogeneous mixture" and "solution" are often used interchangeably, leading to confusion. While closely related, they aren't perfectly synonymous. Understanding the nuances between these concepts is crucial for grasping fundamental chemistry principles. This article delves into the definitions, characteristics, and subtle differences between homogeneous mixtures and solutions, providing a comprehensive exploration of their relationship.
Defining Homogeneous Mixtures
A homogeneous mixture is a type of mixture where the composition is uniform throughout. This means that at the macroscopic level (what we can see with the naked eye or a standard microscope), the different components are indistinguishable. No matter which sample you take from the mixture, its properties—like color, density, and concentration—will remain consistent. Think of things like saltwater, air, or sugar dissolved in water. These are all homogeneous mixtures because the individual components are evenly distributed at a visible level.
Key Characteristics of Homogeneous Mixtures:
- Uniform Composition: The most defining characteristic. Every part of the mixture has the same composition and properties.
- Single Phase: A homogeneous mixture exists in a single phase, whether solid, liquid, or gas. You won't see distinct layers or regions with different properties.
- Microscopically Uniform (Often): While appearing uniform to the naked eye, some homogeneous mixtures might show slight variations at a microscopic level. However, these variations are typically negligible in terms of overall properties.
Defining Solutions
A solution, on the other hand, is a specific type of homogeneous mixture. It's a homogeneous mixture composed of two or more substances where one substance, called the solute, is uniformly dispersed within another substance, called the solvent. The solute is typically present in a smaller amount than the solvent. The crucial distinction here is the molecular-level interaction between the solute and solvent.
Key Characteristics of Solutions:
- Homogeneous at the Molecular Level: This is the key differentiator. In a solution, the solute particles are completely dispersed among the solvent particles at a molecular or ionic level, forming a stable mixture. There's no settling or separation over time.
- Solute-Solvent Interactions: Strong interactions between solute and solvent particles are vital for solution formation. These interactions can be ionic (like dissolving salt in water), covalent (like dissolving sugar in water), or a combination of both.
- Clear and Transparent (Usually): Solutions are usually clear and transparent, meaning light can easily pass through them without significant scattering. However, some solutions can be colored due to the properties of the solute.
- Solubility: The ability of a solute to dissolve in a solvent is crucial. The solubility of a solute depends on factors like temperature, pressure, and the nature of the solute and solvent.
The Relationship Between Homogeneous Mixtures and Solutions
All solutions are homogeneous mixtures, but not all homogeneous mixtures are solutions. This is the critical point of understanding the difference. The distinction lies in the degree of mixing and the nature of the interactions at the molecular level.
Examples illustrating the difference:
- Saltwater: This is both a homogeneous mixture and a solution. Salt (solute) is completely dissolved in water (solvent) at the molecular level, forming a stable, uniform mixture.
- Air: Air is a homogeneous mixture of various gases (nitrogen, oxygen, argon, etc.). However, it's generally not considered a solution because there isn't a clear solute and solvent distinction. The gases are mixed uniformly but don't interact with each other in the same way as a solute and solvent in a true solution.
- Brass: Brass is a homogeneous mixture of copper and zinc. It's a solid solution, or an alloy, where the zinc atoms are uniformly dispersed within the copper matrix at the atomic level. The key is the atomic-level dispersion leading to the formation of a solid solution.
- Milk: While appearing homogeneous at first glance, milk is actually a colloid, a type of heterogeneous mixture. Tiny fat globules are dispersed in the watery phase, but they aren't dissolved at a molecular level. Under closer examination (like with a microscope), the heterogeneous nature of milk becomes apparent.
Factors Affecting Solution Formation
Several factors influence whether a homogeneous mixture will form a true solution:
- Nature of Solute and Solvent: "Like dissolves like" is a common rule of thumb. Polar solvents (like water) tend to dissolve polar solutes (like sugar), while nonpolar solvents (like oil) tend to dissolve nonpolar solutes (like fats).
- Temperature: Increasing the temperature usually increases the solubility of solids and gases in liquids.
- Pressure: Pressure significantly affects the solubility of gases in liquids. Higher pressure leads to greater solubility.
- Particle Size: For a true solution to form, the solute particles must be small enough to be completely dispersed at the molecular level. Larger particles can lead to suspensions or colloids.
Examples of Homogeneous Mixtures that aren't Solutions
Many examples of homogeneous mixtures are not solutions. Consider these:
- Air (as discussed earlier): A homogenous blend of gases, but without the defined solute-solvent relationship.
- Alloys (some): Some alloys are considered solutions (like brass), but others exhibit more complex interactions, leading to a homogenous blend without the strict definition of a solution.
- Certain gases: A mixture of gases like methane and ethane will be homogenous, but doesn't have a clear distinction between solute and solvent.
Advanced Concepts: Types of Solutions and Concentrations
Understanding solutions goes beyond simply defining them. Different types of solutions exist, classified by the physical states of the solute and solvent:
- Solid solutions: Where the solute is dissolved in a solid solvent (e.g., alloys).
- Liquid solutions: Where the solute is dissolved in a liquid solvent (e.g., saltwater).
- Gaseous solutions: Where the solute is dissolved in a gaseous solvent (e.g., air).
Furthermore, the concentration of a solution, expressing the amount of solute dissolved in a given amount of solvent or solution, is a crucial aspect:
- Molarity (M): Moles of solute per liter of solution.
- Molality (m): Moles of solute per kilogram of solvent.
- Percent by mass (%): Mass of solute divided by mass of solution, multiplied by 100.
- Percent by volume (%): Volume of solute divided by volume of solution, multiplied by 100.
Conclusion: A Clear Distinction
While the terms are often used interchangeably, understanding the distinction between a homogeneous mixture and a solution is crucial. All solutions are homogeneous mixtures, characterized by a uniform composition at the molecular level and defined solute-solvent interactions. However, many homogeneous mixtures lack this molecular-level uniformity and clear solute-solvent distinction, thus not fitting the strict definition of a solution. This understanding lays the groundwork for deeper exploration of various chemistry concepts related to mixtures and solutions, including solubility, concentration, and reaction kinetics. Recognizing the nuances allows for a more precise and comprehensive understanding of chemical systems. The level of interaction at a molecular scale is the key differentiator, pushing beyond mere visual uniformity to examine the underlying forces that govern the stability and properties of the mixture.
Latest Posts
Latest Posts
-
Exterior Angle Of A Regular Hexagon
Apr 04, 2025
-
What Is The Radius Of Circle With Centre N
Apr 04, 2025
-
Pick Up The Incorrect Statement From The Following
Apr 04, 2025
-
Fe Iron Rusts Physical Or Chemical Change
Apr 04, 2025
-
The Eye And Ear Are Part Of
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Is A Homogeneous Mixture A Solution . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
