As The Concentration Of An Electrolyte Reduces The Conductivity
Juapaving
Apr 05, 2025 · 6 min read
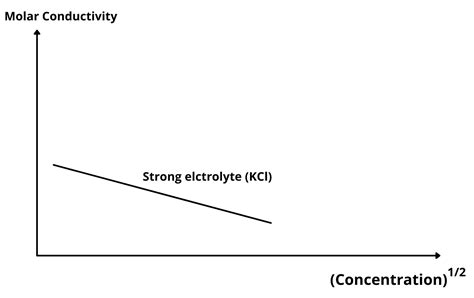
Table of Contents
As the Concentration of an Electrolyte Reduces, Conductivity Decreases: A Deep Dive into Electrolytic Conduction
Electrolytic conductivity, the measure of a solution's ability to conduct electricity, is intrinsically linked to the concentration of its dissolved electrolytes. While it might seem intuitive that higher concentrations lead to higher conductivity, understanding the nuanced relationship requires a deeper exploration of the underlying mechanisms. This article delves into the intricate factors influencing electrolytic conductivity, explaining why conductivity decreases as electrolyte concentration reduces, and exploring the implications across various applications.
The Fundamentals of Electrolytic Conduction
Electrolytic conduction relies on the movement of charged particles, or ions, within a solution. When an electrolyte dissolves in a solvent (like water), it dissociates into its constituent ions – positively charged cations and negatively charged anions. Under the influence of an applied electric field (e.g., from a battery), these ions migrate: cations towards the cathode (negative electrode) and anions towards the anode (positive electrode), thus constituting an electric current. The magnitude of this current is directly proportional to the solution's conductivity.
Factors Influencing Conductivity
Several factors besides electrolyte concentration affect conductivity. These include:
-
Nature of the electrolyte: Different electrolytes dissociate to varying degrees. Strong electrolytes, like NaCl, dissociate almost completely, yielding a high concentration of ions. Weak electrolytes, like acetic acid, only partially dissociate, resulting in fewer charge carriers and lower conductivity. The nature of the ions themselves – their size, charge, and hydration – also plays a crucial role. Smaller, highly charged ions generally exhibit greater mobility.
-
Solvent properties: The solvent's dielectric constant significantly influences ion dissociation and mobility. High dielectric constant solvents, like water, effectively screen the electrostatic attraction between ions, promoting dissociation and enhancing conductivity. The viscosity of the solvent also plays a role; higher viscosity hinders ion movement, reducing conductivity.
-
Temperature: Increasing temperature typically boosts conductivity. Higher temperatures increase the kinetic energy of ions, leading to faster migration and enhanced conductivity. This effect is more pronounced in solutions with weak electrolytes due to increased dissociation at higher temperatures.
-
Concentration: This is the primary focus of this article. The relationship between concentration and conductivity is not linear, but rather follows a complex curve.
The Non-Linear Relationship Between Concentration and Conductivity
The conductivity of an electrolyte solution doesn't increase linearly with concentration. At low concentrations, increasing the concentration dramatically increases conductivity. This is because the number of charge carriers (ions) available to conduct electricity increases proportionally. However, as the concentration increases further, the conductivity increase becomes less pronounced, eventually reaching a maximum and then potentially even decreasing at very high concentrations.
Why Conductivity Decreases at High Concentrations
Several factors contribute to the decrease in conductivity at high concentrations:
-
Increased Ion-Ion Interactions: At high concentrations, ions are much closer together. This leads to increased electrostatic interactions between ions of opposite charges, forming ion pairs or clusters. These aggregates effectively reduce the number of freely moving ions available for conduction, hindering the current flow. The ions are essentially "tied up" and less mobile.
-
Decreased Ion Mobility: Even without forming distinct aggregates, the high concentration of ions creates a significant "ionic atmosphere" around each ion. This atmosphere consists of ions of opposite charge, creating a drag effect that reduces the mobility of individual ions. The ions are constantly colliding with each other, hindering their movement through the solution.
-
Changes in Solvent Structure: At very high concentrations, the electrolyte can significantly alter the structure of the solvent itself. This can lead to changes in viscosity or other properties that further impact ion mobility and conductivity. The solvent's ability to effectively solvate (surround) the ions may be compromised.
The Role of Molar Conductivity
To better understand the concentration-conductivity relationship, the concept of molar conductivity (Λ<sub>m</sub>) is introduced. Molar conductivity represents the conductivity of a solution containing one mole of electrolyte, normalized by the solution's volume. It accounts for the change in the number of ions per unit volume as concentration varies. The equation is:
Λ<sub>m</sub> = κ/c
where κ is the conductivity and c is the concentration.
Kohlrausch's Law
Kohlrausch's Law describes the behavior of molar conductivity at low concentrations. It states that the molar conductivity of an electrolyte at infinite dilution (Λ<sub>m</sub><sup>∞</sup>) is the sum of the limiting molar conductivities of its individual ions:
Λ<sub>m</sub><sup>∞</sup> = λ<sup>+</sup> + λ<sup>-</sup>
where λ<sup>+</sup> and λ<sup>-</sup> are the limiting molar conductivities of the cation and anion, respectively. This law highlights the independent contribution of each ion to the overall conductivity at low concentrations, where ion-ion interactions are minimal.
Deviation from Kohlrausch's Law at Higher Concentrations
At higher concentrations, deviations from Kohlrausch's Law become significant, reflecting the increasing influence of ion-ion interactions and decreased ion mobility. Graphs of molar conductivity versus concentration show an initial increase followed by a plateau or even a decrease, clearly illustrating the non-linear relationship.
Applications and Implications
The relationship between electrolyte concentration and conductivity has crucial implications across various fields:
-
Electroplating: Controlling the electrolyte concentration is crucial in electroplating to achieve a uniform and desired coating thickness. Too high a concentration can lead to uneven deposition, while too low a concentration might result in slow deposition rates.
-
Batteries: Battery performance strongly depends on the electrolyte's conductivity. High conductivity ensures efficient ion transport within the battery, improving its power output and lifespan. Electrolyte composition and concentration are key parameters in battery design.
-
Corrosion Control: Conductivity measurements are employed in assessing the corrosiveness of solutions. High conductivity generally indicates a higher risk of corrosion due to increased ionic activity.
-
Water Quality Monitoring: Conductivity measurements are a simple and rapid method to assess the total dissolved solids (TDS) in water, a vital parameter for monitoring water quality. Higher conductivity indicates higher TDS, which can affect water's potability and suitability for various applications.
-
Analytical Chemistry: Conductivity measurements are used in various analytical techniques, such as titrations, to determine the concentration of electrolytes or to monitor reaction progress.
Conclusion
The relationship between electrolyte concentration and conductivity is more complex than a simple linear relationship. While increasing concentration initially enhances conductivity, at higher concentrations, ion-ion interactions, decreased ion mobility, and changes in solvent structure lead to a decrease or plateau in conductivity. Understanding these nuances is essential for numerous applications, ranging from electroplating and battery technology to water quality monitoring and analytical chemistry. Further research continues to explore this relationship, particularly for complex electrolytes and non-aqueous solvents. By carefully considering the interplay of these factors, scientists and engineers can optimize systems that rely on the efficient conduction of electricity through electrolytic solutions.
Latest Posts
Latest Posts
-
Electrostatics Is A Branch Of Electricity That Focuses On
Apr 05, 2025
-
Surface Area Of Sphere And Hemisphere
Apr 05, 2025
-
An Object Following A Straight Line Path At A Constant Speed
Apr 05, 2025
-
How Long Is 18 Cm In Inches
Apr 05, 2025
-
Is Combination The Same As Synthesis Chemistry
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about As The Concentration Of An Electrolyte Reduces The Conductivity . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
