Where Are The Metals Located In The Periodic Table
Juapaving
Apr 05, 2025 · 6 min read
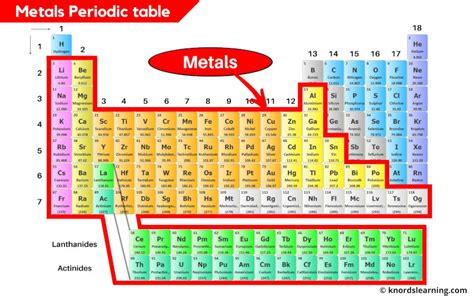
Table of Contents
Where Are the Metals Located in the Periodic Table? A Comprehensive Guide
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. One of the most fundamental classifications of elements is the division into metals, nonmetals, and metalloids. Understanding where metals reside on the periodic table is crucial for comprehending their chemical behavior and practical applications. This comprehensive guide will delve into the location of metals, explore their characteristic properties, and examine specific groups and families within the metallic realm.
The Broad Sweep: Metals on the Periodic Table
The vast majority of elements on the periodic table are metals. They are predominantly located on the left side and towards the bottom of the table. A diagonal line, starting roughly from Boron (B) and extending downwards towards Astatine (At), separates metals from nonmetals. Elements along this line often exhibit properties of both metals and nonmetals, and are classified as metalloids or semimetals.
Think of it like this: Imagine a staircase descending from Boron to Astatine. Elements to the left and below this staircase are generally metals, while those above and to the right are nonmetals. The elements directly on the staircase itself are the metalloids.
Key Characteristics Defining Metals
Before we dive into specific locations, let's briefly review the key properties that define metals:
-
High Electrical Conductivity: Metals readily conduct electricity due to the presence of freely moving electrons in their outer shells. This makes them ideal for electrical wiring and various electronic components.
-
High Thermal Conductivity: Metals efficiently transfer heat, making them useful in applications requiring heat dissipation or transfer, such as cookware and heat sinks.
-
Malleability and Ductility: Metals can be easily hammered into sheets (malleability) and drawn into wires (ductility). This is because the metallic bonding allows atoms to slide past each other without disrupting the overall structure.
-
Luster: Most metals possess a characteristic shine or luster due to their interaction with light. This is often lost when the metal reacts with the environment to form an oxide layer (like rust on iron).
-
High Tensile Strength: Many metals exhibit high tensile strength, meaning they can withstand significant pulling forces before breaking. This makes them suitable for construction and engineering applications.
-
Low Ionization Energy: Metals generally have low ionization energy, meaning they readily lose electrons to form positive ions (cations). This is a key factor in their reactivity and their ability to form various chemical compounds.
Delving Deeper: Metal Groups and Families
Now let's explore some key groups and families of metals located in specific regions of the periodic table:
1. Alkali Metals (Group 1): Highly Reactive Metals
Located in the first column of the periodic table, the alkali metals (lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr)) are extremely reactive metals. Their single valence electron makes them highly prone to losing an electron to form +1 ions. They are soft, silvery-white metals that react vigorously with water and oxygen. Due to their reactivity, they are typically stored under oil to prevent oxidation.
2. Alkaline Earth Metals (Group 2): Reactive, but Less So Than Alkali Metals
Found in the second column, the alkaline earth metals (beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra)) are also reactive, but less so than the alkali metals. They have two valence electrons, readily forming +2 ions. These metals are generally harder and denser than alkali metals. Magnesium, for example, is a lightweight yet strong metal used in alloys for aircraft construction. Calcium is essential for biological processes.
3. Transition Metals (Groups 3-12): A Diverse Group with Variable Oxidation States
The transition metals form a significant block in the middle of the periodic table. They are characterized by their ability to exhibit multiple oxidation states, meaning they can lose varying numbers of electrons to form ions with different charges. This versatility leads to a wide range of chemical properties and applications. Examples include iron (Fe) used in steel, copper (Cu) in electrical wiring, and platinum (Pt) as a catalyst. Their partially filled d orbitals contribute to their unique properties, including colorful compounds and catalytic activity.
4. Post-Transition Metals (Groups 13-15): A Bridge Between Metals and Metalloids
These elements are located to the right of the transition metals. They exhibit some metallic characteristics but also demonstrate properties closer to metalloids. Examples include aluminum (Al), tin (Sn), and lead (Pb). Aluminum is lightweight, strong, and highly corrosion-resistant, making it a versatile metal used in various applications, from aircraft to packaging. Tin is used in solder, while lead, although toxic, has historical applications in plumbing and batteries.
5. Lanthanides and Actinides: Inner Transition Metals
These two series of elements are placed separately at the bottom of the periodic table. They are often referred to as the inner transition metals. The lanthanides (rare earth elements) are characterized by the filling of the 4f orbitals, while the actinides are characterized by the filling of the 5f orbitals. Many of these elements are radioactive and have specialized uses in nuclear technology and medicine.
6. Other Metals: Scattered Throughout the Table
Beyond these major groups, other metals are scattered throughout the periodic table, often exhibiting unique properties. For instance, some metals in the p-block (groups 13-16) show metallic characteristics despite their location.
Metals and Their Real-World Applications: A Glimpse into Our Metal-Dependent World
The widespread presence and diverse properties of metals have made them indispensable to modern society. Their applications span numerous industries and aspects of daily life:
-
Construction and Infrastructure: Steel, iron, aluminum, and concrete (which incorporates metals) form the backbone of our built environment. Bridges, buildings, and transportation systems rely heavily on these materials.
-
Transportation: From automobiles and airplanes to trains and ships, metals are essential components in the design and manufacture of various transportation systems. Lightweight metals like aluminum and titanium are crucial for reducing fuel consumption in aircraft.
-
Electronics and Technology: Copper, silicon, and various transition metals are fundamental to electronics and semiconductor technology. Their electrical conductivity and unique properties are essential for the functioning of computers, smartphones, and other electronic devices.
-
Energy Production: Metals play a significant role in energy production, from the generation of electricity through power plants to the storage of energy in batteries. Lithium-ion batteries, for example, rely on lithium metal for their energy storage capabilities.
-
Medical Applications: Metals are used extensively in medical devices and instruments, including surgical tools, implants, and drug delivery systems. Titanium, for example, is a biocompatible metal frequently used in orthopedic implants.
Conclusion: Understanding the Periodic Table's Metallic Landscape
The periodic table acts as a roadmap to the elements' properties, and the location of metals within this arrangement provides invaluable insight into their behavior and uses. The clear predominance of metals on the left and bottom of the table reflects their abundance and importance in the world around us. From the highly reactive alkali metals to the versatile transition metals, each group contributes uniquely to our technological advancements and our understanding of the material world. By grasping the organization and characteristics of metals within the periodic table, we gain a deeper appreciation for their profound impact on our lives. Further exploration of specific metal groups and their individual properties will reveal even more about the incredible diversity and significance of these essential elements.
Latest Posts
Latest Posts
-
Which Of The Following Is A Product Of Glycolysis
Apr 05, 2025
-
What Is The Test For Hydrogen Gas Called
Apr 05, 2025
-
Common Factors Of 15 And 25
Apr 05, 2025
-
Are Polar Covalent Bonds Stronger Than Nonpolar
Apr 05, 2025
-
What Is A Quarter Of A Half
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Where Are The Metals Located In The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
