Are Polar Covalent Bonds Stronger Than Nonpolar
Juapaving
Apr 05, 2025 · 5 min read
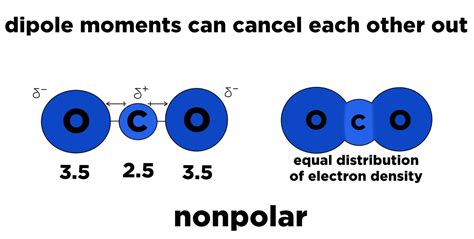
Table of Contents
Are Polar Covalent Bonds Stronger Than Nonpolar? A Deep Dive into Bond Strength
The question of whether polar covalent bonds are stronger than nonpolar covalent bonds doesn't have a simple yes or no answer. While the polarity of a bond can influence its strength, it's not the sole determining factor. Several intertwined factors contribute to the overall strength of a chemical bond, making a direct comparison complex. This article will delve into the intricacies of bond strength, examining the roles of electronegativity, bond length, and other contributing elements to provide a comprehensive understanding.
Understanding Covalent Bonds: The Foundation
Before we compare polar and nonpolar covalent bonds, let's establish a solid understanding of what a covalent bond is. A covalent bond is a chemical bond formed by the sharing of one or more pairs of electrons between two atoms. This sharing allows each atom to achieve a more stable electron configuration, often resembling a noble gas. The strength of this bond is directly related to the strength of the attraction between the shared electrons and the positively charged nuclei of the atoms involved.
Nonpolar Covalent Bonds: Equal Sharing
In a nonpolar covalent bond, the electrons are shared equally between the two atoms. This typically occurs when the atoms involved have similar electronegativities. Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. When electronegativities are nearly identical, neither atom exerts a significantly stronger pull on the shared electrons, resulting in an even distribution. Examples of molecules with predominantly nonpolar covalent bonds include:
- H₂ (Hydrogen gas): Both hydrogen atoms have the same electronegativity.
- Cl₂ (Chlorine gas): Both chlorine atoms have the same electronegativity.
- O₂ (Oxygen gas): While technically possessing a slight polarity due to the molecular geometry, the electronegativity difference is minimal, leading to a largely nonpolar bond.
Polar Covalent Bonds: Unequal Sharing
In contrast, a polar covalent bond involves the unequal sharing of electrons. This occurs when there's a significant difference in electronegativity between the two atoms involved. The atom with the higher electronegativity attracts the shared electrons more strongly, creating a slightly negative charge (δ-) around that atom and a slightly positive charge (δ+) around the other atom. This creates a dipole moment, a measure of the separation of positive and negative charges within the molecule. Examples include:
- HCl (Hydrogen chloride): Chlorine is significantly more electronegative than hydrogen, resulting in a polar covalent bond.
- H₂O (Water): Oxygen is more electronegative than hydrogen, leading to polar covalent bonds between oxygen and each hydrogen atom.
- NH₃ (Ammonia): Nitrogen is more electronegative than hydrogen, creating polar covalent bonds.
Factors Affecting Bond Strength: Beyond Polarity
While polarity plays a role, it's crucial to understand that several other factors significantly influence bond strength:
1. Bond Length: The Distance Matters
Bond length is the average distance between the nuclei of two bonded atoms. Shorter bond lengths generally correspond to stronger bonds. This is because the closer the nuclei are, the stronger the electrostatic attraction between the positively charged nuclei and the negatively charged electrons. In general, smaller atoms form shorter bonds, leading to increased strength.
2. Number of Shared Electron Pairs: Multiple Bonds
The number of shared electron pairs also affects bond strength. Single bonds involve one shared electron pair, double bonds involve two shared pairs, and triple bonds involve three. Multiple bonds are generally stronger than single bonds because more electrons are shared, resulting in a stronger electrostatic attraction. For example, a triple bond in nitrogen gas (N₂) is significantly stronger than a single bond in hydrogen gas (H₂).
3. Atomic Size and Orbital Overlap: Effective Sharing
The size of the atoms involved and the degree of orbital overlap also play crucial roles. Smaller atoms with better orbital overlap form stronger bonds because the shared electrons are more effectively localized between the nuclei. This overlap allows for a greater concentration of electron density in the bonding region, enhancing the attractive forces.
4. Bond Order: A Comprehensive Measure
Bond order is the number of chemical bonds between a pair of atoms. It provides a quantitative measure of bond strength that considers the number of shared electron pairs. Higher bond orders indicate stronger bonds. For example, a triple bond (bond order = 3) is stronger than a double bond (bond order = 2), which in turn is stronger than a single bond (bond order = 1).
The Complex Relationship: Polarity and Strength
Now, let's address the central question more directly. While a polar bond can sometimes be stronger than a nonpolar bond of similar length and bond order, it's not a guaranteed outcome. Polarity introduces a dipole moment, which can contribute to stronger intermolecular forces (forces between molecules). However, the intramolecular force (within the bond itself) might not be stronger in every instance.
For example, a carbon-oxygen double bond (C=O) is polar due to the higher electronegativity of oxygen, and it's quite strong. However, a carbon-carbon triple bond (C≡C), while nonpolar, is also extremely strong due to the three shared electron pairs. In this case, the nonpolar triple bond might be stronger than some polar double bonds.
Conclusion: No Simple Answer, but Important Factors
In summary, there isn't a simple answer to whether polar covalent bonds are always stronger than nonpolar ones. The strength of a covalent bond depends on a complex interplay of factors:
- Bond length: Shorter bonds are generally stronger.
- Bond order: Higher bond order (more shared electron pairs) leads to stronger bonds.
- Atomic size and orbital overlap: Smaller atoms with better overlap form stronger bonds.
- Polarity: While it can contribute to stronger intermolecular forces, its effect on intramolecular bond strength is less straightforward.
To determine which bond is stronger, you must consider all these factors together for specific molecules involved. While polarity can be an important element, it's not the sole determinant of bond strength. Understanding the underlying factors allows for a more nuanced and accurate comparison. The strength of a bond is a testament to the intricate dance of electrostatic forces, electron sharing, and atomic properties at the heart of chemical bonding.
Latest Posts
Latest Posts
-
Are All Angles Of A Parallelogram Congruent
Apr 06, 2025
-
What Is Not A Symptom Of Foodborne Illness
Apr 06, 2025
-
How Many Food Chains Make Up The Food Web
Apr 06, 2025
-
69 1 Liquid Measurement Ratio Calculator
Apr 06, 2025
-
Elements That Are Good Conductors Of Heat And Electricity Are
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Are Polar Covalent Bonds Stronger Than Nonpolar . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
