What Is The Test For Hydrogen Gas Called
Juapaving
Apr 05, 2025 · 6 min read
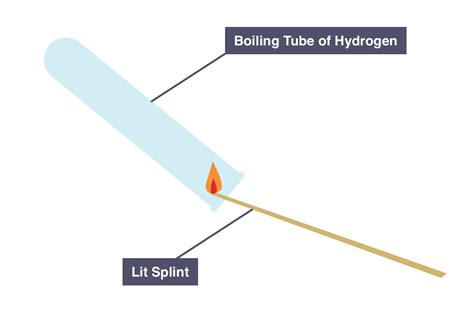
Table of Contents
What is the Test for Hydrogen Gas Called? A Comprehensive Guide
Hydrogen gas, a colorless, odorless, and highly flammable substance, requires specific tests for its detection and confirmation. While its properties make direct observation challenging, several reliable methods exist to identify its presence. Understanding these tests is crucial for safety in various industrial settings and scientific experiments where hydrogen gas might be involved. This article delves deep into the various tests used for hydrogen gas identification, explaining their principles, procedures, and limitations.
Common Tests for Hydrogen Gas: A Detailed Overview
Several tests are employed to detect hydrogen gas, each with its own advantages and disadvantages. The choice of test depends on factors like the expected concentration of hydrogen, the available equipment, and the desired level of accuracy.
1. The "Pop" Test (Burning Splint Test): A Simple Qualitative Test
This is arguably the most well-known and simplest test for hydrogen. It's a qualitative test, meaning it confirms the presence of hydrogen but doesn't quantify the amount.
Procedure:
- Collect a sample of the gas: Carefully collect a sample of the gas you suspect to be hydrogen in a clean, dry test tube or container.
- Approach a burning splint: Bring a lighted splint (a small wooden stick) close to the mouth of the container holding the gas sample.
- Observe the result: If the gas is hydrogen, a characteristic squeaky "pop" sound will be heard as the hydrogen ignites.
Principle:
Hydrogen gas is highly flammable. When mixed with air in the correct proportions, it forms an explosive mixture. The burning splint provides the ignition source, causing the hydrogen to combust rapidly, producing the characteristic "pop."
Limitations:
- Not quantitative: This test only confirms the presence of hydrogen, not its concentration.
- Safety concerns: The test involves handling a lit splint and potentially explosive mixtures. Appropriate safety precautions are paramount.
- Interference: Other flammable gases could also produce a similar "pop" sound, leading to false positives. Therefore, the "pop" test should only be considered preliminary and ideally should be followed up with confirmatory tests.
2. Palladium Chloride Test: A Sensitive and Specific Test
The palladium chloride test is a more sensitive and specific chemical test for hydrogen gas. It's based on the reducing properties of hydrogen.
Procedure:
- Prepare palladium chloride solution: A solution of palladium chloride (PdCl₂) is prepared.
- Expose to hydrogen: The solution is exposed to the gas sample suspected to contain hydrogen.
- Observe the change: If hydrogen is present, it will reduce the palladium chloride, resulting in the formation of metallic palladium, which appears as a black precipitate.
Principle:
Hydrogen acts as a reducing agent, causing the reduction of palladium(II) chloride (PdCl₂) to metallic palladium (Pd), which is insoluble and forms a black precipitate. The reaction can be represented as:
PdCl₂ + H₂ → Pd + 2HCl
Advantages:
- Higher sensitivity: Compared to the "pop" test, it's more sensitive and can detect lower concentrations of hydrogen.
- Specificity: It's more specific to hydrogen than the "pop" test, reducing the chances of false positives.
Limitations:
- Interference: Other reducing agents can interfere with this test, leading to false positives. Careful control experiments are necessary.
- Requires specialized reagents: The test requires the use of palladium chloride, a specialized reagent.
3. Gas Chromatography (GC): A Quantitative Analysis Method
Gas chromatography is a powerful analytical technique used to separate and quantify the components of a gaseous mixture. It's a highly accurate and quantitative method for determining the concentration of hydrogen in a gas sample.
Procedure:
- Sample injection: A sample of the gas mixture is injected into the gas chromatograph.
- Separation: The components of the mixture are separated based on their different affinities for a stationary phase within a column.
- Detection: A detector (often a thermal conductivity detector or flame ionization detector) measures the amount of each component eluting from the column.
- Quantification: The area under the peak corresponding to hydrogen is used to determine its concentration.
Principle:
Gas chromatography separates the components of a gas mixture based on their different boiling points and interactions with the stationary phase. The detector then measures the amount of each component, providing a quantitative analysis.
Advantages:
- High accuracy and precision: GC provides highly accurate and precise measurements of hydrogen concentration.
- Quantitative analysis: It's a quantitative method, providing the exact concentration of hydrogen in the sample.
- Multi-component analysis: GC can simultaneously analyze multiple components in a gas mixture, not just hydrogen.
Limitations:
- Expensive equipment: GC requires specialized and relatively expensive equipment.
- Requires skilled operator: Proper operation and data interpretation require training and expertise.
- Sample preparation: Sample preparation might be required depending on the matrix.
4. Electrochemical Sensors: Real-time Monitoring
Electrochemical sensors, often based on metal oxide semiconductors or solid-state electrochemical cells, provide a real-time, continuous monitoring capability for hydrogen gas.
Principle:
These sensors work by detecting the change in electrical conductivity or potential caused by the interaction of hydrogen with the sensing material. The change in electrical signal is directly proportional to the concentration of hydrogen.
Advantages:
- Real-time monitoring: These sensors can continuously monitor hydrogen levels.
- Compact and portable: Many electrochemical sensors are compact and portable, suitable for various applications.
Limitations:
- Sensitivity and range: The sensitivity and measurement range vary depending on the specific sensor.
- Drift and lifespan: Sensor readings can drift over time, requiring calibration. The sensors also have a limited lifespan.
- Interference: Other gases might interfere with the sensor’s response.
Choosing the Right Test: Factors to Consider
The selection of the appropriate hydrogen gas test depends on several factors:
- Required sensitivity: For high sensitivity, GC or palladium chloride tests are preferred. For simple detection, the "pop" test might suffice.
- Accuracy and precision: GC offers the highest accuracy and precision.
- Cost: The "pop" test is the least expensive, while GC is the most expensive.
- Availability of equipment: The "pop" test requires minimal equipment, while GC requires specialized instrumentation.
- Required speed: Electrochemical sensors provide real-time monitoring, while GC requires some time for analysis.
- Safety: All tests involving hydrogen should be conducted with appropriate safety precautions.
Safety Precautions when Testing for Hydrogen Gas
Hydrogen is highly flammable and can form explosive mixtures with air. Therefore, all tests involving hydrogen gas must be conducted with utmost care:
- Proper ventilation: Ensure adequate ventilation to prevent the accumulation of hydrogen gas.
- No open flames: Avoid open flames near the test area.
- Grounding: Ground all equipment to prevent static electricity discharge.
- Personal protective equipment (PPE): Wear appropriate PPE, including safety glasses and gloves.
- Emergency procedures: Have emergency procedures in place in case of a fire or explosion.
Conclusion: A Multifaceted Approach to Hydrogen Detection
The detection and quantification of hydrogen gas require a multifaceted approach. While the simple "pop" test serves as a preliminary indicator, more sophisticated techniques such as palladium chloride tests, gas chromatography, and electrochemical sensors are crucial for accurate and quantitative analysis. The choice of method depends on the specific application, available resources, and required level of accuracy. Regardless of the chosen method, safety remains paramount, emphasizing the importance of proper training and adhering to strict safety protocols when working with hydrogen gas. Understanding these tests and their limitations empowers safe and efficient handling of hydrogen in various scientific and industrial contexts.
Latest Posts
Latest Posts
-
The Lcm Of 6 And 10
Apr 06, 2025
-
Type Of Energy Transformed By A Toaster Into Thermal Energy
Apr 06, 2025
-
Are All Angles Of A Parallelogram Congruent
Apr 06, 2025
-
What Is Not A Symptom Of Foodborne Illness
Apr 06, 2025
-
How Many Food Chains Make Up The Food Web
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Is The Test For Hydrogen Gas Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
