Where Are The Metal Located On The Periodic Table
Juapaving
Apr 02, 2025 · 5 min read
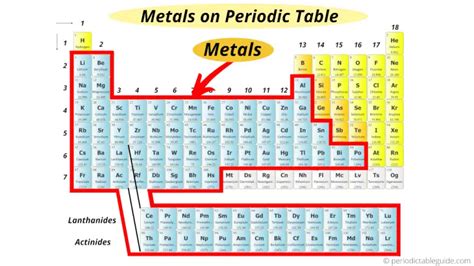
Table of Contents
Where Are the Metals Located on the Periodic Table? A Comprehensive Guide
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding its layout is crucial for predicting an element's behavior, including its metallic character. This comprehensive guide delves into the location of metals on the periodic table, exploring their characteristics and exceptions to the general rules.
The Broad Strokes: Metals Dominate the Table
Metals constitute the vast majority of elements on the periodic table. They're predominantly found on the left-hand side and towards the center, forming a large, roughly stair-step shaped block. This isn't a hard and fast line, however, with some elements exhibiting properties that blur the lines between metals and nonmetals. This transition zone is where the fascinating world of metalloids resides.
Identifying the Metal Zone: A Visual Guide
Imagine drawing a diagonal line from Boron (B) to Astatine (At). While not perfectly precise, this line generally separates metals from nonmetals. Elements to the left and below this line are primarily metals. This includes the alkali metals, alkaline earth metals, transition metals, lanthanides, actinides, and post-transition metals. Elements to the right and above the line are generally nonmetals, while those directly on the line are metalloids, demonstrating a blend of metallic and nonmetallic characteristics.
Key Metal Groups on the Periodic Table
Let's examine the major groups of metals, highlighting their locations and key properties:
1. Alkali Metals (Group 1): The Highly Reactive Family
Located in the first column of the periodic table, alkali metals are highly reactive, soft metals with low melting points. Their reactivity stems from their single valence electron, readily donated to form +1 ions. This high reactivity makes them extremely unstable in their pure form and necessitates storage under oil or inert atmospheres. Examples include Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Cesium (Cs), and Francium (Fr).
2. Alkaline Earth Metals (Group 2): Reactive, but Less So
Residing in the second column, alkaline earth metals are also reactive metals, though less so than alkali metals. They have two valence electrons, readily forming +2 ions. They are generally harder, denser, and have higher melting points than alkali metals. Examples include Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba), and Radium (Ra).
3. Transition Metals (Groups 3-12): The Diverse Group
Occupying the central block of the periodic table (groups 3-12), transition metals are characterized by their partially filled d orbitals. This leads to a diverse range of oxidation states and complex ion formation. They are known for their strength, high melting and boiling points, and excellent conductivity. Examples include Iron (Fe), Copper (Cu), Gold (Au), Platinum (Pt), and many others. This group is essential for various industrial applications and biological processes.
4. Post-Transition Metals (Groups 13-15): Bridging the Gap
Located adjacent to the transition metals, post-transition metals exhibit properties intermediate between transition and nonmetals. They are relatively soft, with lower melting points than transition metals. Their reactivity varies significantly across the group. Examples include Aluminum (Al), Tin (Sn), and Lead (Pb).
5. Lanthanides and Actinides: The Inner Transition Metals
These two series of elements, collectively known as the inner transition metals, are located at the bottom of the periodic table. They are often placed separately to maintain the table's compactness. Lanthanides (filling the 4f orbitals) and actinides (filling the 5f orbitals) are characterized by similar chemical properties within their respective series. Many are radioactive, particularly the actinides.
Beyond the Simple Classification: Exceptions and Gray Areas
While the general trend is clear, several exceptions and nuanced classifications exist:
-
Metalloids: These elements, such as Silicon (Si), Germanium (Ge), and Arsenic (As), lie along the metalloid staircase. They exhibit properties of both metals and nonmetals, making them crucial in semiconductor technology. Their position is crucial to understanding the gradual transition in properties across the periodic table.
-
Amphoteric Metals: Some metals, like Aluminum (Al) and Zinc (Zn), display amphoteric behavior. They can react with both acids and bases, showcasing a unique dual reactivity not common in all metals.
-
Noble Metals: Elements like Gold (Au), Silver (Ag), and Platinum (Pt) are known as noble metals due to their high resistance to corrosion and oxidation. While still metallic, their low reactivity distinguishes them from other metals.
The Importance of Understanding Metal Location
Understanding the location of metals on the periodic table is essential for several reasons:
-
Predicting Properties: The position of an element on the table provides valuable insights into its physical and chemical properties, including reactivity, conductivity, and melting point.
-
Understanding Chemical Reactions: Knowing the metallic character of an element helps predict how it will react with other elements and compounds, guiding chemical synthesis and analysis.
-
Material Science Applications: The properties of metals are crucial for developing new materials with specific characteristics, such as strength, conductivity, and resistance to corrosion. Understanding their location on the periodic table is critical for designing and engineering materials.
-
Biological Significance: Many metals are essential for biological processes, acting as cofactors in enzymes and playing crucial roles in cellular function. Knowledge of their properties helps understand their biological roles.
Conclusion: The Periodic Table – A Map to Metallic Properties
The periodic table provides a powerful framework for understanding the properties of elements, particularly metals. While a simplified view positions metals predominantly on the left and center, nuanced considerations like metalloids and amphoteric metals highlight the complexities of elemental classification. Mastering the relationships between location and properties empowers deeper understanding in chemistry, materials science, and various other fields. Further exploration into the electron configurations, oxidation states, and bonding characteristics of metals further elucidates their diverse properties and applications. The periodic table, in essence, serves as a comprehensive roadmap, guiding our understanding of the diverse and fascinating world of metallic elements.
Latest Posts
Latest Posts
-
The Pectoral Girdle Consists Of The
Apr 03, 2025
-
Find The Unit Vector In The Direction Of V
Apr 03, 2025
-
5 Letter Words Starting With Ho
Apr 03, 2025
-
Partial Fraction Decomposition Calculator Step By Step
Apr 03, 2025
-
5 Letter Words Starting With Har
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Where Are The Metal Located On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
