What Is The Electron Configuration For Aluminum
Juapaving
Apr 05, 2025 · 5 min read
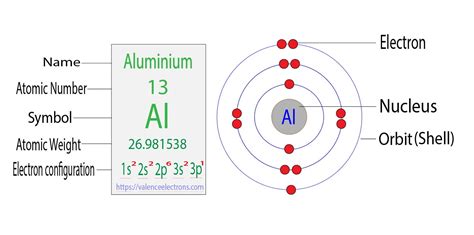
Table of Contents
What is the Electron Configuration for Aluminum? A Deep Dive into Atomic Structure
Aluminum, a ubiquitous metal found in everything from soda cans to airplanes, boasts a fascinating atomic structure that dictates its unique properties. Understanding its electron configuration is key to unlocking this fascinating world. This comprehensive guide will explore the electron configuration of aluminum, delve into the principles behind it, and examine its implications for the element's chemical behavior.
Understanding Electron Configuration
Before diving into aluminum's specific configuration, let's establish the foundational principles. Electron configuration describes the arrangement of electrons in the various energy levels and sublevels within an atom. This arrangement follows specific rules dictated by quantum mechanics, ensuring stability and reactivity. The electrons occupy orbitals, regions of space around the nucleus where there's a high probability of finding an electron.
Key Principles Governing Electron Configuration:
- Aufbau Principle: Electrons fill orbitals starting from the lowest energy level and progressing upwards. Think of it like filling a building from the ground floor up.
- Pauli Exclusion Principle: Each orbital can hold a maximum of two electrons, each with opposite spins (represented as ↑ and ↓). This is like having two people occupy a single apartment, but they must have different characteristics.
- Hund's Rule: When filling orbitals of equal energy (degenerate orbitals), electrons initially occupy each orbital singly before pairing up. Imagine filling seats on a bus – individuals will spread out before doubling up.
These principles are crucial in predicting the electron configuration of any element, including aluminum.
Determining the Electron Configuration of Aluminum (Al)
Aluminum's atomic number is 13, meaning it has 13 protons and 13 electrons in a neutral atom. To determine its electron configuration, we systematically fill the orbitals according to the Aufbau principle, Pauli exclusion principle, and Hund's rule.
Step-by-Step Configuration:
-
First Energy Level (n=1): This level contains only the 1s subshell, which can hold a maximum of two electrons. Therefore, we fill it completely: 1s².
-
Second Energy Level (n=2): This level contains the 2s and 2p subshells. The 2s subshell holds two electrons (2s²), leaving us with 9 more electrons to place. The 2p subshell has three orbitals, each capable of holding two electrons, for a total of six electrons. We fill these completely: 2p⁶.
-
Third Energy Level (n=3): We have three electrons remaining. The third energy level starts with the 3s subshell, which can hold two electrons (3s²). This leaves one electron for the 3p subshell. We place this electron in one of the three 3p orbitals: 3p¹.
The Complete Electron Configuration of Aluminum:
Therefore, the complete electron configuration for aluminum is 1s²2s²2p⁶3s²3p¹. This concise notation signifies the distribution of aluminum's 13 electrons across its various energy levels and sublevels.
Orbital Diagrams and Electron Configuration
While the electron configuration notation is concise, orbital diagrams provide a more visual representation of electron placement within the orbitals. Each orbital is represented by a box, and electrons are represented by arrows (↑ and ↓).
Aluminum's Orbital Diagram:
1s: ↑↓
2s: ↑↓
2p: ↑↓ ↑↓ ↑↓
3s: ↑↓
3p: ↑ _ _
This diagram visually confirms the filling of orbitals according to Hund's rule, with the single electron in the 3p subshell occupying an orbital by itself before pairing.
Aluminum's Valence Electrons and Chemical Behavior
The valence electrons are the electrons in the outermost energy level, which are responsible for an atom's chemical reactivity. In aluminum's case, the valence electrons are located in the third energy level (3s²3p¹), totaling three valence electrons.
Aluminum's three valence electrons readily participate in chemical bonding, explaining its characteristic properties:
-
Reactivity: Aluminum readily loses its three valence electrons to achieve a stable octet configuration, resulting in a 3+ oxidation state (Al³⁺). This explains its high reactivity with many elements, particularly oxygen and halogens.
-
Conductivity: The loosely held valence electrons contribute to aluminum's excellent electrical and thermal conductivity. These electrons are mobile and can easily carry charge and energy.
-
Alloy Formation: Aluminum's ability to readily bond with other metals makes it a valuable component in various alloys, enhancing strength and other desirable properties.
Variations and Exceptions in Electron Configurations
While the Aufbau principle provides a generally accurate prediction of electron configurations, certain exceptions exist due to subtle energy level variations and electron-electron interactions. These exceptions primarily occur in transition metals and lanthanides/actinides where the energy differences between subshells are smaller. However, Aluminum's electron configuration follows the standard Aufbau principle without any exceptions.
Applications and Importance of Understanding Aluminum's Electron Configuration
Understanding aluminum's electron configuration is crucial for several reasons:
-
Predicting Chemical Reactions: Knowing its three valence electrons allows us to predict its chemical behavior, reactivity, and bonding patterns with other elements.
-
Material Science: The configuration explains aluminum's unique properties, making it essential for designing alloys and materials with specific characteristics.
-
Catalysis: Aluminum compounds play a significant role in catalysis, and the understanding of electron configuration helps in designing and optimizing catalytic processes.
Conclusion: A Deeper Understanding of Aluminum
The electron configuration of aluminum, 1s²2s²2p⁶3s²3p¹, is a fundamental aspect of its atomic structure and chemical behavior. By understanding the principles governing electron configuration and the specific arrangement of aluminum's electrons, we gain insight into its reactivity, conductivity, and its extensive applications in diverse fields. This comprehensive overview provides a solid foundation for further exploration of aluminum's chemistry and material properties. The study of electron configurations is not just an academic exercise; it's a key to unlocking the secrets of matter and its interactions, providing a foundation for innovation and technological advancement.
Latest Posts
Latest Posts
-
Why Activation Energy Is Not Affected By Temperature
Apr 06, 2025
-
Whats The Square Root Of 196
Apr 06, 2025
-
What Is The Least Common Multiple Of 2 And 9
Apr 06, 2025
-
How Do You Find The Numerator
Apr 06, 2025
-
What Is The Lowest Common Multiple Of 3 And 4
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Is The Electron Configuration For Aluminum . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
