What Is The Charge Of Sodium
Juapaving
Apr 02, 2025 · 5 min read
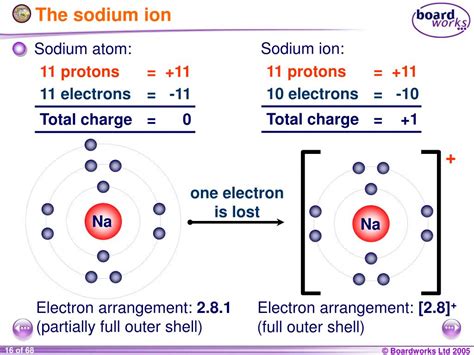
Table of Contents
What is the Charge of Sodium? Understanding Sodium Ions and Their Importance
Sodium, a ubiquitous element in our lives, plays a crucial role in various biological and chemical processes. Understanding its charge, specifically its ionic charge, is essential to grasping its behavior and function. This comprehensive article delves into the intricacies of sodium's charge, exploring its atomic structure, ionic formation, and significance in various contexts.
The Atomic Structure of Sodium and its Tendency to Ionize
Sodium (Na), an alkali metal found in Group 1 of the periodic table, possesses 11 electrons orbiting its nucleus. These electrons are arranged in specific energy levels or shells: two electrons in the first shell, eight in the second, and a single electron in the outermost, or valence, shell. This lone valence electron is relatively loosely bound to the atom.
This single valence electron is key to understanding sodium's charge. Atoms strive for stability, usually achieved by having a full outermost electron shell. For sodium, achieving this full outer shell is far easier by losing its single valence electron than by gaining seven more.
The Formation of Sodium Ions (Na⁺)
The process of sodium losing its valence electron is called ionization. When sodium loses this electron, it becomes a cation, specifically a sodium ion (Na⁺). The loss of a negatively charged electron leaves the sodium atom with one more proton (positively charged) than electrons, resulting in a net positive charge of +1. This positive charge is what defines the charge of sodium in its ionic form.
Key takeaway: Sodium doesn't inherently have a positive charge; it acquires a positive charge through the loss of an electron to achieve a stable electron configuration.
The Significance of the +1 Charge of Sodium Ions
The +1 charge of sodium ions significantly influences their chemical and biological behavior. This charge allows sodium ions to participate in a wide range of interactions, including:
1. Ionic Bonding: The Foundation of Many Compounds
The positive charge of the sodium ion (Na⁺) allows it to form strong ionic bonds with negatively charged ions (anions). A classic example is sodium chloride (NaCl), or common table salt. The electrostatic attraction between the positively charged sodium ion and the negatively charged chloride ion (Cl⁻) forms the crystalline structure of salt. This principle applies to many other sodium compounds.
2. Electrolyte Balance: Crucial for Biological Processes
Sodium ions play a vital role in maintaining electrolyte balance in biological systems. Electrolytes are minerals that carry an electric charge when dissolved in fluids, such as blood and bodily fluids. The precise balance of sodium ions is critical for:
- Nerve impulse transmission: The movement of sodium ions across cell membranes is crucial for the transmission of nerve impulses. Changes in sodium ion concentration trigger electrical signals that allow communication between nerve cells.
- Muscle contraction: Similar to nerve impulse transmission, sodium ion movement is essential for muscle contraction. The influx of sodium ions into muscle cells initiates the process of muscle fiber shortening.
- Fluid balance: Sodium ions help regulate the balance of water in the body. They contribute to osmotic pressure, influencing the movement of water across cell membranes. Maintaining proper fluid balance is vital for various physiological functions.
- Blood pressure regulation: Sodium levels are closely linked to blood pressure. High sodium intake can lead to increased blood volume and consequently, higher blood pressure.
3. Chemical Reactions: Participating in Reactions
The +1 charge also allows sodium ions to participate in various chemical reactions. Its charge enables it to interact with other charged species, acting as a reactant or catalyst in numerous processes, both in industrial and biological settings.
4. Industrial Applications: Leveraging Sodium's Properties
Sodium's properties, largely influenced by its ionic charge, find extensive use in various industrial applications, including:
- Sodium lamps: Sodium vapor lamps emit a characteristic yellow-orange light, which is efficient and widely used in street lighting.
- Sodium hydroxide (NaOH) production: Sodium hydroxide, a strong base, is a crucial industrial chemical used in various applications, from soap making to paper production. Its production involves the interaction of sodium with water.
- Sodium metal in chemical synthesis: Sodium metal, although reactive, finds use in certain organic synthesis reactions, acting as a reducing agent.
Understanding the Difference Between Sodium Atom and Sodium Ion
It's vital to distinguish between the neutral sodium atom (Na) and the sodium ion (Na⁺). The sodium atom, with its 11 electrons and 11 protons, is electrically neutral. The sodium ion, having lost one electron, carries a net positive charge of +1. This difference in charge drastically alters its chemical reactivity and biological function.
The sodium atom is highly reactive, readily losing its single valence electron to achieve stability. The sodium ion, on the other hand, is relatively stable due to its full outer electron shell. This stability is what allows sodium ions to play the crucial roles outlined above.
Sodium's Role in Human Health: A Delicate Balance
Maintaining the right balance of sodium in the body is crucial for overall health. While sodium is essential for various physiological processes, excessive sodium intake can lead to several health problems, including:
- High blood pressure (hypertension): Excess sodium can lead to fluid retention, increasing blood volume and consequently, blood pressure. Hypertension is a major risk factor for heart disease, stroke, and kidney disease.
- Heart disease: High blood pressure, directly linked to excessive sodium intake, significantly increases the risk of developing heart disease.
- Stroke: High blood pressure damages blood vessels, increasing the risk of stroke.
- Kidney disease: The kidneys play a vital role in regulating sodium levels. High sodium intake can strain the kidneys, potentially leading to kidney damage and dysfunction.
- Osteoporosis: High sodium intake can increase calcium excretion in urine, potentially contributing to bone loss and osteoporosis.
Conclusion: The Ubiquitous and Vital +1 Charge of Sodium
The +1 charge of the sodium ion is not simply a number; it's a fundamental characteristic that dictates its chemical and biological behavior. This positive charge is responsible for its participation in ionic bonds, its crucial role in electrolyte balance, its involvement in various chemical reactions, and its widespread industrial applications. Understanding this charge is essential for appreciating the significance of sodium in both the natural world and our daily lives. Maintaining a balanced sodium intake is critical for preserving overall health and preventing various health complications. Further research continues to unravel the intricate complexities of sodium's behavior and its impact on biological systems, underscoring the importance of continued exploration in this field.
Latest Posts
Latest Posts
-
Identify The Structure Indicated By The Lines
Apr 03, 2025
-
How To Find The Perimeter Of A Hexagon
Apr 03, 2025
-
This Is The Functional Unit Of The Kidney
Apr 03, 2025
-
Four Kingdoms Of The Domain Eukarya
Apr 03, 2025
-
Do Metals Lose Or Gain Electrons
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Charge Of Sodium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
