Do Metals Lose Or Gain Electrons
Juapaving
Apr 03, 2025 · 5 min read
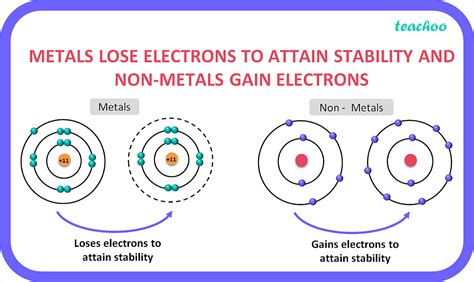
Table of Contents
Do Metals Lose or Gain Electrons? Understanding Metallic Properties and Reactivity
The question of whether metals lose or gain electrons is fundamental to understanding their chemical behavior and properties. The short answer is that metals generally lose electrons to form positive ions (cations). This electron loss is what defines their metallic character and drives many of their characteristic reactions. However, the complexity of electron behavior in metals goes far beyond this simple statement, encompassing concepts like metallic bonding, oxidation states, and exceptions to the rule. This article delves deep into the intricacies of electron transfer in metals, exploring the underlying principles and examining some key examples.
The Nature of Metallic Bonding
To fully grasp why metals lose electrons, we need to understand the nature of metallic bonding. Unlike ionic or covalent bonds, where electrons are shared or transferred completely between atoms, metallic bonding involves a "sea" of delocalized electrons. This means that the valence electrons (outermost electrons) of metal atoms are not associated with any single atom but rather are free to move throughout the entire metal lattice.
Delocalized Electrons and Metallic Properties
This "sea" of delocalized electrons is responsible for several characteristic properties of metals, including:
- Electrical conductivity: The free movement of electrons allows for the easy flow of electric current.
- Thermal conductivity: The delocalized electrons readily transfer thermal energy throughout the metal.
- Malleability and ductility: The non-directional nature of metallic bonding allows metal atoms to slide past each other without disrupting the overall structure, enabling metals to be hammered into sheets (malleability) or drawn into wires (ductility).
- Metallic luster: The delocalized electrons interact with light, causing the reflection of light waves and imparting the characteristic shiny appearance of metals.
Ionization Energy and Electron Loss
The tendency of metals to lose electrons is directly related to their relatively low ionization energies. Ionization energy is the energy required to remove an electron from an atom or ion in its gaseous state. Metals have low ionization energies because their valence electrons are relatively loosely held by the nucleus. This is due to several factors, including:
- Shielding effect: The inner electrons shield the outer electrons from the full positive charge of the nucleus, reducing the attraction between the nucleus and valence electrons.
- Atomic radius: Metals generally have larger atomic radii compared to non-metals, increasing the distance between the nucleus and valence electrons, thus weakening the electrostatic attraction.
The lower the ionization energy, the easier it is to remove an electron, making the metal more prone to losing electrons and forming positive ions. This electron loss is a crucial aspect of many chemical reactions involving metals.
Oxidation States and Electron Loss
When metals lose electrons, they form positive ions, with the charge of the ion representing the number of electrons lost. This charge is often expressed as the oxidation state of the metal. For example, when sodium (Na) loses one electron, it forms a sodium ion (Na⁺) with an oxidation state of +1. Similarly, magnesium (Mg) loses two electrons to form Mg²⁺ with an oxidation state of +2.
The most common oxidation state for a metal is determined by the number of valence electrons it possesses. Group 1 metals (alkali metals) typically have an oxidation state of +1, Group 2 metals (alkaline earth metals) have an oxidation state of +2, and so on. However, transition metals can exhibit multiple oxidation states due to the involvement of electrons from both the s and d orbitals.
Exceptions and Complexities
While the general rule is that metals lose electrons, there are some exceptions and complexities to consider.
Transition Metals and Variable Oxidation States
Transition metals are well-known for their ability to exhibit multiple oxidation states. This is because the electrons in their d orbitals are relatively close in energy, making it possible for them to lose different numbers of electrons depending on the specific reaction conditions. For example, iron (Fe) can have oxidation states of +2 (ferrous) and +3 (ferric). This variability in oxidation states contributes to the wide range of compounds and complexes formed by transition metals.
Organometallic Compounds
In organometallic compounds, metals can form bonds with carbon-containing ligands. In some cases, these bonds involve the metal gaining electrons, resulting in a negative oxidation state for the metal. This is a departure from the typical electron-losing behavior observed in simple metal compounds.
Alloy Formation
In alloys, which are mixtures of two or more metals, the electron transfer is less straightforward. The delocalized electrons are shared among the different metal atoms, creating a complex system of electron interactions that cannot be simply described as electron loss or gain.
Examples of Metals Losing Electrons
Let's look at some specific examples illustrating metal electron loss:
1. Reaction of Sodium with Chlorine:
Sodium (Na), an alkali metal, readily loses one electron to form Na⁺, while chlorine (Cl), a non-metal, gains one electron to form Cl⁻. This electron transfer results in the formation of sodium chloride (NaCl), a common table salt:
2Na(s) + Cl₂(g) → 2NaCl(s)
2. Corrosion of Iron:
Iron (Fe) corrodes in the presence of oxygen and water, losing electrons to form Fe²⁺ or Fe³⁺ ions. This process, known as rusting, involves a complex electrochemical reaction where iron acts as an anode, losing electrons, and oxygen acts as a cathode, gaining electrons:
4Fe(s) + 3O₂(g) + 6H₂O(l) → 4Fe(OH)₃(s)
3. Displacement Reactions:
More reactive metals can displace less reactive metals from their compounds. For example, zinc (Zn) can displace copper (Cu) from copper(II) sulfate solution:
Zn(s) + CuSO₄(aq) → ZnSO₄(aq) + Cu(s)
In this reaction, zinc loses electrons to form Zn²⁺, while copper(II) ions (Cu²⁺) gain electrons to form copper metal (Cu).
Conclusion
In summary, while metals generally lose electrons to form positive ions, the complexity of electron behavior in metals extends beyond this simple generalization. The concepts of metallic bonding, ionization energy, oxidation states, and the existence of exceptions like transition metal complexes and organometallic compounds paint a more nuanced picture of electron transfer in metals. Understanding these concepts is crucial to comprehending the diverse chemical and physical properties that make metals such an important class of materials. The study of electron behavior in metals continues to be a vibrant area of research, with ongoing efforts to explore the subtle interactions and emergent properties arising from the dynamic interactions of electrons within metallic structures. Further research in areas such as advanced materials science and nanotechnology will undoubtedly reveal even greater complexities and applications of these fundamental principles.
Latest Posts
Latest Posts
-
What Is 32 Inches In Feet
Apr 04, 2025
-
5 Letter Words Starting With Ac
Apr 04, 2025
-
How Long Is 120 Minutes In Hours
Apr 04, 2025
-
What Do I Multiply Do Get 216
Apr 04, 2025
-
What Is A Horizontal Row On The Periodic Table Called
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Do Metals Lose Or Gain Electrons . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
