What Are The Chemicals In A Battery
Juapaving
Apr 02, 2025 · 6 min read
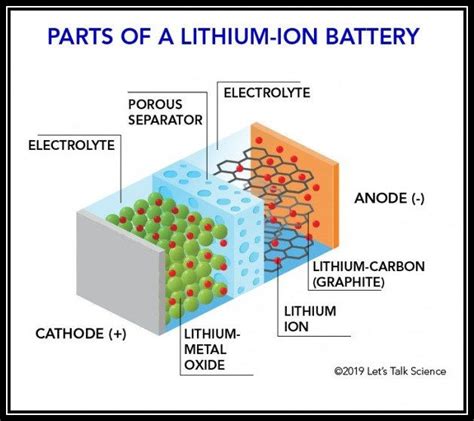
Table of Contents
What are the Chemicals in a Battery? A Deep Dive into Battery Chemistry
Batteries power our modern world, from smartphones and laptops to electric vehicles and grid-scale energy storage. But what exactly is inside these ubiquitous power sources? Understanding the chemical makeup of batteries is crucial to appreciating their functionality, limitations, and environmental impact. This comprehensive guide delves into the diverse chemistry of different battery types, explaining the roles of each component and the ongoing research into safer and more efficient alternatives.
The Fundamentals of Battery Chemistry: Anodes, Cathodes, and Electrolytes
All batteries, regardless of type, share fundamental components:
- Anode: The negative electrode. In a discharging battery, this is where oxidation occurs – the anode loses electrons.
- Cathode: The positive electrode. In a discharging battery, this is where reduction occurs – the cathode gains electrons.
- Electrolyte: The medium that allows ions (charged atoms) to move between the anode and cathode, completing the electrical circuit. This can be a liquid, a gel, or even a solid.
- Separator: A porous membrane that prevents the anode and cathode from directly contacting each other, which would cause a short circuit.
The specific chemical composition of these components dramatically impacts the battery's performance characteristics, including energy density, voltage, lifespan, and safety.
Common Battery Chemistries: A Detailed Look
Let's explore the chemical compositions of some prevalent battery types:
1. Alkaline Batteries (e.g., AA, AAA)
These are the ubiquitous primary (single-use) batteries found in many household devices. They are relatively inexpensive and readily available but have a lower energy density compared to rechargeable options. The chemistry is straightforward:
- Anode: Zinc (Zn) powder. Zinc undergoes oxidation, losing electrons and forming zinc oxide (ZnO).
- Cathode: Manganese dioxide (MnO₂) powder. Manganese dioxide accepts electrons and is reduced to manganese(III) oxide (Mn₂O₃).
- Electrolyte: Potassium hydroxide (KOH) solution – an alkaline solution that allows the movement of potassium ions (K⁺) between the electrodes.
Chemical Reaction (Simplified): Zn + 2MnO₂ → ZnO + Mn₂O₃
2. Lithium-ion Batteries (Li-ion)
Li-ion batteries dominate the rechargeable battery market, powering everything from smartphones to electric vehicles. Their high energy density, relatively long lifespan, and relatively low self-discharge rate make them highly desirable. However, the specific chemistry varies depending on the cathode material:
a) Lithium Cobalt Oxide (LiCoO₂) Cathode Li-ion Batteries
This is a common type of Li-ion battery known for its high energy density but also its relatively short lifespan and safety concerns (potential for thermal runaway).
- Anode: Graphite (C). Lithium ions (Li⁺) intercalate (insert between layers) into the graphite structure during discharge.
- Cathode: Lithium cobalt oxide (LiCoO₂). Lithium ions de-intercalate from LiCoO₂ during discharge, leaving behind cobalt oxide (CoO₂).
- Electrolyte: A lithium salt (e.g., lithium hexafluorophosphate, LiPF₆) dissolved in an organic solvent (e.g., ethylene carbonate, dimethyl carbonate).
Chemical Reaction (Simplified): LiC₆ + LiCoO₂ ⇌ 6C + Li₁₋ₓCoO₂ + Li⁺ + e⁻
b) Lithium Manganese Oxide (LiMn₂O₄) Cathode Li-ion Batteries
LiMn₂O₄ cathodes offer a lower cost and improved thermal stability compared to LiCoO₂, but their energy density is slightly lower.
- Anode: Similar to LiCoO₂ batteries, typically graphite.
- Cathode: Lithium manganese oxide (LiMn₂O₄).
- Electrolyte: Similar to LiCoO₂ batteries, a lithium salt in an organic solvent.
c) Lithium Iron Phosphate (LiFePO₄) Cathode Li-ion Batteries
LiFePO₄ cathodes are known for their excellent safety, long lifespan, and thermal stability. However, they have a lower energy density than LiCoO₂ and LiMn₂O₄.
- Anode: Graphite.
- Cathode: Lithium iron phosphate (LiFePO₄).
- Electrolyte: Similar to other Li-ion batteries.
d) Nickel Manganese Cobalt (NMC) Cathode Li-ion Batteries
NMC batteries are a blend of nickel, manganese, and cobalt oxides. The ratios of these metals can be adjusted to optimize performance characteristics. They offer a balance between energy density, lifespan, and cost.
- Anode: Graphite.
- Cathode: Nickel manganese cobalt oxide (NMC), with varying ratios of Ni, Mn, and Co. (e.g., NMC 111, NMC 523, NMC 811).
- Electrolyte: Similar to other Li-ion batteries.
3. Lead-Acid Batteries
These are a mature technology used in automotive applications and backup power systems. They are relatively inexpensive and robust but have a lower energy density than Li-ion batteries.
- Anode: Lead (Pb) sponge. Lead is oxidized to lead sulfate (PbSO₄).
- Cathode: Lead dioxide (PbO₂). Lead dioxide is reduced to lead sulfate (PbSO₄).
- Electrolyte: Sulfuric acid (H₂SO₄) solution. Sulfuric acid is consumed during discharge and regenerated during charging.
Chemical Reaction (Simplified): Pb + PbO₂ + 2H₂SO₄ ⇌ 2PbSO₄ + 2H₂O
4. Nickel-Metal Hydride (NiMH) Batteries
NiMH batteries are rechargeable and offer a higher energy density than alkaline batteries but lower than Li-ion batteries. They are less prone to the memory effect than older Nickel-Cadmium (NiCd) batteries.
- Anode: Hydrogen-absorbing alloy (e.g., LaNi₅). Hydrogen atoms are released during discharge.
- Cathode: Nickel hydroxide (Ni(OH)₂). Nickel hydroxide is reduced to nickel oxyhydroxide (NiOOH).
- Electrolyte: Potassium hydroxide (KOH) solution.
Chemical Reaction (Simplified): MH + NiOOH ⇌ M + Ni(OH)₂
Beyond the Basics: Additives and Other Components
The core chemical components discussed above represent the essential building blocks of batteries. However, many other additives and components play critical roles:
- Binders: These materials hold the active materials (anode and cathode) together, maintaining structural integrity. Common binders include polyvinylidene fluoride (PVDF) and styrene-butadiene rubber (SBR).
- Conductive Additives: These enhance the electron conductivity within the electrodes. Common examples include carbon black and graphite.
- Current Collectors: These are typically metal foils (e.g., aluminum or copper) that provide electrical contact to the electrodes.
- Additives to Electrolytes: Various additives modify the electrolyte's properties, such as improving its ionic conductivity, stability, and safety.
Environmental Considerations and Recycling
The chemical composition of batteries plays a significant role in their environmental impact. Some battery components, such as heavy metals (e.g., lead, cadmium, cobalt), are toxic and require careful handling and disposal. Recycling is crucial to recover valuable materials and minimize environmental damage. Research into environmentally friendly battery chemistries (e.g., those using abundant and less toxic materials) is an active area of research.
The Future of Battery Chemistry: Innovations and Research
The quest for better batteries is relentless. Ongoing research focuses on:
- Solid-state batteries: Replacing liquid or gel electrolytes with solid electrolytes, enhancing safety and potentially energy density.
- Lithium-sulfur batteries: Utilizing sulfur as a cathode material, offering potentially very high energy density.
- Sodium-ion batteries: Exploring sodium as an alternative to lithium, leveraging its abundance and lower cost.
- Magnesium-ion batteries: Investigating magnesium as a potential alternative to lithium.
The chemical composition of batteries is a complex and fascinating field. Understanding these complexities is crucial to developing safer, more efficient, and sustainable energy storage solutions for the future. As technology advances, we can expect to see even more innovative battery chemistries emerging, further shaping the landscape of energy storage and our technological world.
Latest Posts
Latest Posts
-
Aluminum Foil Hydrochloric Acid Balanced Equation
Apr 03, 2025
-
How Many Sides Does An Octogon Have
Apr 03, 2025
-
The Pectoral Girdle Consists Of The
Apr 03, 2025
-
Find The Unit Vector In The Direction Of V
Apr 03, 2025
-
5 Letter Words Starting With Ho
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Are The Chemicals In A Battery . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
