The Smallest Particle Of An Element Is A N
Juapaving
Apr 02, 2025 · 5 min read
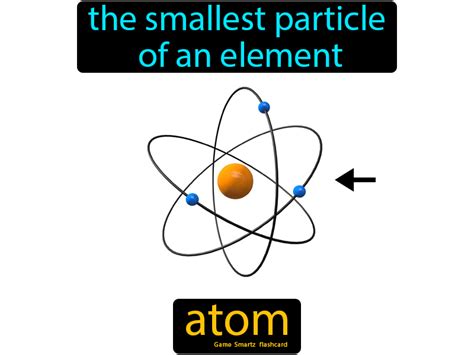
Table of Contents
The Smallest Particle of an Element is an Atom: A Deep Dive into Atomic Structure and Behavior
The question, "What is the smallest particle of an element?" has intrigued scientists for centuries. The answer, while seemingly simple – an atom – belies a universe of complexity and fascinating properties. This article delves deep into the world of atoms, exploring their structure, behavior, and significance in shaping the universe as we know it.
Understanding the Atom: A Building Block of Matter
At its core, an atom is the fundamental building block of all matter. Everything around us, from the air we breathe to the ground we walk on, is composed of countless atoms bonded together in various ways. While indivisible in the ancient Greek sense, modern science has revealed a rich internal structure to the atom.
Subatomic Particles: The Inner Workings of an Atom
Atoms are not solid, impenetrable spheres as once thought. Instead, they consist of even smaller particles:
-
Protons: Positively charged particles residing within the atom's nucleus. The number of protons in an atom's nucleus defines its atomic number and determines which element it is. For example, all hydrogen atoms have one proton, all helium atoms have two, and so on.
-
Neutrons: Neutrally charged particles also located in the atom's nucleus. Neutrons contribute to an atom's mass but not its charge. Isotopes of an element differ in the number of neutrons they possess.
-
Electrons: Negatively charged particles that orbit the nucleus in electron shells or energy levels. The number of electrons typically equals the number of protons, resulting in a neutral atom. However, atoms can gain or lose electrons, forming ions with a net positive or negative charge.
Atomic Structure and Models: From Bohr to Quantum Mechanics
Our understanding of atomic structure has evolved significantly over time. Early models, such as the Bohr model, depicted electrons orbiting the nucleus in distinct energy levels like planets orbiting a star. While a simplified representation, it helped explain the spectral lines observed in atomic emissions.
Modern understanding relies on quantum mechanics, a more complex and accurate framework. This model describes electrons not as orbiting particles but as existing in probability clouds called orbitals. These orbitals represent regions of space where there's a high probability of finding an electron. This probabilistic nature of electron location is a fundamental aspect of quantum mechanics.
The Quantum Mechanical Model: This more accurate model accounts for the wave-particle duality of electrons and provides a far more nuanced picture of atomic behavior. It's crucial for understanding chemical bonding and the properties of materials.
Isotopes and Isobars: Variations Within Elements
While the number of protons defines an element, the number of neutrons can vary. Atoms of the same element with different numbers of neutrons are called isotopes. For example, carbon-12 and carbon-14 are isotopes of carbon; both have six protons, but carbon-12 has six neutrons, while carbon-14 has eight. Some isotopes are stable, while others are radioactive, undergoing decay and emitting particles or energy.
Isobars, on the other hand, are atoms of different elements with the same mass number (total number of protons and neutrons). For instance, an isotope of calcium (20 protons, 20 neutrons) and an isotope of argon (18 protons, 22 neutrons) would be isobars.
Ions: Charged Atoms
Atoms can gain or lose electrons, forming ions. When an atom loses an electron, it becomes a positively charged cation. When an atom gains an electron, it becomes a negatively charged anion. The formation of ions is crucial for chemical bonding and plays a significant role in many chemical and biological processes.
The Significance of Atoms: From Chemistry to Cosmology
The concept of the atom is fundamental to numerous fields of science:
Chemistry: The Science of Matter and Change
Chemistry is essentially the study of matter and its transformations, and at the heart of this study lies the atom. Chemical reactions involve the rearrangement of atoms, forming and breaking bonds between them. Understanding atomic structure and behavior is vital for predicting and controlling chemical reactions, designing new materials, and developing new technologies.
Chemical Bonding: Atoms combine to form molecules through various types of bonds: ionic, covalent, and metallic. These bonds arise from the interactions of electrons between atoms and dictate the properties of molecules and materials.
Physics: Exploring the Fundamental Forces
Atomic physics explores the behavior of atoms and their interactions with electromagnetic radiation. It's crucial for understanding phenomena such as spectroscopy, laser technology, and nuclear reactions. Nuclear physics, a branch of physics, focuses specifically on the nucleus of the atom and its properties, including radioactivity and nuclear fission and fusion.
Biology: The Building Blocks of Life
Living organisms are incredibly complex systems built from atoms. Biological molecules such as proteins, DNA, and carbohydrates are all composed of atoms arranged in specific ways. Understanding the atomic structure and properties of these molecules is essential for comprehending biological processes such as metabolism, replication, and cell signaling.
Materials Science: Designing New Materials
Materials scientists use their knowledge of atomic structure and interactions to design and synthesize new materials with specific properties. This involves tailoring the arrangement and bonding of atoms to create materials with enhanced strength, conductivity, or other desired characteristics. Examples include advanced alloys, semiconductors, and superconductors.
Cosmology and Astrophysics: The Origin of Elements
The formation of elements is a fascinating area of study in cosmology and astrophysics. The abundance of elements in the universe is a direct consequence of nucleosynthesis, the process by which elements are created in stars and supernova explosions. By studying the composition of stars and galaxies, we can gain insights into the history and evolution of the universe.
Conclusion: The Atom – A Universe Within
In conclusion, while the atom might seem like a tiny and simple particle, it's a complex and fascinating entity that holds the key to understanding the universe. Its structure, behavior, and interactions with other atoms are fundamental to all aspects of science, from the smallest biological molecules to the largest stars. Continued research into the atom promises to uncover even more wonders and to further advance our knowledge of the universe and our place within it. The journey into the realm of the atom is a continuous exploration, with endless possibilities for discovery and innovation. It's a testament to the power of scientific inquiry and the beauty of nature's fundamental building block.
Latest Posts
Latest Posts
-
Four Kingdoms Of The Domain Eukarya
Apr 03, 2025
-
Do Metals Lose Or Gain Electrons
Apr 03, 2025
-
How Many Electrons Are In Phosphorus
Apr 03, 2025
-
Common Multiples Of 5 And 2
Apr 03, 2025
-
Choose The True Statement About The Krebs Cycle
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about The Smallest Particle Of An Element Is A N . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
