How Many Electrons Are In Phosphorus
Juapaving
Apr 03, 2025 · 5 min read
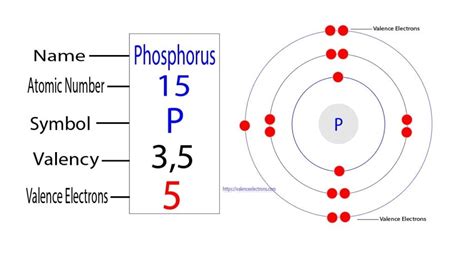
Table of Contents
How Many Electrons Are in Phosphorus? A Deep Dive into Atomic Structure
Phosphorus, a crucial element for life as we know it, plays a vital role in biological processes and numerous industrial applications. Understanding its atomic structure, particularly the number of electrons it possesses, is fundamental to comprehending its chemical behavior and properties. This in-depth article will not only answer the question "How many electrons are in phosphorus?" but also explore the broader context of atomic structure, electron configuration, and the significance of electron count in determining an element's characteristics.
Understanding Atomic Structure: Protons, Neutrons, and Electrons
Before diving into the specifics of phosphorus, let's establish a foundational understanding of atomic structure. Atoms, the basic building blocks of matter, consist of three fundamental subatomic particles:
- Protons: Positively charged particles located in the atom's nucleus. The number of protons defines the atomic number of an element and uniquely identifies it on the periodic table.
- Neutrons: Neutrally charged particles also residing in the atom's nucleus. Neutrons contribute to the atom's mass but not its charge. Isotopes of an element differ in the number of neutrons.
- Electrons: Negatively charged particles that orbit the nucleus in shells or energy levels. These electrons are responsible for the chemical properties and reactivity of an element. The number of electrons in a neutral atom is equal to the number of protons.
The Atomic Number of Phosphorus: The Key to Electron Count
The periodic table organizes elements based on their atomic number, which is the number of protons in the atom's nucleus. Phosphorus (P) is located in the third row (period) and the fifteenth column (group 15) of the periodic table. Its atomic number is 15.
Therefore, a neutral phosphorus atom contains 15 electrons.
Electron Configuration: Distributing Electrons in Shells
Electrons don't randomly orbit the nucleus. They occupy specific energy levels or shells, each capable of holding a maximum number of electrons. The distribution of electrons in these shells is described by the element's electron configuration. This configuration determines an element's reactivity and bonding behavior.
The electron shells are labeled with principal quantum numbers (n = 1, 2, 3, etc.), with lower numbers indicating shells closer to the nucleus. Each shell can accommodate a specific number of electrons:
- Shell 1 (n=1): Holds a maximum of 2 electrons.
- Shell 2 (n=2): Holds a maximum of 8 electrons.
- Shell 3 (n=3): Holds a maximum of 18 electrons (although only up to 8 electrons are typically filled in the third shell for phosphorus).
The electron configuration of phosphorus (atomic number 15) is 1s²2s²2p⁶3s²3p³. Let's break this down:
- 1s²: Two electrons in the first shell (s subshell).
- 2s²: Two electrons in the second shell (s subshell).
- 2p⁶: Six electrons in the second shell (p subshell).
- 3s²: Two electrons in the third shell (s subshell).
- 3p³: Three electrons in the third shell (p subshell).
This configuration explains why phosphorus has 15 electrons and how they are arranged within the atom. The outermost shell (valence shell) contains 5 electrons (3s²3p³), which are crucial for determining its chemical reactivity.
The Significance of Valence Electrons in Phosphorus's Reactivity
The valence electrons, those in the outermost shell, are the electrons involved in chemical bonding. Phosphorus, with 5 valence electrons, tends to gain 3 electrons to achieve a stable octet (8 electrons in the outermost shell) or share 3 electrons with other atoms to achieve a stable electron configuration. This explains why phosphorus forms compounds with other elements and exhibits a variety of oxidation states.
Phosphorus's Allotropes and Electron Configuration
Phosphorus exists in several allotropic forms, meaning it can exist in different structural modifications with varying properties. The most common allotropes are white phosphorus and red phosphorus. While the number of electrons remains consistent (15) in each allotrope, the arrangement of atoms and the bonding between them differ significantly, leading to distinct physical and chemical properties. For instance, white phosphorus is highly reactive and toxic, while red phosphorus is less reactive and more stable.
Applications of Phosphorus: A Consequence of its Electron Configuration
The unique electron configuration of phosphorus underpins its wide range of applications. Its ability to form strong covalent bonds makes it a crucial component in various materials and biological systems:
- Fertilizers: Phosphorus is an essential nutrient for plant growth, and phosphate-containing fertilizers are vital for agriculture worldwide.
- Detergents: Phosphates were once commonly used in detergents as water softeners, but their environmental impact has led to their reduction in many formulations.
- Food Additives: Phosphorus compounds are used as food additives, particularly in processed foods.
- Matches: Red phosphorus is used in the striking surface of safety matches.
- Biological Systems: Phosphorus is a crucial component of DNA, RNA, and ATP (adenosine triphosphate), the energy currency of cells. It's an essential element for all living organisms.
Isotopes of Phosphorus: Variations in Neutron Count
While the electron count remains constant for a neutral phosphorus atom, isotopes of phosphorus exist with different numbers of neutrons. The most common isotope is ³¹P, with 16 neutrons. Other, less stable isotopes of phosphorus also exist, but they have a short half-life and are radioactive. These isotopes differ in their mass but maintain the same electron configuration and chemical behavior as the most common isotope.
Conclusion: The Importance of Understanding Electron Count
Determining the number of electrons in phosphorus—15—is not just a simple numerical answer. It's a fundamental piece of information that unlocks a deeper understanding of its atomic structure, electron configuration, chemical reactivity, and its vital role in biological systems and various industrial applications. The arrangement of these 15 electrons, particularly the 5 valence electrons, dictates phosphorus's behavior and determines its position and function within the broader world of chemistry and biology. Furthermore, exploring isotopes highlights the subtle variations possible within the same element while retaining its defining characteristics. Understanding this fundamental aspect of phosphorus is essential for appreciating its widespread significance and its impact on various fields of science and technology.
Latest Posts
Latest Posts
-
Factors That Affect The Rate Of Osmosis
Apr 04, 2025
-
What Is 32 Inches In Feet
Apr 04, 2025
-
5 Letter Words Starting With Ac
Apr 04, 2025
-
How Long Is 120 Minutes In Hours
Apr 04, 2025
-
What Do I Multiply Do Get 216
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about How Many Electrons Are In Phosphorus . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
