The Scattering Of Light By A Colloid Is Called The
Juapaving
Apr 03, 2025 · 6 min read
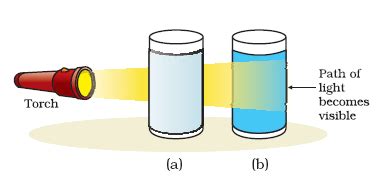
Table of Contents
The Scattering of Light by a Colloid is Called the Tyndall Effect
The shimmering beauty of a sunbeam piercing through a misty forest, the vibrant blues of a twilight sky, the opalescence of a milky gemstone – these captivating phenomena all share a common underlying principle: the scattering of light by particles. Specifically, the scattering of light by a colloid is known as the Tyndall effect. This article delves deep into the Tyndall effect, exploring its underlying physics, its diverse applications, and its significance across various scientific fields.
Understanding Colloids: A Realm Between Solutions and Suspensions
Before diving into the Tyndall effect, let's first establish a clear understanding of colloids. Colloids are mixtures where one substance is dispersed evenly throughout another, forming a stable system. The crucial distinction lies in the size of the dispersed particles: they are significantly larger than those found in solutions (e.g., salt dissolved in water) but smaller than those in suspensions (e.g., sand in water). This size range, typically between 1 and 1000 nanometers, is what imparts unique properties to colloids, including the fascinating Tyndall effect.
Examples of Colloids: A Diverse Spectrum
Colloids are ubiquitous in our everyday lives and the natural world. Some common examples include:
- Milk: Fat globules dispersed in water.
- Fog: Water droplets suspended in air.
- Blood: Blood cells dispersed in plasma.
- Paint: Pigment particles dispersed in a liquid medium.
- Mayonnaise: Oil droplets emulsified in water.
- Jell-O: A solid network holding a liquid phase.
These seemingly disparate substances all share the commonality of exhibiting colloidal behavior, making them candidates for demonstrating the Tyndall effect.
The Tyndall Effect: Unveiling the Physics of Light Scattering
The Tyndall effect, named after the 19th-century physicist John Tyndall, refers to the scattering of light as a light beam passes through a colloid. This scattering occurs because the colloidal particles are large enough to interact with the wavelengths of visible light. Unlike solutions, where the solute particles are too small to scatter visible light significantly, the larger particles in a colloid effectively scatter the light in all directions.
Rayleigh Scattering vs. Mie Scattering: Two Sides of the Same Coin
The nature of the scattering depends on the relative size of the colloidal particles compared to the wavelength of light. Two primary scattering mechanisms are at play:
-
Rayleigh scattering: This occurs when the particle size is much smaller than the wavelength of light. Rayleigh scattering is inversely proportional to the fourth power of the wavelength (λ⁻⁴). This explains why the sky appears blue: shorter wavelengths (blue) are scattered more strongly than longer wavelengths (red).
-
Mie scattering: This occurs when the particle size is comparable to or larger than the wavelength of light. Mie scattering is less dependent on wavelength, resulting in a less selective scattering of light. This explains why clouds appear white or gray: all wavelengths are scattered relatively equally.
In colloids, both Rayleigh and Mie scattering can contribute to the overall Tyndall effect, depending on the size distribution of the particles. The Tyndall effect is more pronounced with larger particles where Mie scattering is dominant.
Factors Influencing the Tyndall Effect: A Multifaceted Phenomenon
Several factors influence the intensity and visibility of the Tyndall effect:
- Particle size: Larger particles scatter more light than smaller particles.
- Particle concentration: Higher concentrations of colloidal particles lead to greater scattering.
- Wavelength of light: Shorter wavelengths are scattered more effectively than longer wavelengths, especially in Rayleigh scattering.
- Refractive index difference: A greater difference in the refractive indices of the dispersed phase and the dispersion medium enhances scattering.
Understanding these factors is crucial for controlling and utilizing the Tyndall effect in various applications.
Applications of the Tyndall Effect: A Spectrum of Uses
The Tyndall effect finds applications in numerous scientific and industrial settings, extending its reach far beyond simple observation.
1. Determining Colloidal Nature: A Diagnostic Tool
The Tyndall effect serves as a simple yet powerful method for distinguishing between true solutions and colloids. Shining a beam of light through a sample will reveal whether the Tyndall effect is present: a visible scattering path indicates a colloid, while the absence of scattering suggests a true solution. This simple test is widely employed in analytical chemistry and material science.
2. Atmospheric Optics: Unveiling the Beauty of the Sky
The Tyndall effect plays a crucial role in shaping the colors and appearances we observe in the atmosphere. The scattering of sunlight by atmospheric colloids, such as water droplets in clouds and dust particles, leads to phenomena like:
- Blue skies: Rayleigh scattering of sunlight by air molecules.
- Red sunsets and sunrises: Longer wavelengths (red) penetrate the atmosphere more readily when the sun is low on the horizon.
- White clouds: Mie scattering by larger water droplets.
- Crepuscular rays: Sunlight scattering through gaps in clouds.
These atmospheric optics illustrate the pervasive influence of light scattering on our perception of the natural world.
3. Industrial Applications: Enhancing Products and Processes
The Tyndall effect finds diverse applications in industrial processes:
- Food science: Monitoring the size and distribution of fat globules in dairy products.
- Pharmaceutical industry: Characterizing the size and stability of drug nanoparticles.
- Paint industry: Controlling the opacity and color of paints.
- Material science: Developing new materials with tailored optical properties.
These applications highlight the importance of understanding and controlling light scattering in industrial manufacturing and quality control.
4. Medical Diagnostics: Illuminating Biological Systems
The Tyndall effect plays a significant role in various medical diagnostic techniques:
- Nephelometry: Measuring the turbidity of fluids based on light scattering, used in assessing protein levels and detecting infections.
- Laser light scattering: Analyzing the size and distribution of particles in biological samples.
These applications demonstrate the potential of light scattering techniques to improve medical diagnostics and patient care.
Beyond the Basics: Exploring Advanced Concepts
The Tyndall effect is more than just a simple observation; it opens doors to a deeper understanding of light-matter interactions at the nanoscale. Advanced concepts related to the Tyndall effect include:
- Dynamic light scattering: Measuring the Brownian motion of particles to determine their size and hydrodynamic radius.
- Photon correlation spectroscopy: Analyzing the fluctuations in scattered light to obtain information about particle size and diffusion coefficients.
- Light scattering spectroscopy: Employing different light sources and detection techniques to obtain detailed information about particle shape, size, and composition.
These advanced techniques provide powerful tools for analyzing colloidal systems and gaining deeper insights into their structure and dynamics.
Conclusion: The Enduring Significance of the Tyndall Effect
The scattering of light by a colloid, known as the Tyndall effect, is a fundamental phenomenon with far-reaching consequences. From the vibrant colors of sunsets to the precise characterization of nanoparticles, the Tyndall effect plays a crucial role in various scientific disciplines and industrial applications. Understanding the underlying physics, factors influencing the effect, and its diverse applications are essential for harnessing its power in numerous fields, paving the way for technological advancements and a deeper understanding of the natural world around us. The simple observation of a light beam scattering through a colloid reveals a universe of complexity and potential. This seemingly simple phenomenon continues to fascinate and inspire scientists, revealing ever more insights into the fundamental interactions between light and matter. The study of the Tyndall effect remains a vital area of research, promising further breakthroughs and applications in the future. The seemingly simple beauty of a sunbeam cutting through fog or mist is, in reality, a testament to the profound physical principles governing our world.
Latest Posts
Latest Posts
-
415 Rounded To The Nearest Hundred
Apr 04, 2025
-
Addition Of Water To An Alkyne
Apr 04, 2025
-
What Is 5 5 In Centimeters
Apr 04, 2025
-
Changes Of State Occur When There Is A Change In
Apr 04, 2025
-
8 Centimeters Is How Many Millimeters
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about The Scattering Of Light By A Colloid Is Called The . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
