Changes Of State Occur When There Is A Change In
Juapaving
Apr 04, 2025 · 6 min read
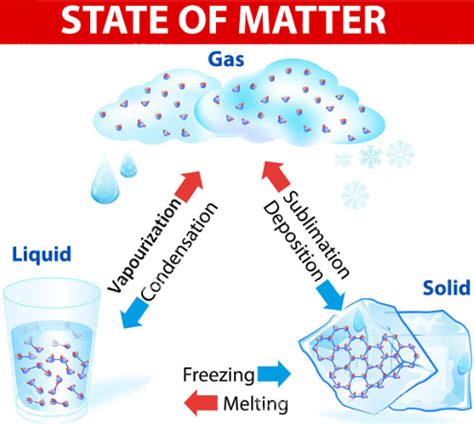
Table of Contents
Changes of State Occur When There Is a Change In... Energy!
Changes of state, also known as phase transitions, are fascinating phenomena that occur when the physical state of a substance transforms from one form to another. Water, for instance, can exist as ice (solid), liquid water, or water vapor (gas). These transformations aren't random; they're governed by fundamental principles of physics, specifically the transfer of energy. This article delves deep into the mechanics of changes of state, exploring the role of energy, temperature, and pressure in driving these transitions. We'll examine the different types of phase transitions and look at real-world examples to solidify our understanding.
The Role of Energy in Phase Transitions
At the heart of every change of state lies a shift in the kinetic and potential energy of the molecules comprising the substance. Let's break this down:
Kinetic Energy: The Energy of Motion
Kinetic energy refers to the energy associated with the motion of molecules. In a solid, molecules are tightly packed and vibrate in place with relatively low kinetic energy. As you add energy (typically in the form of heat), these vibrations become more vigorous. This increased kinetic energy is what eventually leads to a change in state.
Potential Energy: The Energy of Attraction
Potential energy, on the other hand, represents the energy stored within the bonds and interactions between molecules. In solids, strong intermolecular forces hold molecules in fixed positions. Adding energy weakens these forces, allowing molecules to overcome the attractive forces holding them together. This is crucial for transitions from solid to liquid and liquid to gas.
The Interplay of Kinetic and Potential Energy
The transition from one state to another isn't simply about increasing kinetic energy; it's a complex interplay between kinetic and potential energy. For example, when ice melts, the added heat increases the kinetic energy of water molecules, causing them to vibrate more forcefully. This increased vibration overcomes the strong intermolecular forces (hydrogen bonds) holding the water molecules in a rigid lattice structure, leading to a decrease in potential energy and the transition to the liquid state.
Types of Phase Transitions
There are several key types of phase transitions, each characterized by a specific change in the arrangement and energy of molecules:
1. Melting (Solid to Liquid)
Melting occurs when a solid transitions to a liquid. This requires an input of energy, overcoming the intermolecular forces that maintain the rigid structure of the solid. The melting point is the temperature at which this transition occurs at a given pressure.
2. Freezing (Liquid to Solid)
Freezing is the reverse of melting; it's the transition from a liquid to a solid. This process releases energy as the molecules lose kinetic energy and become more ordered, forming a solid structure. The freezing point is the temperature at which this transition occurs at a given pressure; under normal conditions, the freezing and melting points are the same.
3. Vaporization (Liquid to Gas)
Vaporization involves the transition from a liquid to a gas. This requires a significant input of energy to overcome the intermolecular forces holding the liquid molecules together, allowing them to escape into the gaseous phase. Vaporization can occur through boiling (at a specific boiling point) or evaporation (at temperatures below the boiling point).
4. Condensation (Gas to Liquid)
Condensation is the reverse of vaporization; it's the transition from a gas to a liquid. This process releases energy as gas molecules lose kinetic energy and come closer together, forming a liquid. Condensation often occurs when a gas cools below its dew point.
5. Sublimation (Solid to Gas)
Sublimation is the direct transition from a solid to a gas, bypassing the liquid phase. This process requires an input of energy to overcome the intermolecular forces in the solid and allow the molecules to escape directly into the gaseous phase. Dry ice (solid carbon dioxide) is a classic example of a substance that sublimates.
6. Deposition (Gas to Solid)
Deposition is the reverse of sublimation; it's the direct transition from a gas to a solid, bypassing the liquid phase. This process releases energy as gas molecules lose kinetic energy and directly form a solid structure. Frost formation is a common example of deposition.
Factors Influencing Phase Transitions: Temperature and Pressure
Temperature and pressure play crucial roles in determining the state of a substance and influencing phase transitions.
Temperature's Influence
Temperature is directly related to the kinetic energy of molecules. Higher temperatures mean higher kinetic energy, making it easier for molecules to overcome intermolecular forces and transition to a higher-energy state (e.g., solid to liquid, liquid to gas).
Pressure's Influence
Pressure affects the intermolecular forces and the spacing between molecules. Higher pressure forces molecules closer together, increasing intermolecular forces and making it more difficult for a substance to transition to a less dense state (e.g., solid to liquid, liquid to gas). Conversely, lower pressure reduces intermolecular forces, facilitating transitions to less dense states.
Phase Diagrams: Visualizing Phase Transitions
Phase diagrams are graphical representations that illustrate the relationship between temperature, pressure, and the state of a substance. These diagrams show the conditions under which different phases exist and the transitions between them. Analyzing a phase diagram allows one to predict the state of a substance at specific temperature and pressure conditions.
Real-World Examples of Changes of State
Changes of state are ubiquitous in our everyday lives:
- Boiling water: The transition from liquid water to water vapor is a critical part of cooking and many industrial processes.
- Formation of ice: The freezing of water into ice is essential for preserving food and creating winter landscapes.
- Evaporation of sweat: The vaporization of sweat from our skin helps regulate our body temperature.
- Condensation on a cold glass: The condensation of water vapor from the air onto a cold glass demonstrates the transition from gas to liquid.
- Dry ice sublimating: The direct transition of solid carbon dioxide to gaseous carbon dioxide is used in special effects and cooling applications.
- Frost formation: The deposition of water vapor directly onto surfaces as ice is a common winter phenomenon.
Conclusion
Changes of state are fundamental physical processes driven by changes in the energy of molecules. Understanding the interplay between kinetic and potential energy, along with the influence of temperature and pressure, is crucial for comprehending these transitions. From the melting of ice to the boiling of water, these phenomena are integral to various natural and technological processes, highlighting the importance of studying phase transitions in different fields of science and engineering. By continuing to explore and understand these processes, we can further develop innovative technologies and enhance our comprehension of the physical world around us. Further research into the intricacies of phase transitions continues to unravel new discoveries and refine our models of molecular behavior, constantly expanding our knowledge of this fascinating area of physics. The seemingly simple shift from one state of matter to another unveils a complex world of molecular dynamics, showcasing the elegance and precision of the laws of nature.
Latest Posts
Latest Posts
-
What Is The Square Root Of 112
Apr 04, 2025
-
Balanced Equation For Nitric Acid And Sodium Hydroxide
Apr 04, 2025
-
Energy Of Electron In Nth Orbit Formula
Apr 04, 2025
-
Find Moment Of Inertia Of Solid Sphere
Apr 04, 2025
-
6 Letter Word Starting With Sta
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Changes Of State Occur When There Is A Change In . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
