Addition Of Water To An Alkyne
Juapaving
Apr 04, 2025 · 6 min read
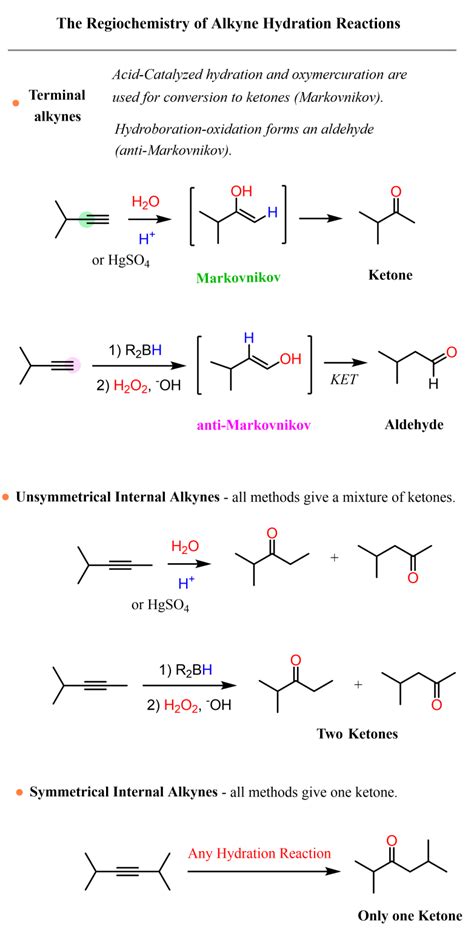
Table of Contents
The Hydration of Alkynes: A Deep Dive into Reaction Mechanisms, Regioselectivity, and Applications
The addition of water to alkynes, also known as alkyne hydration, is a fascinating and significant reaction in organic chemistry. This process transforms a relatively unreactive alkyne into a valuable carbonyl compound – a ketone or aldehyde – opening up a world of synthetic possibilities. Understanding the intricacies of this reaction, including its mechanism, regioselectivity, and practical applications, is crucial for any aspiring organic chemist.
Understanding the Reaction: A Mechanistic Overview
The direct hydration of alkynes doesn't occur readily under normal conditions. Alkynes, with their relatively strong C≡C triple bond, are not easily attacked by water molecules. To facilitate the reaction, the presence of a catalyst is essential. The most common catalyst employed is mercury(II) sulfate (HgSO₄) in the presence of a strong acid, typically sulfuric acid (H₂SO₄). This combination promotes the addition of water across the triple bond.
The reaction proceeds through a series of steps, broadly categorized as follows:
Step 1: Mercury(II) Ion Catalyzed Protonation
The alkyne first interacts with the mercury(II) ion, which acts as a Lewis acid. This coordination forms a π-complex, activating the triple bond and making it more susceptible to electrophilic attack. Subsequently, a proton (H⁺) from the sulfuric acid adds to one of the carbon atoms of the alkyne, creating a vinyl cation intermediate. This step is crucial for setting up the regiochemistry of the final product. Markovnikov's rule generally governs this protonation step.
Step 2: Nucleophilic Attack by Water
The vinyl cation intermediate, being highly electrophilic, is readily attacked by a water molecule, a nucleophile. This attack occurs at the carbon atom bearing the partial positive charge, resulting in a neutral intermediate.
Step 3: Proton Transfer and Tautomerization
A proton transfer ensues, typically involving the loss of a proton from the oxygen atom and its acquisition by another oxygen atom from the catalyst. This facilitates the formation of an enol, an alcohol with the hydroxyl group attached to a carbon-carbon double bond.
Step 4: Keto-Enol Tautomerization
The enol undergoes a rapid tautomerization to the more stable keto form. This tautomerization, catalyzed by acid, involves the migration of a proton from the hydroxyl group to the adjacent carbon atom, resulting in the formation of a carbonyl group (C=O) and ultimately, a ketone or an aldehyde. The exact structure of the ketone or aldehyde depends on the structure of the starting alkyne.
Regioselectivity: Markovnikov's Rule and Beyond
The regioselectivity of alkyne hydration is largely governed by Markovnikov's rule. This rule states that in the addition of a protic acid to an unsymmetrical alkyne, the proton adds to the carbon atom that already possesses more hydrogen atoms. This results in the formation of a more substituted carbocation intermediate, which is more stable. Consequently, the carbonyl group will be positioned on the more substituted carbon atom.
However, it's important to note that certain conditions can influence the regioselectivity. The use of alternative catalysts or reaction conditions can lead to deviations from Markovnikov's rule, particularly with terminal alkynes. In some cases, anti-Markovnikov addition can be achieved through the use of specific reagents and catalysts.
Applications of Alkyne Hydration
Alkyne hydration is a powerful synthetic tool with a wide array of applications in organic synthesis and industrial processes.
Synthesis of Ketones and Aldehydes
The most prominent application is the straightforward synthesis of ketones and aldehydes. This reaction provides a convenient and efficient route to these important functional groups, which are building blocks for many organic molecules. The ability to control the regioselectivity of the reaction allows for the selective synthesis of specific isomers, offering fine-tuned control over the product's structure.
Synthesis of Pharmaceuticals and Fine Chemicals
The ability to access ketones and aldehydes through alkyne hydration makes this reaction vital in pharmaceutical and fine chemical synthesis. Many biologically active molecules contain these functional groups, and alkyne hydration offers a valuable strategy to incorporate them into complex molecular scaffolds. This is especially valuable when the desired ketone or aldehyde cannot be easily accessed via alternative synthetic methods.
Industrial Applications
Alkyne hydration finds its place in several industrial processes as well. It is used in the synthesis of certain polymers and other materials. The reaction's efficiency and selectivity contribute to its suitability for large-scale production.
Factors Influencing the Reaction
Several factors can significantly influence the success and outcome of alkyne hydration. Careful control of these variables is crucial to optimizing the yield and selectivity of the reaction.
Catalyst Concentration
The concentration of the mercury(II) sulfate catalyst plays a pivotal role. An insufficient amount of catalyst may lead to a slow reaction rate or incomplete conversion of the alkyne. Conversely, an excessive amount of catalyst can lead to unwanted side reactions. Optimization of the catalyst concentration is essential for achieving maximum efficiency.
Acid Concentration
The concentration of the strong acid, typically sulfuric acid, also impacts the reaction. The acid not only provides the protons necessary for the reaction but also helps to stabilize the intermediates and promote tautomerization. The acid concentration needs to be carefully controlled to avoid undesirable side reactions or decomposition of the reactants.
Temperature
Temperature significantly influences the reaction rate. Higher temperatures generally lead to faster reaction rates, but excessive heat can cause undesirable side reactions or decomposition. Optimization of the reaction temperature is essential to achieve the optimal balance between speed and selectivity.
Solvent Selection
The choice of solvent can influence the reaction's outcome. A suitable solvent should effectively dissolve both the alkyne and the catalyst while not participating in any unwanted side reactions. The solvent's polarity and ability to stabilize the intermediates play a key role.
Safety Precautions
Mercury(II) sulfate is a toxic compound, and its handling requires appropriate safety precautions. The reaction should be conducted in a well-ventilated area, and proper personal protective equipment, including gloves and eye protection, should be used. Disposal of the waste products should follow established safety protocols. The use of alternative catalysts that are less toxic is an active area of research, aiming to enhance the sustainability and safety of this valuable reaction.
Future Directions and Research
Despite its extensive use, research continues to explore improvements and alternatives to the traditional mercury-catalyzed hydration of alkynes. The main focus of current research lies in the development of more environmentally friendly and less toxic catalytic systems. This includes investigating the use of noble metals, metal complexes, and organocatalysts as replacements for mercury(II) sulfate. Moreover, research is focused on enhancing the regio- and stereoselectivity of the reaction, allowing for greater control over the final product. The development of new catalytic systems and improved reaction conditions will undoubtedly continue to broaden the scope and applications of alkyne hydration in organic synthesis.
Conclusion
The hydration of alkynes remains a cornerstone reaction in organic chemistry. Its ability to transform alkynes into valuable carbonyl compounds, coupled with its relatively straightforward mechanism, makes it a highly versatile tool for synthetic chemists. Understanding the factors that influence the reaction, including the catalyst, reaction conditions, and regioselectivity, is crucial for its successful application. As research continues to explore greener and more efficient catalytic systems, the importance of alkyne hydration in organic synthesis and industrial applications is only set to increase. This versatile reaction will undoubtedly remain a powerful synthetic strategy for years to come, paving the way for innovative advancements in various fields.
Latest Posts
Latest Posts
-
Glycogen Cellulose And Starch Are All
Apr 04, 2025
-
The Stored Form Of Carbohydrate In Humans Is Called Term1
Apr 04, 2025
-
Boiling Point Of Water In Celsius Scale
Apr 04, 2025
-
Dna Synthesis Occurs During Which Phase Of The Cell Cycle
Apr 04, 2025
-
Chemicals Used In The Therapy Of Infectious Diseases Are Called
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Addition Of Water To An Alkyne . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
