Number Of Valence Electrons In S
Juapaving
Apr 02, 2025 · 5 min read
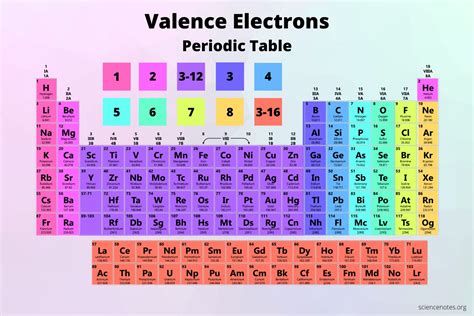
Table of Contents
Unveiling the Secrets of Valence Electrons in the 's' Block: A Comprehensive Guide
Understanding valence electrons is fundamental to grasping the behavior of elements and their interactions in chemical reactions. This in-depth guide focuses specifically on the number of valence electrons found in the 's' block elements of the periodic table, exploring their significance and how this number dictates their reactivity and properties.
What are Valence Electrons?
Before diving into the specifics of 's' block elements, let's solidify our understanding of valence electrons. Valence electrons are the electrons located in the outermost shell (or energy level) of an atom. These electrons are the primary players in chemical bonding, determining an element's reactivity and the types of bonds it can form (ionic, covalent, metallic). They are the "hands" that atoms use to interact with each other. The number of valence electrons directly impacts an element's chemical properties and its position within the periodic table.
The 's' Block: A Home for Valence Electron Simplicity
The periodic table is organized to reflect the electronic configuration of elements. The 's' block, encompassing Groups 1 (alkali metals) and 2 (alkaline earth metals), is characterized by its elements having their valence electrons occupying the 's' subshell. This makes understanding their valence electron count remarkably straightforward.
Group 1: Alkali Metals – One Valence Electron
Alkali metals, including lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr), are defined by having one valence electron in their outermost 's' subshell. This single electron is easily lost, making these metals highly reactive. They readily form +1 cations (positively charged ions) to achieve a stable electron configuration, mimicking the noble gas configuration of the previous period. This drive for stability explains their high reactivity and tendency to participate in ionic bonding.
Example: Sodium (Na) has an electron configuration of 1s²2s²2p⁶3s¹. The 3s¹ electron is its single valence electron. It readily loses this electron to form Na⁺, achieving the stable electron configuration of neon (Ne).
Group 2: Alkaline Earth Metals – Two Valence Electrons
Alkaline earth metals, including beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra), possess two valence electrons in their outermost 's' subshell. These elements are also relatively reactive, although less so than alkali metals. They tend to lose both valence electrons to form +2 cations, striving for the noble gas configuration. However, the increased nuclear charge holding onto the two electrons results in slightly higher ionization energies compared to alkali metals.
Example: Magnesium (Mg) has an electron configuration of 1s²2s²2p⁶3s². Its two 3s² electrons are its valence electrons. It loses both electrons to form Mg²⁺, achieving the stable electron configuration of neon (Ne).
The Significance of Valence Electron Count in 's' Block Elements
The consistent and predictable number of valence electrons in the 's' block elements has profound implications for their chemical and physical properties:
-
Reactivity: The ease with which 's' block elements lose their valence electrons directly correlates with their reactivity. Alkali metals, with only one valence electron, are the most reactive in their respective periods, followed by alkaline earth metals with two valence electrons.
-
Ionization Energy: The energy required to remove an electron from an atom is called ionization energy. 's' block elements have relatively low ionization energies, reflecting the relatively weak attraction between the nucleus and their outermost valence electrons. This low ionization energy further contributes to their reactivity.
-
Ionic Bonding: The tendency of 's' block elements to readily lose their valence electrons leads to the formation of ionic bonds. They readily form ionic compounds with nonmetals, which readily gain electrons to achieve a stable electron configuration.
-
Metallic Bonding: The 's' block elements are all metals, characterized by metallic bonding. This type of bonding involves the delocalization of valence electrons across a lattice of metal atoms, resulting in the characteristic properties of metals such as conductivity and malleability.
-
Oxidation States: The number of valence electrons dictates the typical oxidation states exhibited by these elements. Alkali metals almost always exhibit a +1 oxidation state, while alkaline earth metals predominantly exhibit a +2 oxidation state.
Exploring Exceptions and Nuances
While the number of valence electrons in the 's' block is generally straightforward, some nuances warrant consideration:
-
Hydrogen and Helium: While hydrogen and helium are placed in the 's' block, their valence electron behavior slightly differs. Hydrogen possesses one valence electron and can either lose it to form a +1 cation or share it to form a covalent bond. Helium, with a full outer shell (1s²), is a noble gas and is chemically inert, showcasing the stability of a completely filled 's' subshell.
-
Relativistic Effects: For heavier 's' block elements like francium and radium, relativistic effects become increasingly significant. These effects alter the electron distribution and can subtly affect chemical properties, leading to slight deviations from expected behavior based solely on the number of valence electrons.
Practical Applications and Real-World Examples
The properties dictated by the valence electrons of 's' block elements are exploited in numerous practical applications:
-
Sodium (Na): Used in sodium vapor lamps for street lighting, due to its low ionization energy and ability to emit light at specific wavelengths.
-
Magnesium (Mg): A crucial component in lightweight alloys used in the aerospace and automotive industries. Its reactivity and ability to form strong bonds contribute to the strength and durability of these alloys.
-
Calcium (Ca): Essential for many biological processes, playing a vital role in bone formation, muscle contraction, and nerve impulse transmission.
-
Lithium (Li): Used extensively in rechargeable batteries for portable electronics due to its high electrochemical potential.
Conclusion: A Foundation for Chemical Understanding
The number of valence electrons in the 's' block elements, whether one (alkali metals) or two (alkaline earth metals), is a cornerstone in predicting and understanding their chemical behavior. This straightforward relationship between valence electrons and properties makes the 's' block an excellent starting point for learning about chemical bonding, reactivity, and the broader principles governing the behavior of matter. This understanding lays the groundwork for exploring the more complex electronic configurations and chemical behaviors of elements in other blocks of the periodic table. Mastering this fundamental concept empowers further exploration of chemical principles and their diverse applications in various fields. The predictable nature of 's' block elements provides a solid foundation for developing a strong understanding of the periodic table and the power of valence electrons in determining chemical properties.
Latest Posts
Latest Posts
-
Identify The Structure Indicated By The Lines
Apr 03, 2025
-
How To Find The Perimeter Of A Hexagon
Apr 03, 2025
-
This Is The Functional Unit Of The Kidney
Apr 03, 2025
-
Four Kingdoms Of The Domain Eukarya
Apr 03, 2025
-
Do Metals Lose Or Gain Electrons
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Number Of Valence Electrons In S . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
