Number Of Valence Electrons In Na
Juapaving
Apr 05, 2025 · 5 min read
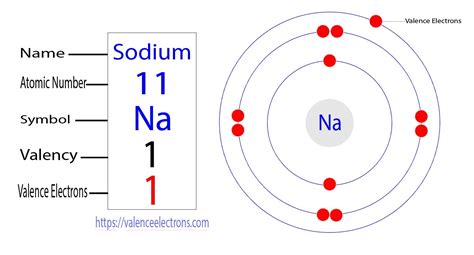
Table of Contents
Unveiling the Secrets of Sodium's Valence Electrons: A Deep Dive into Atomic Structure and Reactivity
Sodium (Na), a ubiquitous element found in table salt and essential for human life, holds a fascinating place in the periodic table. Understanding its atomic structure, particularly the number of valence electrons, is key to grasping its chemical behavior and reactivity. This comprehensive guide will delve into the intricacies of sodium's electron configuration, explaining why it possesses only one valence electron and how this single electron dictates its remarkable properties. We'll explore the implications of this valence electron count in various contexts, including its bonding behavior, ionic interactions, and significance in biological systems.
Understanding Valence Electrons: The Key to Chemical Reactivity
Before focusing specifically on sodium, let's establish a foundational understanding of valence electrons. These are the electrons located in the outermost shell of an atom, also known as the valence shell. These electrons are the primary players in chemical bonding, determining an element's reactivity and the types of bonds it can form. The number of valence electrons directly influences an atom's ability to gain, lose, or share electrons to achieve a stable electron configuration, typically resembling that of a noble gas. This stable configuration is often characterized by a full outermost shell, providing exceptional stability.
The Significance of Octet Rule
The octet rule, a cornerstone of chemical bonding theory, states that atoms tend to gain, lose, or share electrons to achieve a stable configuration of eight electrons in their valence shell. This configuration mirrors the stable electron arrangement of noble gases, which are notoriously unreactive. While there are exceptions, the octet rule provides a valuable framework for predicting the reactivity and bonding behavior of many elements.
Delving into Sodium's Atomic Structure: Uncovering its Single Valence Electron
Sodium, with its atomic number of 11, possesses 11 protons and 11 electrons in its neutral state. To understand the distribution of these electrons, we need to examine its electron configuration. This configuration dictates how electrons are arranged within various energy levels or shells surrounding the nucleus.
Electron Configuration of Sodium (Na)
The electron configuration of sodium is written as 1s²2s²2p⁶3s¹. This seemingly simple notation reveals a wealth of information:
- 1s²: Two electrons occupy the first energy level (shell), specifically the 1s orbital.
- 2s²2p⁶: Eight electrons fill the second energy level, with two in the 2s orbital and six in the 2p orbitals.
- 3s¹: A single electron resides in the outermost energy level (third shell), specifically in the 3s orbital. This lone electron is the valence electron of sodium.
This single valence electron in the 3s orbital is crucial in determining sodium's chemical behavior. It is relatively loosely held by the nucleus and easily participates in chemical reactions.
Sodium's Reactivity: A Consequence of its Single Valence Electron
The presence of only one valence electron makes sodium highly reactive. To achieve a stable octet, sodium readily loses this single electron, forming a positively charged ion known as a sodium cation (Na⁺). This process is known as ionization. The resulting Na⁺ ion has a complete octet, mirroring the electron configuration of neon (Ne), a noble gas.
Ionic Bonding: Sodium's Preferred Bonding Method
Sodium's tendency to lose an electron often leads to the formation of ionic bonds. Ionic bonds arise from the electrostatic attraction between oppositely charged ions. For instance, when sodium reacts with chlorine (Cl), which has seven valence electrons, sodium readily donates its valence electron to chlorine. Chlorine, accepting this electron, becomes a negatively charged chloride ion (Cl⁻), achieving a stable octet. The electrostatic attraction between the positively charged Na⁺ and negatively charged Cl⁻ ions forms the ionic compound sodium chloride (NaCl), commonly known as table salt.
Other Reactions and Compounds: Exploring Sodium's Versatility
Sodium's reactivity extends beyond its interaction with chlorine. It readily reacts with other nonmetals, such as oxygen (O) and sulfur (S), forming ionic compounds. For example, sodium reacts vigorously with oxygen to produce sodium oxide (Na₂O). The reaction with water is particularly dramatic, producing sodium hydroxide (NaOH) and hydrogen gas (H₂), often accompanied by a significant release of heat.
The Biological Significance of Sodium and its Valence Electron
Sodium's unique chemical properties, stemming from its single valence electron, have profound implications in biological systems. Sodium ions (Na⁺) play vital roles in various physiological processes:
- Nerve Impulse Transmission: Sodium ions are crucial for the propagation of nerve impulses. The movement of Na⁺ ions across neuronal membranes generates electrical signals essential for communication within the nervous system.
- Muscle Contraction: Sodium ions also participate in muscle contraction. Changes in the concentration of Na⁺ ions across muscle cell membranes trigger the contraction mechanism.
- Fluid Balance: Sodium ions contribute to maintaining the proper balance of fluids within the body. This balance is critical for numerous physiological functions.
- Nutrient Absorption: Sodium ions aid in the absorption of nutrients from the digestive tract.
These biological functions highlight the critical role of sodium, whose reactivity and ion formation are all directly linked to its single valence electron.
Beyond the Basics: Advanced Concepts and Applications
Understanding sodium's valence electron is fundamental, but it's only the starting point for a deeper exploration. Further investigations could explore:
- Sodium's spectral lines: The single valence electron influences the absorption and emission spectra of sodium, contributing to its unique spectral fingerprint. This is used extensively in analytical chemistry.
- Sodium in alloys: Sodium's properties are exploited in various metal alloys, influencing their melting points and other characteristics.
- Sodium's role in industrial processes: Sodium's high reactivity makes it an important reactant in several industrial processes, including the production of other chemicals.
- Isotopes of Sodium: Exploring different isotopes of sodium and their nuclear properties expands understanding of its broader atomic characteristics.
Conclusion: A Single Electron with a Vast Impact
The seemingly simple fact that sodium has one valence electron underpins its remarkable chemical reactivity and its crucial role in biological systems. This single electron governs its bonding behavior, driving the formation of ionic compounds and enabling its participation in critical physiological processes. From the formation of table salt to the transmission of nerve impulses, sodium's single valence electron exerts a disproportionately large influence on the world around us. Further study and exploration of this fundamental aspect of sodium's atomic structure unlock a deeper understanding of its diverse applications and biological significance. This single electron acts as a key that unlocks a universe of chemical and biological phenomena.
Latest Posts
Latest Posts
-
Which Of The Following Is Rational
Apr 06, 2025
-
Difference Between An Alternator And A Generator
Apr 06, 2025
-
How Long Is 16 Cm In Inches
Apr 06, 2025
-
Boiling Point Of Water In Kelvin Scale
Apr 06, 2025
-
How Many Feet Is 180 Inches
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Number Of Valence Electrons In Na . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
