How Are Compounds And Mixtures Alike
Juapaving
Apr 02, 2025 · 6 min read
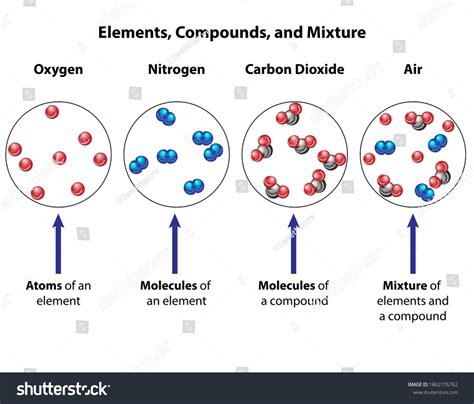
Table of Contents
- How Are Compounds And Mixtures Alike
- Table of Contents
- How Are Compounds and Mixtures Alike? Exploring Similarities in Chemical Combinations
- Similarities in Physical State and Appearance
- The Possibility of Separation: A Shared Characteristic
- The Role of Proportion: A Subtle Similarity
- Everyday Examples: Sharing the Same Space
- The Importance of Understanding the Differences Despite the Similarities
- Conclusion: A Holistic Perspective on Chemical Combinations
- Latest Posts
- Latest Posts
- Related Post
How Are Compounds and Mixtures Alike? Exploring Similarities in Chemical Combinations
Compounds and mixtures are both fundamental concepts in chemistry, representing ways in which different substances can combine. While they differ significantly in their chemical composition and properties, exploring their similarities can provide valuable insights into the nature of matter. This article delves into the surprising commonalities between compounds and mixtures, highlighting their shared characteristics and providing a deeper understanding of these crucial chemical concepts. We'll explore their physical states, the possibility of separation, and how both can be found throughout our everyday lives.
Similarities in Physical State and Appearance
One striking similarity between compounds and mixtures is that they can both exist in all three fundamental states of matter: solid, liquid, and gas. A compound like water (H₂O) can exist as ice (solid), liquid water, or steam (gas), while a mixture like saltwater can similarly exist in all three states. This demonstrates that the manner of combination – whether a compound or a mixture – doesn't inherently dictate the physical state.
Furthermore, both compounds and mixtures can appear homogeneous or heterogeneous. A homogeneous mixture, like saltwater, appears uniform throughout, meaning its components are indistinguishable at a macroscopic level. Similarly, a compound like sugar (sucrose, C₁₂H₂₂O₁₁) appears homogeneous. In contrast, a heterogeneous mixture, such as sand and water, exhibits visibly distinct components. However, even some compounds can appear heterogeneous depending on their crystalline structure. For example, a large crystal of salt might look heterogeneous at a macroscopic level, even though it's chemically a uniform compound. This highlights that appearance alone cannot definitively distinguish between a compound and a mixture; chemical analysis is often necessary.
The Possibility of Separation: A Shared Characteristic
Both compounds and mixtures can, in principle, be separated into their constituent parts. However, the methods used and the resulting products are dramatically different. Mixtures can be separated using physical methods that exploit the differences in physical properties of their components, such as filtration, distillation, evaporation, chromatography, and magnetism. For instance, sand can be separated from water using filtration, while components of crude oil are separated through distillation based on their boiling points.
Compounds, on the other hand, require chemical methods for separation. This is because the components of a compound are chemically bonded, requiring a chemical reaction to break these bonds and separate the elements. For example, the electrolysis of water separates water into its constituent elements, hydrogen and oxygen, through the application of an electric current which initiates a chemical reaction. Therefore, while both compounds and mixtures can be separated, the methods employed differ significantly, reflecting the fundamental difference in their chemical bonding.
The Role of Proportion: A Subtle Similarity
Although the proportions of elements in a compound are fixed and defined by its chemical formula, the proportions of components in a mixture are variable. However, this difference doesn't negate a subtle similarity: both compounds and mixtures can exist in various proportions of their constituents. The crucial difference lies in the fact that the composition of a compound is invariant, while the composition of a mixture is variable. For example, water always contains hydrogen and oxygen in a 2:1 ratio by volume. In contrast, a mixture of sand and water can have varying proportions of sand and water.
While a specific compound always has a definite and constant proportion of its constituent elements, the proportions within a mixture can vary significantly. This might be viewed as a similarity in a broad sense: the ability to have different quantities of the constituent parts in existence. Think of the different kinds of steel alloys; the mixture of iron, carbon, and other trace elements varies significantly to get different properties. This variability itself is a defining feature of mixtures. However, in the context of the constituents' relative proportions, there is a distinction. In a compound, changing the relative proportions changes the substance itself. In a mixture, changing the relative proportions creates a different mixture of the same components.
Everyday Examples: Sharing the Same Space
Both compounds and mixtures are ubiquitous in our everyday lives, often existing side-by-side without our explicit awareness. Consider the air we breathe – it's a mixture of different gases (primarily nitrogen, oxygen, and argon), each retaining its individual chemical properties. However, water, a compound, is vital to this mixture, present in the form of water vapor. This simple example underscores that compounds and mixtures frequently coexist within the same system.
Similarly, consider the food we eat. Many foods are complex mixtures of various compounds. A salad, for instance, is a mixture of different vegetables and dressings. Each vegetable is made of various compounds, such as carbohydrates, proteins, vitamins, and minerals, all present in varying proportions. The dressing itself is a mixture of oil, vinegar, and other ingredients, each composed of diverse compounds. This intricately interwoven combination of compounds and mixtures highlights their pervasive nature in our world.
The Importance of Understanding the Differences Despite the Similarities
While this article has highlighted several interesting similarities between compounds and mixtures, it's crucial to reiterate their fundamental differences. These differences stem from the nature of the bonding between their constituents. Compounds are formed through chemical reactions that create strong chemical bonds between atoms of different elements, resulting in a completely new substance with unique properties. Mixtures, conversely, are merely physical combinations of different substances, retaining their individual chemical properties.
The distinctions are critical because they underpin our understanding of chemical reactions, material properties, and countless other phenomena in the natural world. The properties of a compound are vastly different from the properties of its constituent elements. For example, sodium is a highly reactive metal, and chlorine is a poisonous gas. However, their combination forms sodium chloride (table salt), a compound that is essential for life and quite different in properties from its components. No such drastic change in properties occurs when substances are mixed.
Understanding the subtle similarities, alongside the crucial differences, between compounds and mixtures provides a more holistic view of chemical combinations, enabling us to appreciate the complexity and diversity of matter. It's this nuanced understanding that fuels advancements in various fields, from materials science and medicine to environmental science and engineering.
Conclusion: A Holistic Perspective on Chemical Combinations
The exploration of the similarities between compounds and mixtures unveils a deeper understanding of their nature, highlighting both their shared characteristics and fundamental differences. While both can exist in various physical states and be separated into their components, the methods of separation and the nature of the resulting products clearly distinguish between them. Recognizing these similarities, alongside the crucial differences, allows for a richer appreciation of chemical systems and the intricate interplay between compounds and mixtures in our everyday lives. This balanced perspective is crucial for developing a thorough understanding of chemistry and its applications across diverse scientific fields. Continued research and deeper investigation into these areas will surely reveal more subtle connections between compounds and mixtures.
Latest Posts
Latest Posts
-
Elements That Are Good Conductors Of Heat And Electricity Are
Apr 06, 2025
-
Which Of The Following Is Not A Property Of Bases
Apr 06, 2025
-
What Is Difference Between Gas And Vapour
Apr 06, 2025
-
Words That Begin And End With R
Apr 06, 2025
-
What Is The Tangent Of 30 Degrees
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How Are Compounds And Mixtures Alike . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
