What Is A Horizontal Row On The Periodic Table Called
Juapaving
Apr 04, 2025 · 6 min read
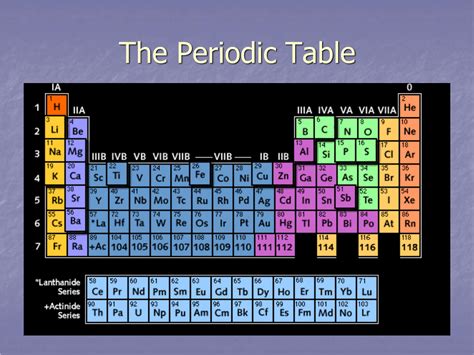
Table of Contents
What is a Horizontal Row on the Periodic Table Called? A Deep Dive into Periods and Their Significance
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic number and recurring chemical properties. Understanding its structure is crucial to grasping the fundamental principles of chemistry. One key aspect of this structure is the arrangement of elements into rows. But what exactly is a horizontal row on the periodic table called? The answer is simple: it's called a period. However, understanding the meaning behind the term "period" and its implications for the properties of elements within a row is far more complex and fascinating. This comprehensive guide delves into the nature of periods, exploring their significance, the trends observed within them, and their overall contribution to our understanding of the chemical world.
Understanding Periods: More Than Just a Row
A period in the periodic table represents a horizontal row of elements. Each period signifies the principal energy level (or shell) that the outermost electrons of the elements in that row occupy. This means elements within the same period have the same number of electron shells. This seemingly simple concept has profound implications for the properties of the elements involved.
The Significance of Electron Shells and Periodicity
The number of electron shells directly influences an element's atomic radius, ionization energy, electron affinity, and electronegativity. These properties, in turn, dictate how an element will behave chemically, influencing its reactivity and the types of compounds it can form.
-
Atomic Radius: As you move across a period from left to right, the atomic radius generally decreases. This is because the increasing nuclear charge attracts the electrons more strongly, pulling them closer to the nucleus.
-
Ionization Energy: Ionization energy is the energy required to remove an electron from an atom. Across a period, ionization energy generally increases. This is again due to the stronger nuclear pull on the electrons, making them harder to remove.
-
Electron Affinity: Electron affinity is the energy change when an electron is added to a neutral atom. While the trend isn't as consistent as ionization energy, generally, electron affinity increases across a period as the atom becomes more eager to gain an electron to achieve a stable electron configuration.
-
Electronegativity: Electronegativity is the ability of an atom to attract electrons in a chemical bond. Similar to ionization energy, electronegativity generally increases across a period due to the increased nuclear charge.
Exploring Each Period: A Detailed Look
Let's examine each period in more detail, highlighting the unique characteristics of the elements they contain:
Period 1: The Simplest Period
Period 1 is the shortest period, containing only two elements: hydrogen (H) and helium (He). Both elements have their electrons in the first principal energy level (n=1), which can hold a maximum of two electrons. Hydrogen is a reactive nonmetal, while helium is an inert noble gas.
Period 2: The Beginning of the 'p' Block
Period 2 contains eight elements, starting with lithium (Li) and ending with neon (Ne). This period introduces the p subshell, meaning elements in this period start to demonstrate a wider range of chemical behaviors. The period includes alkali metals (Li, Na), alkaline earth metals (Be, Mg), and halogens (F, Cl). The trend of increasing ionization energy and electronegativity is readily observable in this period.
Period 3: Similar Trends, Increased Complexity
Period 3, also with eight elements, mirrors many of the trends seen in period 2. It shows a similar progression of properties from alkali metals (Na) to noble gases (Ar). However, the increased number of electrons and the presence of d-orbitals in subsequent periods add complexity to the interactions.
Period 4 and Beyond: The Transition Metals and the Expanding Table
Periods 4 and beyond become considerably more complex due to the introduction of the d and f subshells, leading to the appearance of transition metals and inner transition metals (lanthanides and actinides). The transition metals show variable oxidation states, exhibiting a range of chemical properties. The f-block elements, particularly the actinides, are largely radioactive.
-
Transition Metals: These elements are found in the middle of the periodic table, between the s-block and the p-block elements. They are characterized by their ability to form multiple oxidation states, leading to diverse chemical behavior and vibrant coloration in many of their compounds. The transition metals are known for their catalytic properties and their importance in various industrial applications.
-
Inner Transition Metals (Lanthanides and Actinides): These elements are typically placed below the main body of the periodic table. The lanthanides and actinides are characterized by the filling of the 4f and 5f orbitals respectively. Many of the actinides are radioactive and have short half-lives.
The Importance of Periodicity
The repeating patterns of properties observed across periods are known as periodicity. This concept is fundamental to understanding the behavior of elements. By recognizing periodic trends, chemists can predict the properties of elements and understand their reactivity. This predictability is essential for designing new materials, understanding chemical reactions, and developing numerous applications across various scientific disciplines.
Using Periods to Predict Properties: Practical Applications
The knowledge of periodic trends within rows (periods) has numerous practical applications:
-
Predicting Reactivity: Chemists can predict the reactivity of an element based on its position within a period. For example, elements on the left side of a period (alkali metals) are highly reactive, while elements on the right side (noble gases) are inert.
-
Designing New Materials: By understanding the properties of elements within specific periods, materials scientists can design new materials with desired characteristics, such as strength, conductivity, or reactivity.
-
Understanding Chemical Reactions: The knowledge of periodic trends helps chemists understand how elements react with each other, forming the basis for predicting the outcome of chemical reactions.
-
Developing New Technologies: The principles of periodicity play a crucial role in the development of new technologies, including batteries, catalysts, and electronic devices.
Beyond the Basics: Further Exploration of Periodicity
While the basic understanding of periods as horizontal rows with similar outermost electron configurations is crucial, a deeper dive reveals more subtle yet significant details:
-
Shielding Effect: The inner electrons shield the outer electrons from the full nuclear charge, influencing the trends in atomic radius and ionization energy.
-
Electron-Electron Repulsion: The repulsion between electrons within the same shell can also influence the observed properties, sometimes leading to deviations from the expected trends.
-
Anomalous Behavior: Certain elements exhibit behavior that deviates slightly from the general trends observed within their periods. These anomalies are often caused by specific electronic configurations or other factors influencing electron-electron and electron-nucleus interactions.
Conclusion: The Enduring Importance of Periods in Chemistry
In conclusion, a horizontal row on the periodic table is called a period. While seemingly simple, the concept of periods and the understanding of the trends observed within them are fundamental to comprehending chemical behavior. The number of electron shells, the progressive filling of subshells, and the resulting variations in atomic properties offer a powerful framework for predicting and understanding the interactions of elements, driving innovations in materials science, chemical engineering, and countless other fields. The continued study of periods and their implications will undoubtedly uncover further nuances and contribute to an ever-deepening appreciation of the periodic table's importance as a cornerstone of scientific understanding.
Latest Posts
Latest Posts
-
What Is The Current Through 10 Volt Battery
Apr 04, 2025
-
Cam Plants Keep Stomata Closed In The Daytime
Apr 04, 2025
-
36 Inches Is How Many Centimeters
Apr 04, 2025
-
Five Letter Words Ending In O N
Apr 04, 2025
-
What Is Keratinization And Where Does It Occur
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is A Horizontal Row On The Periodic Table Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
