Do Electrons Flow From Anode To Cathode In Electrolytic Cell
Juapaving
Apr 03, 2025 · 5 min read
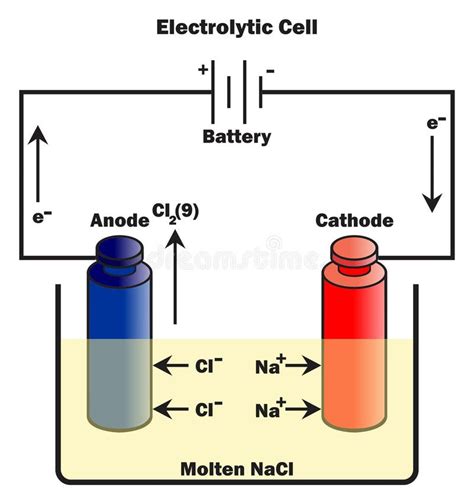
Table of Contents
Do Electrons Flow from Anode to Cathode in an Electrolytic Cell? Understanding Electron Flow in Electrolysis
The question of electron flow in an electrolytic cell – specifically, whether electrons flow from the anode to the cathode – is a fundamental concept in electrochemistry. While seemingly straightforward, a complete understanding requires delving into the definitions of anode and cathode in electrolytic cells, the nature of electrolytic processes, and the movement of ions and electrons within the system. This detailed explanation will clarify the process and address common misconceptions.
Defining Anode and Cathode in Electrolytic Cells
Unlike in galvanic (voltaic) cells where redox reactions spontaneously generate electricity, electrolytic cells require an external power source (like a battery) to drive a non-spontaneous redox reaction. This crucial difference affects the definition of anode and cathode.
Anode: Site of Oxidation
In electrolytic cells, the anode is the electrode where oxidation occurs. Oxidation is the loss of electrons. Therefore, at the anode, negatively charged electrons are released from the substance being oxidized. This is the opposite of what happens at the anode in a galvanic cell.
Cathode: Site of Reduction
Conversely, in electrolytic cells, the cathode is the electrode where reduction occurs. Reduction is the gain of electrons. At the cathode, positively charged ions (cations) gain electrons. Again, this is opposite to the behavior of the cathode in a galvanic cell.
Electron Flow: From Anode to Cathode Through the External Circuit
The key to understanding electron flow lies in recognizing the role of the external power source. The battery connected to the electrolytic cell provides the energy to drive the non-spontaneous redox reaction. This energy is supplied by forcing electrons to move through the external circuit.
The direction of electron flow in the external circuit is always from the anode to the cathode. The electrons released during oxidation at the anode travel through the external wire towards the cathode, driven by the potential difference supplied by the battery. This is a crucial distinction – while ions move within the electrolyte solution, electrons primarily flow through the external circuit.
Ion Movement within the Electrolyte Solution
While electrons travel through the external circuit, ions move within the electrolyte solution. This ionic movement completes the circuit and allows the overall redox reaction to proceed.
-
Anions (negatively charged ions): These ions migrate towards the anode. They are attracted by the positive charge build-up at the anode as electrons are released during oxidation.
-
Cations (positively charged ions): These ions migrate towards the cathode. They are attracted to the negative charge build-up at the cathode, where electrons are supplied by the external circuit for reduction.
The movement of ions within the electrolyte solution maintains electrical neutrality and facilitates the completion of the circuit.
Illustrative Example: Electrolysis of Water
Let’s consider the electrolysis of water as a clear example. Water is decomposed into hydrogen and oxygen gas using an electric current.
Electrolyte: Water (H₂O) with a small amount of an electrolyte, such as sulfuric acid or sodium hydroxide, added to improve conductivity.
Anode Reaction (Oxidation):
2H₂O(l) → O₂(g) + 4H⁺(aq) + 4e⁻
At the anode, water molecules lose electrons, forming oxygen gas, hydrogen ions, and releasing electrons.
Cathode Reaction (Reduction):
4H⁺(aq) + 4e⁻ → 2H₂(g)
At the cathode, hydrogen ions gain electrons, forming hydrogen gas.
Electron Flow:
- Electrons are released at the anode during the oxidation of water.
- These electrons flow through the external circuit towards the cathode (driven by the battery).
- At the cathode, electrons are consumed by hydrogen ions during their reduction.
The overall reaction is:
2H₂O(l) → 2H₂(g) + O₂(g)
Common Misconceptions and Clarifications
Several misconceptions often surround electron flow in electrolytic cells. Let’s address them:
Misconception 1: Electrons flow from cathode to anode.
This is incorrect for electrolytic cells. The external power source forces electrons to flow from the anode to the cathode, as explained above. This is in contrast to galvanic cells, where electrons flow spontaneously from the anode to the cathode.
Misconception 2: Electrons flow directly through the electrolyte.
While ions move through the electrolyte, electrons primarily travel through the external circuit. The electrolyte facilitates ionic conductivity, not electron conductivity. The direct flow of electrons through the electrolyte would be a short circuit, not a controlled electrolytic process.
Misconception 3: The anode is always positive and the cathode is always negative.
This statement is only true when discussing the external connections. Internally, within the electrolytic cell, the charge distribution is dynamic and affected by the electrode reactions and ion movement. The anode develops a positive charge relative to the cathode because of the loss of electrons during oxidation, while the cathode accumulates a negative charge due to the gain of electrons during reduction. But the overall polarity of the electrodes relative to the battery connections remains consistent with the definitions above.
The Importance of Understanding Electron Flow
Understanding the direction of electron flow in electrolytic cells is vital for:
- Predicting the products of electrolysis: Knowing which electrode attracts which ion helps predict the outcome of the electrolysis process.
- Designing and optimizing electrolytic cells: Engineers must understand electron flow to design effective and efficient electrolytic cells for various industrial applications.
- Interpreting electrochemical data: Understanding the flow of electrons aids in the interpretation of experimental data obtained from electrochemical studies.
- Understanding related concepts: Electrolytic cells are a crucial foundation for understanding other related electrochemical concepts like electroplating, electrorefining, and the production of various chemicals.
Conclusion
In summary, electrons in an electrolytic cell flow from the anode to the cathode through the external circuit, driven by the external power source. While ions migrate within the electrolyte solution, their movement complements electron flow through the external circuit to complete the electrical circuit and allow the non-spontaneous redox reaction to occur. Grasping this fundamental principle is crucial for a thorough understanding of electrochemistry and its numerous applications. Remembering the difference between electrolytic and galvanic cells and clarifying your understanding of the anode and cathode in each context is essential for avoiding common misconceptions. By carefully considering the movement of both electrons and ions, a clear and accurate picture of the processes within electrolytic cells can be formed.
Latest Posts
Latest Posts
-
What Are Two Categories Of Pure Substances
Apr 03, 2025
-
1 Bromobutane Primary Secondary Or Tertiary
Apr 03, 2025
-
Does A Solid Have A Definite Volume
Apr 03, 2025
-
What Is A Property Of A Base
Apr 03, 2025
-
Difference Between Physical Mapping And Genetic Mapping
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Do Electrons Flow From Anode To Cathode In Electrolytic Cell . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
