What Is A Property Of A Base
Juapaving
Apr 03, 2025 · 6 min read
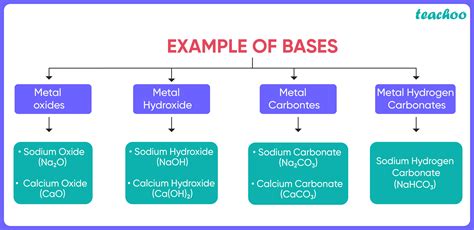
Table of Contents
What is a Property of a Base? A Comprehensive Guide
Understanding the properties of bases is fundamental to chemistry. Bases, along with acids, form the cornerstone of acid-base chemistry, a crucial concept across numerous scientific disciplines. This comprehensive guide will delve deep into the defining characteristics of bases, exploring their various properties from a theoretical and practical standpoint. We will cover key concepts such as Arrhenius bases, Brønsted-Lowry bases, and Lewis bases, showcasing their unique properties and highlighting the distinctions between them.
Defining Bases: Different Perspectives
The definition of a base has evolved over time, with different models offering unique insights. Understanding these different perspectives is crucial to grasping the multifaceted nature of bases.
1. Arrhenius Bases: The Hydroxide Connection
The Arrhenius definition, one of the earliest models, defines a base as a substance that increases the hydroxide ion (OH⁻) concentration when dissolved in water. This definition is straightforward and focuses on the direct impact of the base on the aqueous solution. Classic examples include metal hydroxides like sodium hydroxide (NaOH) and potassium hydroxide (KOH), which readily dissociate in water to release OH⁻ ions, thus increasing the solution's pH.
Key characteristics of Arrhenius bases:
- Produce OH⁻ ions in aqueous solutions: This is their defining feature.
- Typically metal hydroxides: Many common Arrhenius bases fall under this category.
- Limited scope: This definition is limited as it only applies to aqueous solutions and doesn't encompass all substances that exhibit basic properties.
2. Brønsted-Lowry Bases: Proton Acceptors
The Brønsted-Lowry definition offers a broader perspective. A Brønsted-Lowry base is defined as a proton acceptor. This definition transcends the limitations of the Arrhenius definition by focusing on the base's ability to accept a proton (H⁺) rather than its specific behavior in water. This allows for a wider range of substances to be classified as bases, even those that don't directly produce OH⁻ ions.
Key characteristics of Brønsted-Lowry bases:
- Accept protons (H⁺): This is their defining characteristic.
- Includes a wider range of substances: Ammonia (NH₃), for instance, acts as a Brønsted-Lowry base by accepting a proton to form the ammonium ion (NH₄⁺).
- Applies to non-aqueous solutions: This definition isn't limited to aqueous solutions.
3. Lewis Bases: Electron Pair Donors
The Lewis definition provides the most expansive view of basicity. A Lewis base is defined as an electron pair donor. This definition focuses on the base's ability to donate a pair of electrons to form a coordinate covalent bond with an electron-deficient species (a Lewis acid). This definition encompasses all Brønsted-Lowry bases and extends the concept of basicity to numerous other compounds that may not fit the previous definitions.
Key characteristics of Lewis bases:
- Donate electron pairs: This is their defining characteristic.
- Includes a vast range of compounds: Many molecules and ions with lone pairs of electrons can act as Lewis bases, including water, ammonia, and halide ions.
- Expands the scope of acid-base chemistry: This definition opens up acid-base reactions beyond proton transfer, incorporating reactions involving electron pair donation.
Properties of Bases: A Detailed Exploration
Now that we have established the various definitions of bases, let's explore their key properties in detail. These properties are observed across different types of bases and manifest in various ways.
1. pH Greater Than 7: The Alkaline Nature
A fundamental property of bases is their ability to increase the pH of a solution above 7. This is a direct consequence of the increased hydroxide ion (OH⁻) concentration in aqueous solutions, as per the Arrhenius definition. A higher pH indicates a more alkaline solution, contrasting with acidic solutions that have a pH below 7. The pH scale provides a quantitative measure of the acidity or basicity of a solution.
2. Taste: Bitter and Caustic
While not a reliable method for identifying bases in a laboratory setting, many bases possess a characteristic bitter taste and can be caustic or corrosive to living tissue. This property highlights the importance of handling bases with care, always using appropriate safety precautions.
3. Feel: Slippery or Soapy
Another characteristic property, particularly notable in strong bases, is their slippery or soapy feel. This is due to the interaction of the base with the oils and proteins on the skin, producing a saponification reaction. This reaction alters the skin's surface, leading to the characteristic slippery texture. However, it's crucial to avoid direct contact with strong bases as they can cause severe burns.
4. Reaction with Acids: Neutralization
Bases react with acids in a process called neutralization. This reaction involves the combination of H⁺ ions from the acid and OH⁻ ions from the base to form water (H₂O). The other product of the reaction is typically a salt. This neutralization reaction is a fundamental concept in acid-base chemistry, often used in titrations to determine the concentration of an unknown acid or base.
Example: NaOH (aq) + HCl (aq) → NaCl (aq) + H₂O (l)
5. Conductivity: Electrical Conductance
Strong bases, which fully dissociate in water, are good electrical conductors in solution. This is due to the presence of freely moving ions (cations and anions) that carry the electric current. Weak bases, on the other hand, exhibit lower conductivity due to their partial dissociation.
6. Indicators: Color Change
Many chemical indicators change color depending on the pH of the solution. Bases can cause these indicators to change color, signifying the presence of a base in the solution. Examples include litmus paper (turning blue in the presence of a base) and phenolphthalein (turning pink in alkaline solutions). These indicators are commonly used in titrations to visually monitor the progress of a neutralization reaction.
7. Reactivity with Metals: Metal Reactions
Certain bases react with certain metals, particularly amphoteric metals like aluminum and zinc, to produce hydrogen gas (H₂). This reaction is a characteristic property of strongly basic solutions and is an important consideration in handling and storing these materials.
Example: 2Al (s) + 6NaOH (aq) → 2Na₃AlO₃ (aq) + 3H₂ (g)
8. Effect on Solubility: Dissolution of Insoluble Compounds
Some bases, such as ammonia, can improve the solubility of certain insoluble metal compounds through complex formation. These reactions lead to increased dissolution of the metal compounds in the presence of base, affecting the overall solubility equilibrium.
Examples of Bases and their Applications
Numerous substances exhibit basic properties, finding wide applications across various fields.
Strong Bases:
- Sodium hydroxide (NaOH): Used in soap making, paper production, and drain cleaners.
- Potassium hydroxide (KOH): Used in fertilizer production, and as an electrolyte in alkaline batteries.
- Calcium hydroxide (Ca(OH)₂): Used in mortar and plaster, and as a pH adjuster in water treatment.
Weak Bases:
- Ammonia (NH₃): Used in fertilizers, cleaning products, and as a refrigerant.
- Sodium carbonate (Na₂CO₃): Used in glass manufacturing, detergents, and water softening.
- Sodium bicarbonate (NaHCO₃): Used in baking soda, antacids, and fire extinguishers.
Conclusion
The properties of bases are multifaceted and depend on the specific definition employed. From the straightforward Arrhenius definition to the more encompassing Brønsted-Lowry and Lewis definitions, understanding the different perspectives allows for a complete appreciation of the diverse range of substances that exhibit basic properties. Their characteristic properties, such as increased pH, bitter taste, slippery feel, and reactivity with acids, are vital in identifying and utilizing bases in various scientific and industrial applications. The ability to understand and apply this knowledge is fundamental to a wide array of chemical processes and technological advancements. Further exploration into the intricacies of acid-base chemistry will undoubtedly reveal even more fascinating aspects of these fundamental chemical entities.
Latest Posts
Latest Posts
-
Describing Words That Start With P
Apr 04, 2025
-
Which Of The Following Are Functions Of The Skeletal System
Apr 04, 2025
-
Find The Area Of The Region Calculator
Apr 04, 2025
-
Glycogen Cellulose And Starch Are All
Apr 04, 2025
-
The Stored Form Of Carbohydrate In Humans Is Called Term1
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is A Property Of A Base . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
