1 Bromobutane Primary Secondary Or Tertiary
Juapaving
Apr 03, 2025 · 5 min read
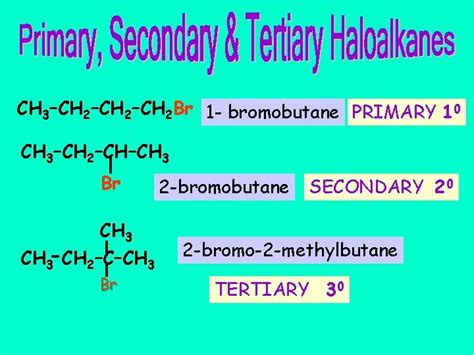
Table of Contents
1-Bromobutane: Primary, Secondary, or Tertiary? Understanding Alkyl Halides
Keywords: 1-bromobutane, primary alkyl halide, secondary alkyl halide, tertiary alkyl halide, alkyl halide classification, organic chemistry, halogenoalkane, SN1 reaction, SN2 reaction, electrophilic substitution, nucleophilic substitution
Understanding the classification of alkyl halides, specifically whether a given compound is primary, secondary, or tertiary, is fundamental in organic chemistry. This knowledge is crucial for predicting reactivity and understanding the mechanisms of various reactions. This article delves into the classification of 1-bromobutane, explaining the principles behind this categorization and its implications for chemical behavior.
What are Alkyl Halides?
Alkyl halides, also known as haloalkanes or halogenoalkanes, are organic compounds derived from alkanes by replacing one or more hydrogen atoms with halogen atoms (fluorine, chlorine, bromine, or iodine). The general formula is R-X, where R represents an alkyl group and X represents a halogen atom. Their properties and reactivity are significantly influenced by the nature of the alkyl group and the halogen atom.
Classifying Alkyl Halides: Primary, Secondary, and Tertiary
The classification of alkyl halides as primary (1°), secondary (2°), or tertiary (3°) depends on the number of carbon atoms directly bonded to the carbon atom bearing the halogen.
-
Primary (1°) alkyl halide: The carbon atom bonded to the halogen is directly attached to only one other carbon atom.
-
Secondary (2°) alkyl halide: The carbon atom bonded to the halogen is directly attached to two other carbon atoms.
-
Tertiary (3°) alkyl halide: The carbon atom bonded to the halogen is directly attached to three other carbon atoms.
1-Bromobutane: A Detailed Analysis
Now, let's analyze 1-bromobutane. Its structural formula is CH₃CH₂CH₂CH₂Br. The bromine atom is attached to a carbon atom at the end of the butane chain. This carbon atom is only bonded to one other carbon atom (the second carbon in the chain).
Therefore, 1-bromobutane is classified as a primary (1°) alkyl halide.
Reactivity and Reaction Mechanisms: SN1 vs. SN2
The classification of an alkyl halide as primary, secondary, or tertiary significantly influences its reactivity and the preferred reaction mechanism it undergoes. Two common nucleophilic substitution reactions are SN1 and SN2.
SN2 Reactions (Bimolecular Nucleophilic Substitution)
SN2 reactions are concerted, meaning the bond breaking and bond forming occur simultaneously in a single step. The nucleophile attacks the carbon atom bearing the halogen from the backside, leading to inversion of configuration. The rate of an SN2 reaction depends on the concentration of both the alkyl halide and the nucleophile.
Steric hindrance plays a crucial role in SN2 reactions. Primary alkyl halides are the most reactive in SN2 reactions because they experience minimal steric hindrance. Secondary alkyl halides react slower, and tertiary alkyl halides are essentially unreactive in SN2 reactions due to significant steric hindrance.
Since 1-bromobutane is a primary alkyl halide, it is highly reactive in SN2 reactions.
SN1 Reactions (Unimolecular Nucleophilic Substitution)
SN1 reactions proceed through a two-step mechanism. The first step involves the formation of a carbocation intermediate by the departure of the leaving group (halogen). The second step involves the nucleophile attacking the carbocation. The rate of an SN1 reaction depends only on the concentration of the alkyl halide.
Carbocation stability plays a critical role in SN1 reactions. Tertiary carbocations are the most stable, followed by secondary, and primary carbocations are the least stable. Therefore, tertiary alkyl halides are the most reactive in SN1 reactions, followed by secondary, and primary alkyl halides are essentially unreactive under typical SN1 conditions.
Because 1-bromobutane is a primary alkyl halide, it forms a relatively unstable primary carbocation. Consequently, it is unreactive in SN1 reactions under normal conditions.
Factors Affecting Reactivity Beyond Primary, Secondary, Tertiary Classification
While the primary, secondary, tertiary classification is a powerful predictor of reactivity, other factors also influence the reaction rates and mechanisms of alkyl halides:
-
The nature of the halogen: The leaving group ability of the halogen affects the reaction rate. Iodine is the best leaving group, followed by bromine, chlorine, and fluorine. Therefore, 1-iodobutane would be more reactive than 1-bromobutane in both SN1 and SN2 reactions.
-
The nature of the nucleophile: Stronger nucleophiles react faster in SN2 reactions. The solvent also plays a significant role. Polar aprotic solvents favor SN2 reactions, while polar protic solvents favor SN1 reactions.
-
Steric effects: Even within primary alkyl halides, steric hindrance from bulky groups can influence the reaction rate.
-
Solvent effects: The solvent can significantly influence the reaction mechanism and rate. Polar protic solvents stabilize the carbocation intermediate, favoring SN1 reactions. Polar aprotic solvents favor SN2 reactions.
Applications of 1-Bromobutane and Alkyl Halides
Alkyl halides, including 1-bromobutane, are versatile intermediates in organic synthesis. They serve as building blocks for a vast array of organic compounds. Some of their applications include:
-
Synthesis of Grignard reagents: Grignard reagents are organomagnesium halides (RMgX) widely used in organic synthesis for carbon-carbon bond formation. 1-bromobutane can be used to synthesize the butylmagnesium bromide Grignard reagent.
-
Synthesis of amines: Alkyl halides can be used to synthesize amines via nucleophilic substitution reactions with ammonia or primary/secondary amines.
-
Synthesis of ethers: Alkyl halides can react with alkoxides to form ethers (Williamson ether synthesis).
-
Synthesis of nitriles: Alkyl halides can react with cyanide ions to form nitriles.
Conclusion
1-bromobutane is definitively a primary alkyl halide. This classification is crucial for understanding its reactivity and predicting its behavior in various reactions. Its preference for SN2 reactions over SN1 reactions stems directly from this classification and the relative instability of primary carbocations. While the primary, secondary, tertiary classification provides a valuable framework for predicting reactivity, other factors like the nature of the halogen, nucleophile, and solvent also play significant roles in determining the reaction mechanism and rate. Alkyl halides are essential building blocks in organic synthesis, showcasing their importance in the creation of a wide variety of organic compounds. Understanding their classification and reactivity is crucial for anyone studying or working in organic chemistry.
Latest Posts
Latest Posts
-
What Is The Square Root Of 112
Apr 04, 2025
-
Balanced Equation For Nitric Acid And Sodium Hydroxide
Apr 04, 2025
-
Energy Of Electron In Nth Orbit Formula
Apr 04, 2025
-
Find Moment Of Inertia Of Solid Sphere
Apr 04, 2025
-
6 Letter Word Starting With Sta
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about 1 Bromobutane Primary Secondary Or Tertiary . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
