Difference Between Starch Glycogen And Cellulose
Juapaving
Apr 05, 2025 · 5 min read
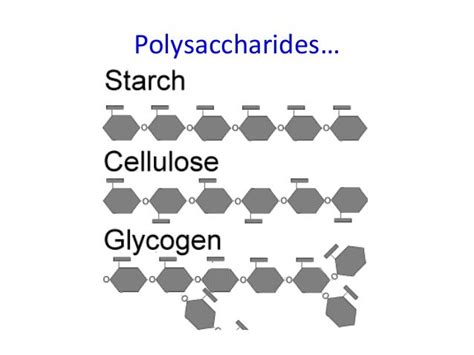
Table of Contents
The Crucial Differences Between Starch, Glycogen, and Cellulose: A Deep Dive
Polysaccharides are complex carbohydrates formed by the joining of many monosaccharide units. Among the most important polysaccharides are starch, glycogen, and cellulose, all composed of glucose units but exhibiting strikingly different structures and functions. These differences stem from variations in the type of glycosidic bonds and the branching patterns within the molecules, leading to vastly different properties and roles in living organisms. This article delves deep into the distinctions between starch, glycogen, and cellulose, exploring their structures, functions, and significance in biology and human nutrition.
Understanding the Building Blocks: Glucose and Glycosidic Bonds
Before exploring the differences, let's establish a common ground. All three – starch, glycogen, and cellulose – are polymers of glucose, a simple sugar. The glucose units are linked together via glycosidic bonds, covalent bonds formed between the hydroxyl (-OH) groups of adjacent glucose molecules. However, the type of glycosidic bond and the manner in which these bonds are formed significantly influence the overall structure and properties of each polysaccharide.
Alpha (α) vs. Beta (β) Glycosidic Bonds: The Key Distinction
The crucial difference lies in the configuration of the glycosidic bonds. Starch and glycogen contain α-1,4-glycosidic bonds and α-1,6-glycosidic bonds (at branch points), while cellulose contains β-1,4-glycosidic bonds. This seemingly minor difference in bond orientation has profound consequences on the three-dimensional structure and biological function of these polymers.
Starch: The Energy Storage Polysaccharide of Plants
Starch serves as the primary energy storage polysaccharide in plants. It exists in two main forms: amylose and amylopectin.
Amylose: A Linear Chain of Glucose
Amylose consists of long, unbranched chains of glucose units linked by α-1,4-glycosidic bonds. This linear structure allows the molecule to coil into a helix, creating a compact structure suitable for storage. The helical structure also provides opportunities for interactions with iodine, resulting in the characteristic blue-black color observed in the iodine test for starch.
Amylopectin: A Branched Structure for Efficient Storage
Amylopectin, the more abundant form of starch, has a branched structure. It also primarily consists of α-1,4-glycosidic bonds, but it introduces α-1,6-glycosidic bonds at branch points approximately every 24-30 glucose units. These branches create a more compact and readily accessible structure for energy mobilization. The branched nature allows enzymes to readily access and hydrolyze glucose units, facilitating rapid energy release when the plant needs it.
Starch Digestion and Human Nutrition
Humans possess enzymes, such as amylase, that can effectively hydrolyze the α-glycosidic bonds in starch, releasing glucose units that can be used for energy production. This is why starch is a crucial component of the human diet, providing a significant source of calories.
Glycogen: The Animal Energy Storage Champion
Glycogen is the primary energy storage polysaccharide in animals and fungi. Its structure closely resembles amylopectin, with a highly branched structure formed by α-1,4-glycosidic bonds and α-1,6-glycosidic bonds at branch points. However, glycogen is even more highly branched than amylopectin, with branches occurring more frequently.
High Branching Density for Rapid Glucose Release
The high degree of branching in glycogen is crucial for its function. The numerous branch points provide numerous sites for enzyme action, enabling the rapid mobilization of glucose when energy demands are high. This rapid release is essential for maintaining blood glucose levels and providing energy for muscle contraction and other metabolic processes.
Glycogen Storage and Depletion
Glycogen is stored primarily in the liver and muscles. Liver glycogen serves as a buffer to maintain blood glucose levels, releasing glucose into the bloodstream when needed. Muscle glycogen provides a readily available energy source for muscle contraction. During periods of fasting or intense exercise, glycogen stores can be rapidly depleted.
Cellulose: The Structural Polysaccharide of Plants
Unlike starch and glycogen, cellulose serves a structural role in plants, forming the main component of plant cell walls. Cellulose consists of long, unbranched chains of glucose units linked by β-1,4-glycosidic bonds.
Beta-Glycosidic Bonds and Linear Structure
The β-1,4-glycosidic bonds cause cellulose molecules to adopt a straight, extended conformation. This linear structure allows cellulose molecules to pack tightly together, forming strong, parallel microfibrils held together by hydrogen bonds. This arrangement gives cellulose its remarkable tensile strength and rigidity, crucial for providing structural support to plants.
Cellulose Digestion: A Challenge for Humans
Humans lack the enzyme cellulase, which is necessary to hydrolyze the β-1,4-glycosidic bonds in cellulose. As a result, humans cannot digest cellulose, and it passes through the digestive system as dietary fiber. However, cellulose is still an important part of a healthy diet, as it promotes gut health, regulates bowel movements, and may have other beneficial effects. Herbivores, on the other hand, possess microorganisms in their digestive tracts that produce cellulase, allowing them to digest cellulose and obtain energy from plant matter.
A Comparative Table: Starch, Glycogen, and Cellulose
| Feature | Starch | Glycogen | Cellulose |
|---|---|---|---|
| Monomer | Glucose | Glucose | Glucose |
| Glycosidic Bond | α-1,4 and α-1,6 (branched) | α-1,4 and α-1,6 (highly branched) | β-1,4 (unbranched) |
| Structure | Linear (amylose), branched (amylopectin) | Highly branched | Linear, unbranched |
| Function | Energy storage in plants | Energy storage in animals | Structural support in plants |
| Digestibility in Humans | Digestible | Digestible | Indigestible |
Beyond the Basics: Further Exploration
The differences between starch, glycogen, and cellulose extend beyond their basic structural features. Their solubility, reactivity, and interactions with other molecules are also significantly influenced by their unique structures. For example, starch is readily soluble in hot water, forming a colloid, while cellulose is insoluble. These differences highlight the remarkable diversity of polysaccharides and their tailored functions within living systems.
The study of these polysaccharides also has important implications for various fields, including:
- Food Science: Understanding starch structure is critical for developing food products with desired textural properties.
- Biotechnology: Cellulose is being investigated as a renewable resource for the production of biofuels and other biomaterials.
- Medicine: Glycogen metabolism is crucial in understanding and managing metabolic diseases such as diabetes.
This article provides a comprehensive overview of the key differences between starch, glycogen, and cellulose. By understanding the intricacies of their structures and functions, we can gain a deeper appreciation for the remarkable diversity and importance of polysaccharides in the biological world and their impact on human health and nutrition. Further research into these molecules continues to uncover new insights and potential applications.
Latest Posts
Latest Posts
-
What Is A Political Party Class 10
Apr 06, 2025
-
Which Of The Following Is An Even Function
Apr 06, 2025
-
How Do You Describe A Book
Apr 06, 2025
-
What Do Both Prokaryotes And Eukaryotes Have
Apr 06, 2025
-
What Is A Whole Number But Not A Natural Number
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Difference Between Starch Glycogen And Cellulose . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
