Difference Between Starch Cellulose And Glycogen
Juapaving
Apr 02, 2025 · 5 min read
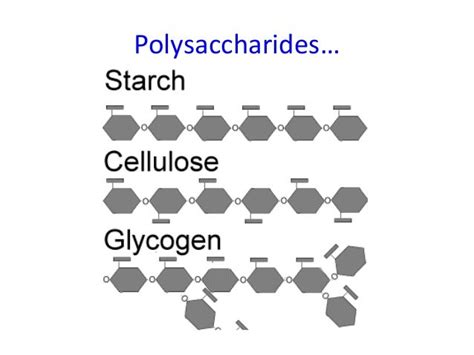
Table of Contents
The Crucial Differences Between Starch, Cellulose, and Glycogen: A Deep Dive
Carbohydrates are essential biomolecules providing energy and structural support to living organisms. Three prominent members of this class – starch, cellulose, and glycogen – share a common building block: glucose. However, their structural differences lead to vastly different properties and biological roles. Understanding these differences is key to appreciating their importance in various biological processes and industrial applications. This comprehensive guide will explore the distinctions between starch, cellulose, and glycogen in detail.
Understanding the Glucose Monomer
Before diving into the specifics of starch, cellulose, and glycogen, it's vital to understand their fundamental unit: glucose. Glucose is a simple sugar (monosaccharide) with the chemical formula C₆H₁₂O₆. It exists in two forms: α-glucose and β-glucose, which differ only in the orientation of a hydroxyl (-OH) group on carbon atom 1. This seemingly minor difference has profound implications for the resulting polysaccharide's structure and function.
Starch: The Energy Store of Plants
Starch is a polysaccharide, meaning it's a long chain of glucose molecules linked together. It's the primary energy storage form in plants. Starch exists in two main forms:
Amylose: The Linear Chain
Amylose is a linear chain of α-glucose molecules linked by α-1,4-glycosidic bonds. This means the bond connects carbon atom 1 of one glucose molecule to carbon atom 4 of the next. The linear structure allows amylose to form a helical coil, making it compact and efficient for storage.
Amylopectin: The Branched Chain
Amylopectin is a branched chain of α-glucose molecules. Like amylose, the main chain is linked by α-1,4-glycosidic bonds. However, amylopectin also contains α-1,6-glycosidic branches approximately every 24-30 glucose units. These branches create a more compact and readily accessible structure compared to amylose. This branching is crucial for efficient enzymatic breakdown and glucose release when the plant needs energy.
Cellulose: The Structural Backbone of Plants
Cellulose, unlike starch, is the primary structural component of plant cell walls. It's also a polysaccharide composed of glucose units, but the key difference lies in the type of glycosidic bond: β-1,4-glycosidic bonds.
This seemingly minor change in bond configuration has massive consequences. β-1,4-glycosidic bonds produce a linear structure that is much less flexible than the α-1,4-glycosidic bonds in starch. These linear cellulose chains aggregate together through hydrogen bonding to form strong, rigid microfibrils, providing the structural integrity necessary for plant cell walls. This strong, insoluble structure makes cellulose indigestible for most animals, including humans. However, some microorganisms possess the necessary enzymes (cellulases) to break down cellulose.
The Importance of Hydrogen Bonding in Cellulose
Hydrogen bonding plays a critical role in the structure and properties of cellulose. The hydroxyl groups (-OH) on the glucose molecules form hydrogen bonds with each other and with water molecules. This extensive hydrogen bonding network contributes to the high tensile strength and insolubility of cellulose. This makes it ideal for providing structural support to plants.
Glycogen: The Animal Energy Reserve
Glycogen is the main energy storage polysaccharide in animals and fungi. Similar to amylopectin, it's a highly branched structure composed of α-glucose molecules linked by α-1,4-glycosidic bonds in the main chain and α-1,6-glycosidic branches. However, glycogen has a higher degree of branching than amylopectin, with branches occurring approximately every 8-12 glucose units.
This extensive branching allows for rapid glucose mobilization when energy is required. The numerous branch points provide many sites for enzymatic action, enabling the efficient breakdown of glycogen into glucose molecules for immediate energy use. Glycogen is stored primarily in the liver and muscles, readily available to meet the body's energy demands.
Glycogen's Rapid Energy Release Mechanism
The high degree of branching in glycogen is crucial for its role as a rapid energy source. When energy is needed, enzymes can simultaneously access many glucose units along the branches, leading to rapid glucose release. This contrasts with the slower release from starch, which has fewer branch points.
A Comparative Table: Starch, Cellulose, and Glycogen
| Feature | Starch | Cellulose | Glycogen |
|---|---|---|---|
| Monomer | α-glucose | β-glucose | α-glucose |
| Glycosidic Bond | α-1,4 (and α-1,6 in amylopectin) | β-1,4 | α-1,4 (and α-1,6) |
| Structure | Linear (amylose), Branched (amylopectin) | Linear, highly organized | Highly branched |
| Function | Energy storage in plants | Structural support in plants | Energy storage in animals |
| Solubility | Soluble in water | Insoluble in water | Soluble in water |
| Digestibility | Digestible by many organisms | Digestible by few organisms | Digestible by animals |
Industrial Applications
The distinct properties of starch, cellulose, and glycogen lead to diverse industrial applications:
Starch Applications:
- Food Industry: Starch is used as a thickener, stabilizer, and binder in various food products.
- Textile Industry: Starch is used as a sizing agent in textiles.
- Paper Industry: Starch is used as a binder and coating agent in paper production.
Cellulose Applications:
- Paper Industry: Cellulose is the primary component of paper.
- Textile Industry: Cellulose is used to produce fabrics like cotton and linen.
- Biofuel Production: Cellulose can be converted into biofuels.
Glycogen Applications:
Glycogen's industrial applications are limited compared to starch and cellulose, primarily due to its higher cost and availability challenges. However, it has some niche applications in research and pharmaceuticals.
Conclusion: The Significance of Structural Differences
The seemingly subtle differences in the glycosidic linkages and branching patterns of starch, cellulose, and glycogen have profound consequences for their properties and biological functions. Starch provides readily available energy storage for plants, cellulose offers structural rigidity for plant cell walls, and glycogen serves as a rapidly mobilizable energy reserve for animals. Understanding these differences is crucial not only for appreciating the intricacies of biological systems but also for developing various industrial applications leveraging the unique properties of these essential polysaccharides. Further research continues to uncover new and innovative uses for these abundant and versatile biomolecules. The development of sustainable and efficient methods for processing and utilizing these resources is of growing importance in the context of a global drive towards bio-based solutions and reduced reliance on fossil fuels.
Latest Posts
Latest Posts
-
Comparison Between Light Microscope And Electron Microscope
Apr 03, 2025
-
Equation For Lead Nitrate And Potassium Iodide
Apr 03, 2025
-
Common Name For Sodium Hydrogen Carbonate
Apr 03, 2025
-
The Rows On The Periodic Table Are Called
Apr 03, 2025
-
Can A Percent Of Change Be Greater Than 100
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Difference Between Starch Cellulose And Glycogen . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
