Difference Between Molecular Geometry And Electron Geometry
Juapaving
Apr 05, 2025 · 6 min read
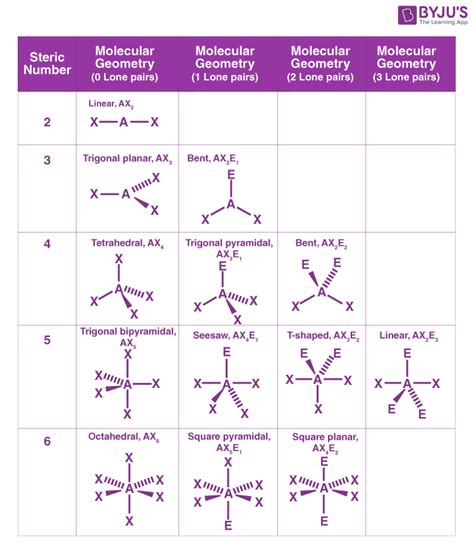
Table of Contents
Decoding the Differences: Molecular Geometry vs. Electron Geometry
Understanding the three-dimensional arrangement of atoms within a molecule is crucial in chemistry. This arrangement dictates a molecule's properties, influencing everything from its reactivity to its physical state. However, two closely related yet distinct concepts often cause confusion: molecular geometry and electron geometry. While both describe the spatial arrangement of atoms and electron pairs, they differ significantly in what they consider. This article delves deep into the nuances of these concepts, providing a clear and comprehensive explanation to eliminate any ambiguity.
What is Electron Geometry?
Electron geometry focuses on the arrangement of all electron domains around the central atom in a molecule. These electron domains include both bonding pairs (electrons shared between atoms in covalent bonds) and lone pairs (non-bonding electrons associated with the central atom). It's a description of the ideal spatial arrangement, ignoring the effects of lone pair repulsion.
Determining Electron Geometry: The VSEPR Theory
The Valence Shell Electron Pair Repulsion (VSEPR) theory is the cornerstone of predicting electron geometry. VSEPR postulates that electron pairs, whether bonding or non-bonding, repel each other and arrange themselves to minimize this repulsion. This results in specific electron geometries, depending on the number of electron domains surrounding the central atom.
Common Electron Geometries:
- Linear: Two electron domains. The bond angle is 180°. Example: BeCl₂
- Trigonal Planar: Three electron domains. The bond angle is approximately 120°. Example: BF₃
- Tetrahedral: Four electron domains. The bond angle is approximately 109.5°. Example: CH₄
- Trigonal Bipyramidal: Five electron domains. The bond angles are 90° and 120°. Example: PCl₅
- Octahedral: Six electron domains. The bond angles are 90°. Example: SF₆
It's vital to remember that electron geometry only considers the number of electron domains, not their nature (bonding vs. lone pairs).
The Influence of Lone Pairs: A Subtle but Significant Factor
While VSEPR theory provides a framework for predicting electron geometry, it's important to acknowledge the influence of lone pairs. Lone pairs occupy more space than bonding pairs due to their greater electron density and less concentrated nature. This results in slight deviations from the ideal bond angles predicted by VSEPR. However, the overall arrangement of electron domains still follows the basic VSEPR geometries. The impact of lone pairs is more prominently observed when considering molecular geometry.
What is Molecular Geometry?
Molecular geometry, also known as molecular shape, focuses solely on the arrangement of atoms in a molecule. Unlike electron geometry, it ignores the positions of lone pairs. Molecular geometry is the actual three-dimensional structure that can be experimentally determined using techniques like X-ray diffraction.
The Role of Lone Pairs in Determining Molecular Geometry
While lone pairs are not explicitly considered in defining molecular geometry, their presence significantly influences the shape. The repulsion between lone pairs and bonding pairs causes the bonding pairs to be compressed, leading to deviations from the ideal electron geometry. This results in different molecular shapes even if the electron geometry is the same.
Common Molecular Geometries:
The molecular geometry is often described in relation to the electron geometry, highlighting the impact of lone pairs. Here are some examples:
- Linear: Two bonding pairs, zero lone pairs. Example: BeCl₂ (both electron and molecular geometry are linear).
- Trigonal Planar: Three bonding pairs, zero lone pairs. Example: BF₃ (both electron and molecular geometry are trigonal planar).
- Bent (V-shaped): Three electron domains, one or two lone pairs. Example: SO₂ (electron geometry is trigonal planar, molecular geometry is bent). H₂O (electron geometry is tetrahedral, molecular geometry is bent).
- Tetrahedral: Four bonding pairs, zero lone pairs. Example: CH₄ (both electron and molecular geometry are tetrahedral).
- Trigonal Pyramidal: Four electron domains, one lone pair. Example: NH₃ (electron geometry is tetrahedral, molecular geometry is trigonal pyramidal).
- See-saw: Five electron domains, one lone pair. Example: SF₄ (electron geometry is trigonal bipyramidal, molecular geometry is see-saw).
- T-shaped: Five electron domains, two lone pairs. Example: ClF₃ (electron geometry is trigonal bipyramidal, molecular geometry is T-shaped).
- Linear (with lone pairs): Five electron domains, three lone pairs. Example: XeF₂ (electron geometry is trigonal bipyramidal, molecular geometry is linear).
- Square Planar: Six electron domains, two lone pairs. Example: XeF₄ (electron geometry is octahedral, molecular geometry is square planar).
- Square Pyramidal: Six electron domains, one lone pair. Example: BrF₅ (electron geometry is octahedral, molecular geometry is square pyramidal).
Key Differences Summarized
The fundamental difference between electron geometry and molecular geometry lies in what they consider:
| Feature | Electron Geometry | Molecular Geometry |
|---|---|---|
| Focus | Arrangement of all electron domains (bonding and lone pairs) | Arrangement of atoms only |
| Consideration of Lone Pairs | Includes lone pairs in determining shape | Ignores lone pairs; their effect is reflected in the deviation from ideal shape |
| Purpose | Provides a framework for understanding the overall spatial distribution of electrons | Describes the actual 3D shape of the molecule |
| Predictive Tool | VSEPR theory | VSEPR theory, but modified to account for lone pair repulsion |
The Importance of Distinguishing Between the Two
Understanding the difference between electron and molecular geometry is crucial for several reasons:
- Predicting Molecular Properties: Molecular geometry directly influences a molecule's polarity, reactivity, and physical properties like boiling point and melting point. The presence of lone pairs, although not directly part of the molecular geometry description, dramatically impacts these properties.
- Interpreting Spectroscopic Data: Spectroscopic techniques like infrared (IR) and nuclear magnetic resonance (NMR) spectroscopy provide insights into molecular structure. Interpreting this data requires understanding both electron and molecular geometries.
- Designing and Synthesizing New Molecules: In the field of drug discovery and materials science, understanding molecular geometry is paramount for designing molecules with specific desired properties.
Advanced Considerations: Beyond VSEPR
While VSEPR theory is a powerful tool for predicting electron and molecular geometries, it has limitations. For more complex molecules, especially those with multiple central atoms or significant electron delocalization, more sophisticated theoretical methods like density functional theory (DFT) are needed for accurate predictions. These methods provide a more quantitative and nuanced description of the electron distribution and molecular shape.
Conclusion
Electron geometry and molecular geometry are closely related yet distinct concepts that describe the three-dimensional arrangement of atoms and electron pairs within a molecule. While electron geometry considers all electron domains, including lone pairs, molecular geometry focuses solely on the arrangement of atoms. Understanding both concepts is critical for comprehending molecular properties, interpreting spectroscopic data, and designing new molecules with desired characteristics. The VSEPR theory serves as a powerful introductory tool, while more advanced methods are needed for complex systems. By grasping these distinctions, chemists gain a deeper understanding of the intricate world of molecular structure and its profound implications.
Latest Posts
Latest Posts
-
Why Is Ice Melting Not A Chemical Reaction
Apr 06, 2025
-
The Lcm Of 6 And 10
Apr 06, 2025
-
Type Of Energy Transformed By A Toaster Into Thermal Energy
Apr 06, 2025
-
Are All Angles Of A Parallelogram Congruent
Apr 06, 2025
-
What Is Not A Symptom Of Foodborne Illness
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Difference Between Molecular Geometry And Electron Geometry . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
