Conclusion Of Millikan Oil Drop Experiment
Juapaving
Apr 05, 2025 · 6 min read
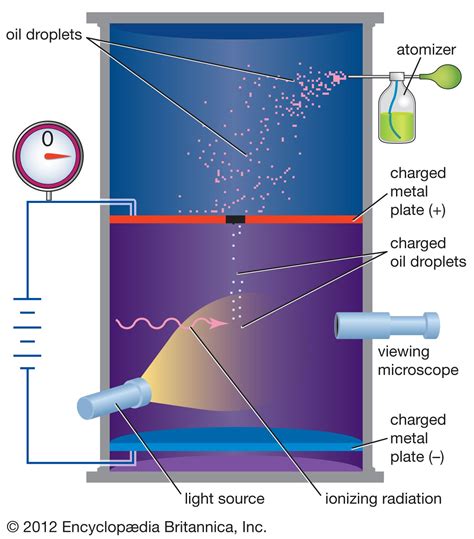
Table of Contents
The Millikan Oil Drop Experiment: Conclusions and Lasting Impact
Robert Millikan's oil drop experiment, conducted in the early 20th century, stands as a landmark achievement in physics. While the experiment itself is relatively straightforward in concept, its conclusions were revolutionary, profoundly impacting our understanding of the fundamental building blocks of matter. This article delves deep into the conclusions drawn from Millikan's experiment, exploring its significance and its lasting legacy on the field of physics and beyond.
Determining the Elementary Charge: The Core Conclusion
The primary and most significant conclusion of Millikan's oil drop experiment was the precise determination of the elementary charge, often denoted as e. This is the fundamental unit of electric charge, the smallest possible unit of electric charge that can exist independently. Before Millikan's work, the existence of a discrete, quantized charge was theorized, but there was no accurate measurement.
Millikan's ingenious method involved observing the motion of tiny oil droplets suspended in an electric field. By carefully balancing the gravitational force on the droplet with the electrostatic force, he could determine the charge on each droplet. Crucially, he observed that the charges measured were always integer multiples of a single, fundamental value: e. This observation provided irrefutable evidence for the quantization of electric charge, a cornerstone of modern physics. This wasn't just an approximation; Millikan's meticulous measurements led to a remarkably accurate value of e, laying the foundation for future advancements.
Refining the Value of e: A Legacy of Precision
The initial value of e obtained by Millikan was remarkably accurate for its time. However, subsequent refinements and improved experimental techniques have further refined the value. The current accepted value of the elementary charge, as determined through numerous experiments building upon Millikan's work, is incredibly precise. This continued refinement demonstrates the power of scientific methodology and the importance of building upon previous discoveries. The initial work laid the groundwork, paving the way for greater precision and deeper understanding.
Beyond the Elementary Charge: Further Implications
While the determination of e is the experiment's most celebrated conclusion, its impact extended far beyond this single measurement. The experiment had several other profound implications:
1. Confirmation of the Atomic Theory:
The successful measurement of the elementary charge provided strong experimental evidence supporting the atomic theory. The concept of atoms and their constituent parts had been debated for centuries, but Millikan's experiment gave concrete physical evidence for the discrete nature of matter and its fundamental units of charge, lending further credence to the atomic model.
2. Validation of the Electron's Existence:
The oil drop experiment validated the existence of the electron as a fundamental particle with a specific charge. While J.J. Thomson had previously discovered the electron through his cathode ray experiments, Millikan's work provided a precise measure of its charge, significantly strengthening the case for its existence as a distinct and fundamental entity. This contributed to a more robust understanding of atomic structure.
3. Advancements in Measurement Techniques:
Millikan's experiment also significantly advanced experimental techniques in physics. The methods he developed for precise observation and measurement of microscopic phenomena laid the foundation for many future experiments in atomic and subatomic physics. His approach, incorporating meticulous control over experimental parameters, became a model for careful scientific investigation.
4. Foundation for Further Discoveries:
The precise value of e became a crucial input in other fundamental calculations in physics. For example, it was instrumental in determining Avogadro's number (the number of atoms in one mole of a substance), which is fundamental to chemistry and related sciences. The accuracy of this calculation depended heavily on the accuracy of e. The value subsequently allowed physicists and chemists to accurately calculate the masses of atoms and molecules, bringing further precision to these fields.
Criticisms and Controversies: Addressing the Challenges
Despite its groundbreaking contributions, Millikan's oil drop experiment wasn't without its critics and controversies. One significant issue revolved around the selection of data. It was later argued that Millikan may have selectively chosen data points that fit his hypothesis, discarding data that seemed inconsistent. This raised questions about the integrity of the experimental methodology and the extent to which his results accurately reflected the phenomena.
However, it's important to note that while criticisms of data selection exist, the overall conclusion of quantized charge remains robust and has been validated extensively through independent experiments and theoretical frameworks. The criticisms highlight the importance of rigorous data analysis and transparency in scientific practice. These critiques, while important, don't diminish the landmark achievement of determining the elementary charge.
The Lasting Legacy: A Milestone in Physics
The Millikan oil drop experiment remains a pivotal experiment in the history of physics. Its conclusions dramatically altered our understanding of the fundamental nature of matter and electricity. The experiment provided compelling evidence for the quantization of charge, the existence of the electron as a fundamental particle, and the validity of the atomic theory.
Beyond its specific conclusions, the experiment serves as a powerful example of the scientific method in action. It demonstrates the importance of careful experimental design, meticulous data analysis, and the iterative nature of scientific progress. The experiment continues to be taught in physics classrooms worldwide, serving as a model of experimental ingenuity and a testament to the power of scientific inquiry.
Impact on Education and Science Communication:
The experiment's relatively simple setup and clear conclusions make it an excellent educational tool. Its accessibility allows for insightful discussions about experimental design, data analysis, and the interpretation of scientific results. This accessibility, combined with its historical significance, makes it a valuable component of physics education. The experiment’s clear visualization of fundamental concepts like quantization of charge and electrostatic forces is invaluable for students grasping these otherwise abstract ideas. Its impact is not confined to physics classrooms; it also serves as a prime example in discussions about the scientific process and the importance of critical analysis within the scientific community.
Continuing Relevance in Modern Physics:
Despite being conducted over a century ago, the fundamental principles underpinning the Millikan oil drop experiment continue to hold relevance in modern physics. The concept of quantized charge remains central to our understanding of the universe, underpinning the Standard Model of particle physics and guiding research in areas such as quantum electrodynamics and quantum field theory. While modern techniques offer far greater precision and sophistication, the experiment's core concept and methodology remain essential for understanding fundamental physics principles. Its legacy continues to inspire and challenge physicists as they delve into the subatomic world.
Conclusion: A Monumental Contribution
In conclusion, Robert Millikan's oil drop experiment was not just a clever experiment; it was a pivotal moment in the history of physics. Its most important conclusion – the precise measurement of the elementary charge – revolutionized our understanding of matter and electricity. The experiment’s impact extends far beyond this singular achievement, providing substantial evidence supporting the atomic theory, solidifying the existence of the electron, and driving significant advancements in experimental techniques. Although subject to historical critiques concerning data selection, the experiment's fundamental conclusions remain unchallenged, a testament to the enduring power of meticulous experimentation and the quest for understanding the fundamental building blocks of our universe. The legacy of the Millikan oil drop experiment endures as a monumental contribution to the field of physics and a shining example of scientific inquiry at its finest.
Latest Posts
Latest Posts
-
What Distinguishes An Element From A Compound
Apr 06, 2025
-
How Many Feet Is 27 Inches
Apr 06, 2025
-
What Are The Factors Of 117
Apr 06, 2025
-
The Energy Currency Used By Cells Is
Apr 06, 2025
-
How Tall Is 39 Inches In Feet
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Conclusion Of Millikan Oil Drop Experiment . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
