Chemical Bonds In Order From Strongest To Weakest
Juapaving
Apr 03, 2025 · 6 min read
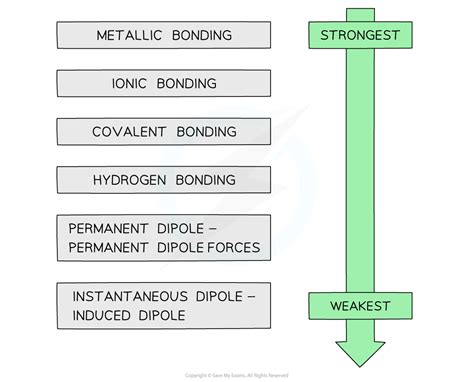
Table of Contents
- Chemical Bonds In Order From Strongest To Weakest
- Table of Contents
- Chemical Bonds: A Comprehensive Guide from Strongest to Weakest
- The Hierarchy of Chemical Bonds: Strength and Stability
- 1. Covalent Bonds (Strongest)
- 2. Ionic Bonds
- 3. Metallic Bonds
- 4. Hydrogen Bonds
- 5. Van der Waals Forces (Weakest)
- Conclusion: The Interplay of Bond Types
- Latest Posts
- Latest Posts
- Related Post
Chemical Bonds: A Comprehensive Guide from Strongest to Weakest
Understanding chemical bonds is fundamental to grasping the behavior of matter. From the formation of simple molecules to the complexity of biological systems, the strength and nature of these bonds dictate properties like melting point, boiling point, solubility, and reactivity. This comprehensive guide explores the various types of chemical bonds, arranged in order from strongest to weakest, delving into their characteristics, formation, and examples.
The Hierarchy of Chemical Bonds: Strength and Stability
Chemical bonds arise from the electrostatic attraction between atoms, resulting from the sharing or transfer of electrons. The strength of a bond is directly related to the energy required to break it – a stronger bond requires more energy to break. Here's a breakdown of bond types, arranged from strongest to weakest:
1. Covalent Bonds (Strongest)
Covalent bonds are formed when atoms share one or more pairs of electrons. This sharing creates a stable electron configuration for each atom, mimicking the electron arrangement of noble gases. The strength of a covalent bond depends on several factors, including:
- Bond order: The number of electron pairs shared between two atoms. A triple bond (e.g., in N₂) is stronger than a double bond (e.g., in O₂), which is stronger than a single bond (e.g., in H₂).
- Atom size: Smaller atoms form stronger covalent bonds because their nuclei are closer to the shared electrons, leading to greater electrostatic attraction.
- Electronegativity difference: While ideally suited for atoms with similar electronegativities, the presence of a larger electronegativity difference leads to polar covalent bonds. While still classified as covalent, this polarity can affect the overall bond strength and reactivity.
Types of Covalent Bonds:
- Nonpolar Covalent Bonds: These bonds occur when electrons are shared equally between atoms with similar electronegativities (e.g., H₂, Cl₂).
- Polar Covalent Bonds: These bonds occur when electrons are shared unequally between atoms with different electronegativities (e.g., H₂O, HCl). The more electronegative atom carries a partial negative charge (δ-), while the less electronegative atom carries a partial positive charge (δ+).
Examples of Covalent Bonding:
- Diamonds: A giant covalent structure with incredibly strong carbon-carbon bonds, resulting in extreme hardness and high melting point.
- Water (H₂O): Polar covalent bonds between oxygen and hydrogen atoms give water its unique properties.
- Methane (CH₄): Nonpolar covalent bonds between carbon and hydrogen atoms.
- Proteins and DNA: Complex molecules held together by numerous covalent bonds (peptide bonds in proteins, phosphodiester bonds in DNA), showcasing the crucial role of covalent bonds in biological systems.
2. Ionic Bonds
Ionic bonds form through the transfer of electrons from one atom to another. This transfer creates ions: positively charged cations (atoms that lose electrons) and negatively charged anions (atoms that gain electrons). The electrostatic attraction between these oppositely charged ions constitutes the ionic bond.
Factors Affecting Ionic Bond Strength:
- Charge of the ions: Higher charges lead to stronger attractions. For example, the bond between Mg²⁺ and O²⁻ is stronger than the bond between Na⁺ and Cl⁻.
- Size of the ions: Smaller ions lead to stronger attractions because the distance between the charges is less.
- Lattice energy: The energy released when ions come together to form a crystal lattice. Higher lattice energy indicates a stronger ionic bond.
Examples of Ionic Bonding:
- Sodium chloride (NaCl): Sodium atoms lose an electron to become Na⁺ ions, while chlorine atoms gain an electron to become Cl⁻ ions. The electrostatic attraction between these ions forms the ionic bond.
- Magnesium oxide (MgO): A stronger ionic bond than NaCl due to the higher charges of the ions.
- Calcium fluoride (CaF₂): An example of an ionic compound with a 1:2 ratio of cations to anions.
3. Metallic Bonds
Metallic bonds occur in metals, where valence electrons are delocalized and shared amongst a "sea" of electrons. These electrons are not associated with any particular atom but move freely throughout the metal lattice. This mobility is responsible for several characteristic properties of metals, including electrical conductivity and malleability.
Factors Affecting Metallic Bond Strength:
- Number of valence electrons: More valence electrons contribute to a stronger metallic bond.
- Charge density of the metal ions: Higher charge density results in a stronger attraction between the metal ions and the delocalized electrons.
- Atomic radius: Smaller atoms generally have stronger metallic bonds.
Examples of Metallic Bonding:
- Iron (Fe): The strong metallic bonding contributes to its high melting point and tensile strength.
- Copper (Cu): Excellent electrical conductivity due to the highly mobile delocalized electrons.
- Gold (Au): Malleable and ductile due to the ability of metal ions to slide past each other without disrupting the metallic bonding.
4. Hydrogen Bonds
Hydrogen bonds are a special type of dipole-dipole interaction. They occur between a hydrogen atom bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine) and another electronegative atom in a different molecule or part of the same molecule. While weaker than covalent, ionic, and metallic bonds, hydrogen bonds are crucial for many biological processes.
Factors Affecting Hydrogen Bond Strength:
- Electronegativity of the acceptor atom: Higher electronegativity results in a stronger hydrogen bond.
- Distance between the atoms: Shorter distances lead to stronger bonds.
- Orientation of the molecules: Optimal alignment of the interacting atoms maximizes bond strength.
Examples of Hydrogen Bonding:
- Water (H₂O): Hydrogen bonds between water molecules are responsible for its high boiling point, surface tension, and its role as a universal solvent.
- DNA: Hydrogen bonds between base pairs hold the double helix structure together.
- Proteins: Hydrogen bonds contribute to the secondary and tertiary structures of proteins.
5. Van der Waals Forces (Weakest)
Van der Waals forces are weak, short-range electrostatic attractions between molecules or parts of molecules. These forces are further categorized into:
- Dipole-dipole interactions: Occur between polar molecules with permanent dipoles. The positive end of one molecule is attracted to the negative end of another.
- London dispersion forces (LDFs): Occur between all molecules (polar or nonpolar) due to temporary, instantaneous dipoles caused by fluctuating electron distribution. Even noble gases exhibit LDFs.
- Ion-dipole interactions: These occur between an ion and a polar molecule, with the charge of the ion influencing the strength of the interaction.
Factors Affecting Van der Waals Forces:
- Molecular size and shape: Larger molecules with greater surface area have stronger LDFs. Molecular shape also plays a role, as more compact molecules have weaker LDFs.
- Polarity: Polar molecules exhibit stronger dipole-dipole interactions than nonpolar molecules.
- Distance: These forces are very short-range; they decrease rapidly with distance.
Examples of Van der Waals Forces:
- Noble gases: Liquid and solid noble gases are held together by LDFs.
- Nonpolar molecules: The forces holding together nonpolar molecules like hydrocarbons are primarily LDFs.
- Gecko feet: The ability of geckos to climb walls is attributed to the large number of weak Van der Waals forces between their feet and the surface.
Conclusion: The Interplay of Bond Types
The strength of a chemical bond is a crucial determinant of a substance's properties. While covalent bonds are generally the strongest, ionic, metallic, and other intermolecular forces such as hydrogen bonds and Van der Waals forces play equally significant roles in shaping the behavior of various materials. Understanding this hierarchy allows for a deeper understanding of the physical and chemical world around us, ranging from the strength of materials to the intricate interactions within biological systems. This knowledge serves as a foundation for numerous scientific disciplines, highlighting the critical importance of comprehending the nature and strength of chemical bonds.
Latest Posts
Latest Posts
-
A Convex Lens Of Focal Length 10cm
Apr 05, 2025
-
Which Expression Is Equal To 72
Apr 05, 2025
-
Substance That Turns Litmus Paper Red
Apr 05, 2025
-
What Are The Characteristics Of Metalloids
Apr 05, 2025
-
Electric Field At The End Of A Charged Wire
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Chemical Bonds In Order From Strongest To Weakest . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
