What Are The Characteristics Of Metalloids
Juapaving
Apr 05, 2025 · 6 min read
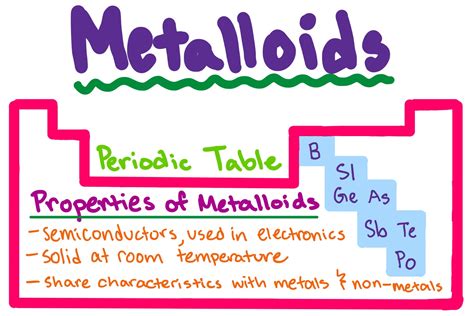
Table of Contents
What are the Characteristics of Metalloids? Exploring the Fuzzy Boundary Between Metals and Nonmetals
Metalloids, also known as semimetals, occupy a fascinating middle ground in the periodic table, exhibiting properties that blend characteristics of both metals and nonmetals. This ambiguity makes them incredibly useful in a wide range of applications, from electronics to medicine. Understanding their unique characteristics is key to appreciating their importance. This in-depth exploration will delve into the defining traits of metalloids, exploring their physical and chemical properties, their diverse applications, and the ongoing scientific research surrounding them.
Defining Metalloids: A Blurred Line
Unlike the clear-cut distinctions between metals and nonmetals, metalloids possess a blend of properties, making their classification somewhat subjective. There isn't a universally agreed-upon list, but elements generally considered metalloids include boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), polonium (Po), and astatine (At). The placement of some elements, like polonium and astatine, is often debated due to their radioactive nature and limited availability for extensive study.
The key to understanding metalloids lies in recognizing their intermediate characteristics. They don't neatly fall into either the metal or nonmetal category, instead showcasing a fascinating mix.
Physical Properties: A Unique Combination
Metalloids display a unique set of physical properties that distinguish them from their metallic and nonmetallic counterparts. Let's explore some key characteristics:
1. Appearance: A Diverse Palette
Metalloids exhibit a diverse range of appearances. Some, like silicon, have a grayish-black, metallic luster. Others, like arsenic, might appear more brittle and less lustrous. This visual variability further emphasizes their intermediary nature. The appearance can also vary significantly based on the crystal structure and purity of the sample.
2. Conductivity: Semiconductors Take Center Stage
One of the most crucial defining features of metalloids is their semiconductivity. Unlike metals, which are excellent conductors of electricity, and nonmetals, which are generally insulators, metalloids exhibit intermediate conductivity. This means their ability to conduct electricity can be altered by various factors, such as temperature, light exposure, and the presence of impurities. This unique characteristic is what makes them indispensable in the electronics industry.
3. Brittle Nature: A Fragile Middle Ground
Metalloids are generally brittle and lack the ductility and malleability commonly associated with metals. They tend to fracture or shatter when subjected to stress, contrasting with the ability of metals to deform under pressure without breaking. This brittle nature is a key difference from metallic elements.
4. Melting and Boiling Points: A Moderate Range
The melting and boiling points of metalloids fall within a moderate range, not as extreme as those of many metals or nonmetals. This intermediate range reflects their mixed characteristics, demonstrating neither the exceptionally high melting points of some metals nor the generally lower melting points of many nonmetals.
Chemical Properties: Reactivity and Beyond
The chemical behavior of metalloids is equally fascinating, exhibiting a range of reactivity depending on the specific element and the conditions of the reaction.
1. Variable Oxidation States: Adaptable Behavior
Metalloids can exhibit multiple oxidation states, meaning they can lose or gain varying numbers of electrons in chemical reactions. This adaptability contributes to their versatility in forming diverse compounds. This contrasts with some metals that tend to have a limited number of stable oxidation states.
2. Reactivity: A Balanced Act
Metalloids demonstrate a moderate reactivity, falling between the highly reactive metals and the relatively unreactive nonmetals. Their reactivity depends significantly on factors such as temperature and the presence of other reactants. This balanced reactivity allows them to participate in a wide range of chemical reactions without being overly vigorous or inert.
3. Semiconductor Behavior: The Heart of Electronics
Their semiconductor behavior is not solely a physical property but also significantly influences their chemical reactions. The ability to control their conductivity through doping (introducing impurities) allows for the creation of materials with precisely tailored electrical properties, crucial for semiconductor devices.
Applications: From Electronics to Medicine
The unique combination of physical and chemical properties makes metalloids incredibly versatile, leading to a wide range of applications across various industries.
1. Electronics: The Foundation of Modern Technology
Metalloids, especially silicon, are the cornerstone of modern electronics. Silicon is used extensively in transistors, integrated circuits, and microprocessors, forming the basis of computers, smartphones, and countless other electronic devices. Germanium also plays a crucial role, particularly in high-frequency applications and certain types of transistors.
2. Optoelectronics: Light and Electronics Combined
Metalloids are essential in optoelectronics, the field dealing with the interaction of light and electricity. They are used in various light-emitting diodes (LEDs), solar cells, and fiber optic cables, taking advantage of their ability to both conduct electricity and interact with light.
3. Medicine: Crucial Roles in Health Care
Certain metalloids find applications in medicine. Arsenic, though toxic in high doses, has found limited use in certain treatments, notably in some anti-cancer drugs. However, the use of arsenic requires strict control due to its high toxicity. Research continues to explore the potential medicinal applications of metalloids while carefully considering safety concerns.
4. Alloys and Materials Science: Enhancing Properties
Metalloids are incorporated into various alloys to enhance their properties. For example, the addition of metalloids can improve the hardness, strength, or corrosion resistance of certain metals. This is an area of ongoing research and development, aiming to create new materials with superior performance characteristics.
5. Catalysts: Accelerating Chemical Reactions
Some metalloids act as catalysts, substances that increase the rate of chemical reactions without being consumed in the process. Their catalytic activity is crucial in various industrial processes, particularly in the production of chemicals and plastics.
Ongoing Research and Future Developments
Research on metalloids is ongoing, driven by the desire to discover new applications and improve existing ones. Here are some key areas of current focus:
- Nanotechnology: The study of metalloid nanomaterials is uncovering unique properties and applications not observed in bulk materials. This field is promising for creating advanced electronic devices, sensors, and catalysts.
- Energy applications: Metalloids are being explored for their potential in renewable energy technologies, including solar cells and batteries. Improving the efficiency and cost-effectiveness of these technologies is a major research goal.
- Biomedical applications: The potential of metalloids in targeted drug delivery, bioimaging, and biosensors is being investigated. This area requires careful consideration of safety and biocompatibility.
- New materials discovery: The search for novel metalloid-based materials with superior properties continues, driven by the needs of various industries, including electronics, aerospace, and automotive.
Conclusion: A Multifaceted Group of Elements
Metalloids represent a fascinating and important group of elements. Their unique blend of metallic and nonmetallic properties makes them indispensable in a vast array of technologies and applications. From the microprocessors powering our computers to the solar cells generating renewable energy, metalloids play a crucial role in shaping our modern world. Continued research and innovation will undoubtedly lead to even more exciting discoveries and applications of these fascinating elements, further solidifying their importance in science and technology. The ambiguity of their classification only enhances their intrigue, highlighting the rich diversity of the chemical elements and the complexities of the periodic table. The study of metalloids is an ongoing journey of discovery, promising a future brimming with technological advancements built on their exceptional versatility.
Latest Posts
Latest Posts
-
Electron Configuration For A Neutral Atom Of Oxygen
Apr 05, 2025
-
What Is The Lcm Of 16 And 12
Apr 05, 2025
-
Which Of The Following Statements Are False
Apr 05, 2025
-
What Type Of Medium Travels The Slowest
Apr 05, 2025
-
Find The Least Common Multiple Of 5 And 3
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Are The Characteristics Of Metalloids . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
