At What Temperature Will Both Solid And Liquid Be Present
Juapaving
Apr 05, 2025 · 5 min read
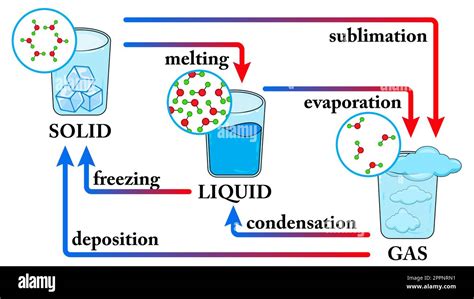
Table of Contents
At What Temperature Will Both Solid and Liquid Be Present? Understanding Phase Transitions and Equilibrium
The question of when both solid and liquid phases of a substance coexist hinges on understanding phase transitions and the concept of equilibrium. It's not simply a single temperature, but rather a range of conditions under which this coexistence occurs. This article delves into the intricacies of phase diagrams, melting and freezing points, and the factors influencing the solid-liquid equilibrium.
Phase Transitions: The Dance of Matter
Matter exists in various phases: solid, liquid, gas, and plasma. These phases are characterized by the arrangement and movement of their constituent particles (atoms, molecules, or ions). Phase transitions are the processes by which matter changes from one phase to another. These transitions are driven by changes in temperature and/or pressure.
Key phase transitions relevant to the solid-liquid coexistence are:
-
Melting (Fusion): The transition from a solid to a liquid. This occurs when sufficient thermal energy is supplied to overcome the intermolecular forces holding the solid together, allowing the particles to move more freely.
-
Freezing (Solidification): The transition from a liquid to a solid. This happens when thermal energy is removed, causing the particles to lose kinetic energy and become more ordered, forming a solid structure.
Factors Influencing Phase Transitions
The temperature at which melting and freezing occur isn't a fixed constant for all substances. Several factors play a critical role:
-
Intermolecular Forces: Stronger intermolecular forces (e.g., hydrogen bonds, dipole-dipole interactions) require more energy to overcome, resulting in higher melting and freezing points. Water, with its strong hydrogen bonds, has a relatively high melting point (0°C).
-
Molecular Weight: Larger molecules generally have higher melting and freezing points due to increased van der Waals forces between them.
-
Pressure: Pressure affects the melting and freezing points, although the effect is generally small for solids and liquids. For most substances, increased pressure raises the melting point, compressing the solid and making it more difficult to transition to the liquid phase. Water is an exception – increased pressure lowers its melting point (this is why ice skates work).
-
Impurities: The presence of impurities in a substance can alter its melting and freezing points. Impurities often depress the freezing point and elevate the melting point, resulting in a range of temperatures over which melting/freezing takes place instead of a single point.
The Solid-Liquid Equilibrium: A Balancing Act
The coexistence of solid and liquid phases is a state of dynamic equilibrium. At the melting/freezing point, the rate of melting (solid to liquid) is equal to the rate of freezing (liquid to solid). This means that the number of particles transitioning from solid to liquid is balanced by the number transitioning from liquid to solid. There's a continuous exchange, but the overall amounts of solid and liquid remain constant.
This equilibrium is best visualized using a phase diagram. A phase diagram is a graphical representation showing the conditions of temperature and pressure at which different phases of a substance exist in equilibrium. The line separating the solid and liquid regions represents the melting/freezing point at different pressures.
Interpreting Phase Diagrams
A typical phase diagram shows three regions: solid, liquid, and gas. The lines separating these regions represent the conditions where two phases coexist in equilibrium. The point where all three phases coexist is called the triple point. The critical point marks the end of the liquid-gas phase boundary.
Focusing on the solid-liquid boundary:
-
Along this line, both solid and liquid phases are present in equilibrium. The exact temperature along this line depends on the pressure.
-
To the left of the line, only the solid phase exists.
-
To the right of the line, only the liquid phase exists.
The Role of Purity and Pressure
As mentioned earlier, impurities significantly influence the solid-liquid equilibrium. A pure substance has a sharp melting/freezing point, meaning the transition occurs at a precise temperature. However, impurities broaden this transition, creating a range of temperatures where both phases coexist. This is because impurities disrupt the ordered structure of the solid, making it easier to melt at lower temperatures. The freezing point depression is directly proportional to the concentration of the impurity.
Pressure also plays a minor but measurable role. While the impact is generally small for solids and liquids, increasing pressure often raises the melting point (except for water). This is because the increased pressure compresses the solid, requiring more energy to break the intermolecular forces and transition to the liquid phase.
Practical Applications and Examples
Understanding the solid-liquid equilibrium is crucial in various applications:
-
Material Science: Determining the melting points of metals and alloys is vital in manufacturing processes like casting and welding. The ability to control the solid-liquid equilibrium is essential for creating materials with desired properties.
-
Chemistry: Phase diagrams are essential tools for understanding chemical reactions and separating mixtures through techniques like fractional crystallization.
-
Food Science: The freezing and melting points of water are critical in food preservation.
-
Meteorology: The solid-liquid equilibrium of water is fundamental to understanding weather patterns and precipitation.
Specific Examples
Let's consider some specific examples to illustrate the concepts:
-
Water: At standard pressure (1 atm), water's melting/freezing point is 0°C. At this temperature, ice and liquid water can coexist in equilibrium. If the temperature is slightly below 0°C, only ice will be present; if slightly above, only liquid water.
-
Ice Cream Making: Ice cream involves a careful control of the temperature to achieve the desired consistency. The presence of sugar and other ingredients lowers the freezing point of the water in the mixture, allowing it to remain partially liquid even at temperatures below 0°C. The freezing process occurs gradually, resulting in a smoother texture.
Conclusion: A Dynamic Equilibrium
The temperature at which both solid and liquid phases are present isn't a single point but rather a range of conditions defined by the substance's phase diagram and influenced by factors such as pressure and purity. Understanding the principles of phase transitions, equilibrium, and the effects of impurities is crucial for numerous scientific and technological applications. The dynamic interplay between melting and freezing, a state of constant molecular flux, underlines the fundamental nature of phase changes in matter. This knowledge empowers us to control and manipulate these transitions for various practical purposes, from crafting alloys to preserving food. The solid-liquid equilibrium, while seemingly simple at first glance, reveals a rich tapestry of physical chemistry at work.
Latest Posts
Latest Posts
-
7 Cm Is How Many Inches
Apr 06, 2025
-
How Many Neutrons Are In Mg
Apr 06, 2025
-
Do Anions Have A Positive Charge
Apr 06, 2025
-
How Many Milliliters Is 3 Liters
Apr 06, 2025
-
Find Area And Perimeter Of A Triangle
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about At What Temperature Will Both Solid And Liquid Be Present . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
