According To Bronsted Lowry Theory An Acid Is
Juapaving
Apr 02, 2025 · 7 min read
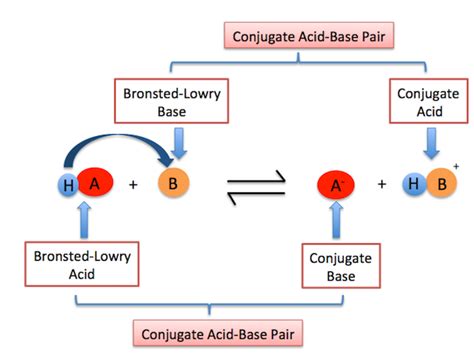
Table of Contents
According to Brønsted-Lowry Theory, an Acid Is... A Proton Donor!
The Brønsted-Lowry theory, a cornerstone of chemistry, offers a comprehensive understanding of acids and bases, going beyond the limitations of previous definitions. Unlike the Arrhenius theory, which restricts acids to substances that produce hydrogen ions (H⁺) in aqueous solutions, the Brønsted-Lowry theory expands the definition to encompass a wider range of chemical reactions. This theory is crucial for understanding various chemical processes, from everyday reactions to complex biological systems. Let's delve deeper into the heart of this theory: what constitutes an acid according to Brønsted-Lowry?
The Brønsted-Lowry Definition: A Proton's Perspective
At its core, the Brønsted-Lowry theory defines an acid as a substance that donates a proton (H⁺). This proton donation is a key characteristic that distinguishes Brønsted-Lowry acids from other chemical species. It's important to note that the proton donation doesn't always occur in an aqueous solution; it can happen in various solvents or even in the gas phase. This broadened scope significantly increases the applicability of the theory.
This proton donation process often involves a chemical reaction where the acid interacts with a base, a substance that accepts the proton. This interaction leads to the formation of a conjugate base (the species remaining after the acid donates a proton) and a conjugate acid (the species formed when the base accepts a proton). This crucial interplay between acids and bases forms the foundation of the Brønsted-Lowry theory.
Understanding Conjugate Acid-Base Pairs
The concept of conjugate acid-base pairs is central to understanding Brønsted-Lowry acid-base reactions. A conjugate acid-base pair is simply an acid and its corresponding base, differing by only one proton (H⁺). For example, consider the reaction between hydrochloric acid (HCl) and water (H₂O):
HCl + H₂O ⇌ H₃O⁺ + Cl⁻
In this reaction:
- HCl is the acid, donating a proton.
- H₂O is the base, accepting a proton.
- Cl⁻ is the conjugate base of HCl.
- H₃O⁺ (hydronium ion) is the conjugate acid of H₂O.
Notice how HCl and Cl⁻ differ by a single proton, as do H₂O and H₃O⁺. This illustrates the fundamental relationship within a conjugate acid-base pair. Understanding these pairs is essential for predicting the outcome of acid-base reactions and analyzing their equilibrium.
Examples of Brønsted-Lowry Acids
The versatility of the Brønsted-Lowry definition extends the classification of acids far beyond the traditional Arrhenius definition. Here are some examples demonstrating the diverse range of substances categorized as Brønsted-Lowry acids:
1. Strong Acids: Complete Proton Donation
Strong acids are characterized by their complete dissociation in aqueous solutions. They readily donate their protons, resulting in a high concentration of H₃O⁺ ions. Examples include:
- Hydrochloric acid (HCl): A common laboratory acid, readily dissociating into H⁺ and Cl⁻ ions.
- Sulfuric acid (H₂SO₄): A strong diprotic acid, meaning it can donate two protons.
- Nitric acid (HNO₃): A highly corrosive acid used in various industrial processes.
- Perchloric acid (HClO₄): One of the strongest known acids.
2. Weak Acids: Partial Proton Donation
Weak acids, unlike strong acids, only partially dissociate in aqueous solutions. This means they donate only a small fraction of their protons, resulting in a lower concentration of H₃O⁺ ions. The equilibrium between the undissociated acid and its conjugate base is significant in weak acid chemistry. Examples include:
- Acetic acid (CH₃COOH): Found in vinegar, it is a common weak acid.
- Formic acid (HCOOH): The simplest carboxylic acid, found in ant venom.
- Carbonic acid (H₂CO₃): Formed when carbon dioxide dissolves in water, crucial in blood buffering systems.
- Hydrofluoric acid (HF): A weak acid, despite the high electronegativity of fluorine.
3. Amphoteric Substances: Acids and Bases in One
Some substances can act as both Brønsted-Lowry acids and bases, depending on the reaction context. These are known as amphoteric substances. A classic example is water (H₂O):
- As an acid: In the reaction with a stronger base like ammonia (NH₃), water donates a proton: H₂O + NH₃ ⇌ NH₄⁺ + OH⁻
- As a base: In the reaction with a stronger acid like HCl, water accepts a proton: HCl + H₂O ⇌ H₃O⁺ + Cl⁻
This dual behavior exemplifies the versatility and adaptability of the Brønsted-Lowry theory in classifying chemical species.
4. Non-Aqueous Acid-Base Reactions
The Brønsted-Lowry theory transcends the limitations of aqueous solutions. Acid-base reactions can occur in various non-aqueous solvents, expanding the range of applicable reactions. For example, liquid ammonia (NH₃) can act as a solvent for acid-base reactions, with NH₄⁺ acting as the acidic species.
Beyond the Basics: Applications of the Brønsted-Lowry Theory
The Brønsted-Lowry theory's impact extends far beyond simple acid-base definitions. Its applications permeate various fields, including:
1. Understanding Biological Systems
The theory is fundamental to understanding biological systems. Many biochemical reactions involve proton transfer, crucial for maintaining pH balance, enzyme activity, and overall cellular function. For example, the buffering systems in blood utilize the Brønsted-Lowry concept to regulate blood pH within a narrow physiological range.
2. Industrial Processes
Many industrial processes rely on acid-base reactions governed by the Brønsted-Lowry theory. Acid-catalyzed reactions are widespread in chemical synthesis, production of pharmaceuticals, and various manufacturing processes. The precise control of pH is often crucial for the success of these processes.
3. Environmental Chemistry
The theory is critical in environmental chemistry for understanding acid rain, water quality, and soil chemistry. Acid deposition, resulting from the release of acidic gases like sulfur dioxide and nitrogen oxides, directly impacts ecosystems and water bodies, causing significant environmental consequences. Understanding the acid-base reactions involved is crucial for mitigating these effects.
4. Analytical Chemistry
Titration, a crucial analytical technique, is based on the principles of acid-base reactions governed by the Brønsted-Lowry theory. Titration allows for precise determination of the concentration of an unknown acid or base solution, having widespread applications in various analytical procedures.
Comparing Brønsted-Lowry to Arrhenius Theory
While both theories describe acids and bases, the Brønsted-Lowry theory offers a significant expansion:
- Arrhenius Theory: Defines acids as substances that produce H⁺ ions in water, and bases as substances that produce OH⁻ ions in water. This theory is limited to aqueous solutions.
- Brønsted-Lowry Theory: Defines acids as proton donors and bases as proton acceptors. This broader definition encompasses a wider range of reactions, including those occurring in non-aqueous solvents and those not involving OH⁻ ions.
The Brønsted-Lowry theory effectively incorporates the Arrhenius theory as a special case within its broader framework. It provides a more comprehensive and versatile understanding of acid-base chemistry.
Limitations of the Brønsted-Lowry Theory
Despite its widespread applicability, the Brønsted-Lowry theory has some limitations:
- It does not explain acid-base reactions where protons are not involved. For instance, reactions involving Lewis acids and bases, where electron pairs are donated and accepted, are not explained by this theory.
- The theory focuses primarily on proton transfer reactions, neglecting other aspects of acid-base interactions.
The Lewis theory of acids and bases addresses some of these limitations by expanding the definition beyond proton transfer to include electron pair donation and acceptance. While the Brønsted-Lowry theory remains a powerful tool, it's important to acknowledge its limitations and appreciate the broader context provided by other acid-base theories.
Conclusion: A Powerful Framework for Understanding Acid-Base Chemistry
The Brønsted-Lowry theory provides a powerful and versatile framework for understanding acid-base chemistry. Its definition of an acid as a proton donor, coupled with the concept of conjugate acid-base pairs, allows for a comprehensive analysis of a wide range of reactions. From biological processes to industrial applications, the theory’s impact is undeniable. While it has limitations, the Brønsted-Lowry theory remains a cornerstone of chemistry education and research, providing a fundamental understanding of the behavior of acids and bases in diverse chemical environments. Understanding this theory is essential for anyone seeking a deeper grasp of chemical reactions and their significance in the world around us.
Latest Posts
Latest Posts
-
How Many Hearts Does Worm Have
Apr 03, 2025
-
5 2 Simplified As A Mixed Number
Apr 03, 2025
-
Is 87 A Composite Or Prime Number
Apr 03, 2025
-
Does A Eukaryotic Cell Have Ribosomes
Apr 03, 2025
-
Equation For Charging And Discharging Of Capacitor
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about According To Bronsted Lowry Theory An Acid Is . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
