Write The Electron Configuration For A Neutral Atom Of Oxygen
Juapaving
Mar 30, 2025 · 6 min read
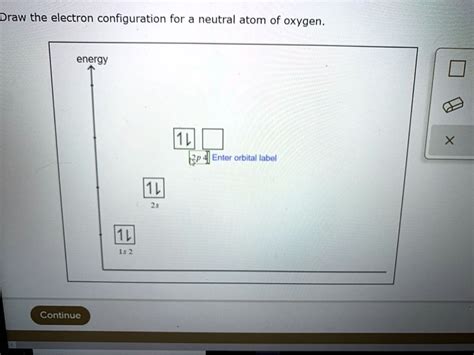
Table of Contents
- Write The Electron Configuration For A Neutral Atom Of Oxygen
- Table of Contents
- Unveiling the Secrets of Oxygen: A Deep Dive into its Electron Configuration
- Understanding Electron Configuration: The Foundation
- The Quantum Mechanical Model: Shells, Subshells, and Orbitals
- Determining the Electron Configuration of Oxygen (O)
- Orbital Diagram Representation
- Implications of Oxygen's Electron Configuration: Reactivity and Bonding
- Oxygen's Role in Life and the Environment
- Beyond the Basics: Excited States and Ions
- Excited States:
- Ions:
- Conclusion: The Significance of Understanding Electron Configuration
- Latest Posts
- Latest Posts
- Related Post
Unveiling the Secrets of Oxygen: A Deep Dive into its Electron Configuration
Oxygen, the life-giving element that fuels our respiration and underpins countless biological processes, holds a fascinating story within its atomic structure. Understanding its electron configuration is key to unlocking its unique properties and reactivity. This comprehensive guide will not only provide the electron configuration for a neutral oxygen atom but will also delve into the underlying principles, explore its implications for oxygen's chemical behavior, and examine related concepts in atomic structure.
Understanding Electron Configuration: The Foundation
Before we dive into oxygen's specific electron configuration, let's establish a solid understanding of the fundamental principles. Electron configuration describes the arrangement of electrons within an atom's energy levels and sublevels. These arrangements follow specific rules dictated by quantum mechanics, ensuring stability and influencing the atom's interactions with other atoms.
The Quantum Mechanical Model: Shells, Subshells, and Orbitals
The modern understanding of atomic structure relies on the quantum mechanical model, which postulates that electrons occupy specific energy levels, often visualized as shells surrounding the nucleus. Each shell is further divided into subshells (s, p, d, and f), each characterized by a different shape and energy level. Within each subshell are orbitals, which are regions of space where there's a high probability of finding an electron.
- Principal Quantum Number (n): This number represents the energy level or shell (n = 1, 2, 3...). Higher 'n' values indicate higher energy levels and greater distance from the nucleus.
- Azimuthal Quantum Number (l): This number defines the subshell (l = 0 for s, 1 for p, 2 for d, 3 for f).
- Magnetic Quantum Number (ml): This specifies the orbital within a subshell (-l ≤ ml ≤ +l).
- Spin Quantum Number (ms): This describes the intrinsic angular momentum of an electron (+1/2 or -1/2, often represented as ↑ and ↓).
The Pauli Exclusion Principle states that no two electrons within an atom can have the same set of four quantum numbers. This means each orbital can hold a maximum of two electrons with opposite spins. The Aufbau Principle dictates that electrons fill the lowest energy levels first, and Hund's Rule states that electrons will individually occupy each orbital within a subshell before pairing up.
Determining the Electron Configuration of Oxygen (O)
Oxygen, with an atomic number of 8, possesses 8 protons and, in a neutral atom, 8 electrons. Following the Aufbau principle and the rules of quantum mechanics, let's determine its electron configuration:
-
First shell (n=1): This shell contains only the s subshell, which can hold a maximum of two electrons. Therefore, oxygen's first shell is completely filled: 1s².
-
Second shell (n=2): This shell contains both the s and p subshells. The s subshell holds two electrons, filling it completely: 2s². The p subshell can hold up to six electrons (three orbitals, each with two electrons). Oxygen has four remaining electrons, so these fill the 2p subshell: 2p⁴.
Therefore, the complete electron configuration of a neutral oxygen atom is: 1s²2s²2p⁴.
Orbital Diagram Representation
A more detailed visualization uses orbital diagrams, showing individual orbitals and electron spins:
1s: ↑↓ 2s: ↑↓ 2p: ↑ ↑ ↑ (one electron in each 2p orbital before pairing)
This diagram clearly shows that the 2p subshell is not fully filled. This partially filled subshell is crucial in explaining oxygen's reactivity.
Implications of Oxygen's Electron Configuration: Reactivity and Bonding
The electron configuration directly influences oxygen's chemical behavior. The four electrons in the 2p subshell are valence electrons, meaning they participate in chemical bonding. Oxygen readily forms bonds to achieve a stable octet (eight electrons in its outermost shell), following the octet rule. This can be achieved in a few ways:
-
Covalent Bonding: Oxygen readily forms covalent bonds by sharing electrons with other atoms. This is evidenced in the formation of the oxygen molecule (O₂), where two oxygen atoms share two pairs of electrons to achieve a stable octet. This double bond explains the relatively high bond strength and stability of the O₂ molecule.
-
Ionic Bonding: Although less common than covalent bonding, oxygen can also form ionic bonds, particularly with highly electropositive metals. In these cases, oxygen gains two electrons to attain a stable octet, acquiring a 2- charge (oxide ion, O²⁻). This is exemplified in the formation of metal oxides like magnesium oxide (MgO).
Oxygen's Role in Life and the Environment
Oxygen's unique electron configuration and its consequent reactivity are fundamental to life on Earth. Its role is multifaceted:
-
Respiration: Oxygen acts as the final electron acceptor in cellular respiration, a process that generates the energy needed for life. This process relies on the reactivity of oxygen and its ability to form stable bonds.
-
Combustion: Oxygen is a powerful oxidizing agent, fueling combustion reactions that release significant amounts of energy. This is crucial for many industrial processes and, unfortunately, also contributes to environmental challenges like wildfires and air pollution.
-
Ozone Layer: Ozone (O₃), an allotrope of oxygen, plays a vital role in protecting life from harmful ultraviolet radiation from the sun. The formation and stability of ozone are directly related to the electronic structure of oxygen.
Beyond the Basics: Excited States and Ions
The electron configuration we've discussed refers to the ground state of a neutral oxygen atom. However, oxygen can also exist in excited states or as ions.
Excited States:
When an oxygen atom absorbs energy, an electron can jump to a higher energy level. This results in an excited state, which is less stable and more reactive. The excited state electron configuration will show at least one electron promoted to a higher energy level. These excited states play a role in various processes, such as light absorption and emission.
Ions:
Oxygen readily forms ions, most notably the oxide ion (O²⁻), which has gained two electrons. The electron configuration of the oxide ion (O²⁻) is 1s²2s²2p⁶, achieving a stable octet.
Conclusion: The Significance of Understanding Electron Configuration
The electron configuration of oxygen, 1s²2s²2p⁴, is far more than just a set of numbers and letters. It's the blueprint that dictates oxygen's properties, its reactivity, and its vital role in countless natural processes. By understanding the fundamental principles behind electron configuration and its implications, we gain a deeper appreciation for the fascinating world of atomic structure and its influence on the macroscopic world around us. This understanding forms a bedrock for exploring the chemistry of countless compounds and the complexities of biological systems. The information presented here provides a solid foundation for further explorations into advanced chemistry concepts, including bonding theories, molecular orbital theory, and spectroscopy. Further research into these areas will solidify your understanding of atomic structure and its powerful influence on the properties and interactions of matter.
Latest Posts
Latest Posts
-
Are Minerals A Renewable Resource Why Or Why Not
Apr 03, 2025
-
What Organelles Are Found Only In Plant Cells
Apr 03, 2025
-
Laplace Transform Of Piecewise Function Calculator
Apr 03, 2025
-
What Are The Factors For 10
Apr 03, 2025
-
Balaneced Chemical Reaction Between C4h10 Oz Yeliding Co2
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Write The Electron Configuration For A Neutral Atom Of Oxygen . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
