Which Of The Following Is Strongest Acid
Juapaving
Apr 03, 2025 · 5 min read
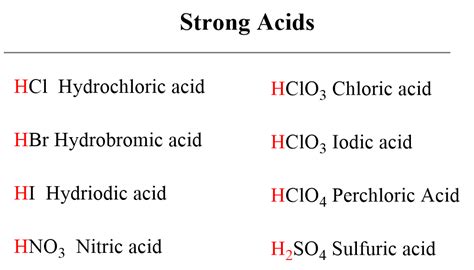
Table of Contents
Which of the Following is the Strongest Acid? Understanding Acid Strength
Determining the strongest acid among a group requires a clear understanding of what constitutes acid strength and the factors influencing it. This article delves deep into the concept of acid strength, exploring various theories and factors that dictate how readily an acid donates a proton (H⁺). We'll examine specific examples and provide you with the tools to confidently compare and contrast the strengths of different acids.
What is Acid Strength?
Acid strength is a measure of how readily an acid donates a proton (H⁺) in a solution. Strong acids completely dissociate in water, meaning all their molecules donate a proton, resulting in a high concentration of H⁺ ions. Weak acids, on the other hand, only partially dissociate, resulting in a much lower concentration of H⁺ ions. This dissociation is an equilibrium reaction, represented by the following equation for a generic acid HA:
HA(aq) ⇌ H⁺(aq) + A⁻(aq)
The equilibrium constant for this reaction, denoted as K<sub>a</sub>, is the acid dissociation constant. A higher K<sub>a</sub> value indicates a stronger acid. The pK<sub>a</sub> value, which is the negative logarithm of K<sub>a</sub>, is also commonly used. A lower pK<sub>a</sub> value indicates a stronger acid.
Factors Affecting Acid Strength
Several key factors influence the strength of an acid:
1. Bond Strength:
The strength of the bond between the hydrogen atom and the rest of the molecule significantly impacts acid strength. A weaker H-X bond leads to easier proton donation and thus a stronger acid. For example, HI is a stronger acid than HF because the H-I bond is weaker than the H-F bond.
2. Electronegativity:
The electronegativity of the atom bonded to the hydrogen atom plays a crucial role. A more electronegative atom pulls electron density away from the H-X bond, weakening it and making proton donation easier. This is why acids with more electronegative atoms are generally stronger. Consider the series of hydrohalic acids (HF, HCl, HBr, HI). Fluorine is the most electronegative, yet HI is the strongest acid. This highlights the interplay between electronegativity and bond strength; while fluorine's electronegativity strengthens the bond, the overall size of the halogen dominates. The much larger size of the iodine atom results in a weaker bond, making HI the strongest.
3. Resonance Stabilization:
Resonance stabilization of the conjugate base (A⁻) significantly affects acid strength. If the conjugate base is stabilized by resonance, the equilibrium shifts to the right, favoring the dissociation of the proton and resulting in a stronger acid. Carboxylic acids, for instance, exhibit resonance stabilization of their conjugate base (carboxylate ion), making them relatively strong acids compared to other organic acids.
4. Inductive Effects:
Electron-withdrawing groups (like halogens) near the acidic proton can stabilize the conjugate base through inductive effects, increasing acid strength. Conversely, electron-donating groups decrease acid strength. The presence of electron-withdrawing groups pulls electron density away from the acidic hydrogen, making it easier to lose a proton.
5. Solvent Effects:
The solvent in which the acid is dissolved also affects its strength. Water, being a polar solvent, stabilizes both the acid and its conjugate base, but the extent of stabilization varies depending on the acid's structure. In non-polar solvents, acid strength can be significantly different from that observed in aqueous solutions.
Comparing Acid Strengths: Examples
Let's consider a few examples to illustrate the concepts discussed above. Suppose we need to compare the acid strengths of HCl, CH₃COOH (acetic acid), and H₂SO₄ (sulfuric acid).
-
HCl (Hydrochloric Acid): This is a strong acid, meaning it completely dissociates in water. It has a very low pK<sub>a</sub> value (approximately -7). The high electronegativity of chlorine and the relatively weak H-Cl bond contribute to its strength.
-
CH₃COOH (Acetic Acid): This is a weak acid, only partially dissociating in water. It has a pK<sub>a</sub> value of around 4.76. The presence of the methyl group (CH₃) is an electron-donating group that destabilizes the conjugate base (acetate ion), making it a weaker acid than HCl.
-
H₂SO₄ (Sulfuric Acid): This is a strong diprotic acid, meaning it can donate two protons. Its first dissociation is essentially complete in water, making it a strong acid. However, its second dissociation is weaker. Its high electronegativity of the sulfur atom and resonance stabilization of the bisulfate ion contribute to its strength.
Based on these examples, the order of acid strength would be: H₂SO₄ > HCl > CH₃COOH. However, the precise ranking can change depending on the solvent and concentration.
Advanced Concepts: Beyond Simple Comparisons
Comparing the strength of more complex acids necessitates a deeper understanding of advanced concepts:
-
Lewis Acids and Bases: The Brønsted-Lowry definition of acids (proton donors) is not exhaustive. Lewis acids are electron-pair acceptors, encompassing a wider range of compounds. The strength of Lewis acids depends on their ability to accept electron pairs, which relates to factors like size, charge, and electronic structure.
-
Hammett Acidity Function (H₀): This function is used to measure the acidity of extremely strong acids in highly concentrated solutions, where the standard pK<sub>a</sub> scale becomes inadequate. H₀ values account for the deviations from ideal behavior in these concentrated acidic media.
-
Superacids: These are acids stronger than 100% sulfuric acid. They are often mixtures of strong acids, and their strength results from complex interactions between the component acids. Examples include magic acid (fluorosulfuric acid and antimony pentafluoride) and fluoroantimonic acid.
Conclusion: A Multifaceted Understanding
Determining the strongest acid among a group isn't a simple task. It requires a thorough understanding of the factors influencing acid strength: bond strength, electronegativity, resonance, inductive effects, and solvent effects. While comparing simple acids relies heavily on pK<sub>a</sub> values and general guidelines, more complex situations might demand consideration of Lewis acidity, Hammett acidity functions, and the concept of superacids. This article provides a comprehensive foundation for accurately comparing acid strengths and further exploring the fascinating world of acid-base chemistry. Remember that context matters; the relative strengths of acids can vary based on the specific conditions, making a nuanced understanding crucial.
Latest Posts
Latest Posts
-
What Is The Hardest Substance In The Body
Apr 03, 2025
-
Do Frying Pans Need A Low Specific Heat Capacity
Apr 03, 2025
-
Electron Configuration For A Neutral Atom Of Manganese
Apr 03, 2025
-
Least Common Multiple Of 7 And 14
Apr 03, 2025
-
A Simple Pendulum With Bob Of Mass M
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is Strongest Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
