What Is The Last Element In Period 4
Juapaving
Apr 02, 2025 · 5 min read
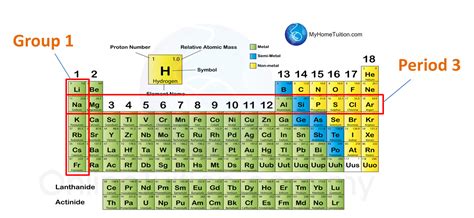
Table of Contents
What is the Last Element in Period 4? Unraveling the Mysteries of Krypton
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding its structure is crucial for grasping the fundamental principles of chemistry. One common question that arises, particularly for students beginning their chemistry journey, is: What is the last element in Period 4? The answer is Krypton, and this article delves deep into its properties, characteristics, and significance within the broader context of the periodic table.
Understanding the Periodic Table: Periods and Groups
Before we dive into the specifics of Krypton, let's briefly review the organization of the periodic table. Elements are arranged into periods (rows) and groups (columns). Periods represent the principal energy level or shell occupied by the outermost electrons of an element's atoms. As we move across a period from left to right, the number of protons and electrons increases, leading to variations in chemical and physical properties.
Groups, on the other hand, represent elements with similar outer electron configurations, resulting in similar chemical behavior. For example, elements in Group 18 (noble gases) are all chemically inert due to their full valence electron shells.
Period 4: A Detailed Look
Period 4 encompasses elements with electrons filling the fourth principal energy level (n=4). This period is particularly interesting because it showcases the transition from the s and p blocks to the d block, introducing a new set of properties and complexities. The elements in Period 4 range from Potassium (K) with its highly reactive nature to Krypton (Kr) with its inertness. The elements in this period demonstrate a wide array of physical and chemical properties, reflecting the intricate interplay of electron configurations and nuclear forces.
The Transition Metals of Period 4
The inclusion of transition metals significantly impacts the diversity of properties seen in Period 4. These elements, from Scandium (Sc) to Zinc (Zn), have partially filled d orbitals, leading to a variety of oxidation states and complex ion formation. This explains their use in various catalytic processes and their vibrant coloration in compounds. Their variable oxidation states contribute to their complex and fascinating chemistry.
The Impact of Electron Configuration
The electron configuration of elements profoundly influences their chemical behavior. As we move across Period 4, the number of electrons increases, affecting the element's electronegativity, ionization energy, and atomic radius. These variations lead to observable differences in reactivity and bonding characteristics. Elements on the left-hand side tend to be highly reactive metals readily losing electrons, while elements on the right-hand side show a tendency to gain electrons or remain inert.
Krypton: The Noble Gas at the End of Period 4
Finally, we arrive at Krypton (Kr), the last element in Period 4. Krypton is a noble gas, meaning it exists as a monatomic gas under standard conditions. Its electron configuration, [Ar] 3d¹⁰ 4s² 4p⁶, indicates a full valence shell, making it exceptionally unreactive. This full outer shell is the key to understanding its inert nature.
Key Properties of Krypton:
- Atomic Number: 36
- Atomic Symbol: Kr
- Atomic Weight: 83.798 u
- Electron Configuration: [Ar] 3d¹⁰ 4s² 4p⁶
- Melting Point: -157.37 °C
- Boiling Point: -153.4 °C
- State at Room Temperature: Gas
- Group: 18 (Noble Gases)
- Period: 4
Chemical Inertness and Applications:
Krypton's chemical inertness stems from its complete valence electron shell. This lack of reactivity limits its direct chemical applications. However, it's worth noting that under extreme conditions, like high pressure and high energy, Krypton can form compounds with highly electronegative elements like fluorine. These are typically unstable and rare.
Despite its inertness, Krypton finds applications in various fields:
-
Lighting: Krypton is used in some types of fluorescent lights and high-intensity discharge lamps to produce a brighter, whiter light than traditional neon lights. The unique spectral lines emitted by Krypton contribute to the distinctive color properties.
-
Lasers: Krypton-based lasers, particularly krypton fluoride lasers, are used in various medical and industrial applications, including laser surgery and micromachining. The energy emitted by Krypton lasers makes them suitable for precision applications.
-
Photography: Historically, Krypton was used in high-speed flash photography, although this has largely been replaced by other technologies.
-
Scientific Research: Krypton isotopes find applications in radioactive dating techniques and in various scientific research areas.
Isotopes of Krypton:
Krypton has several naturally occurring isotopes, with varying abundances. Some of these isotopes are stable, while others are radioactive with varying half-lives. These isotopes are studied extensively in nuclear physics and related fields.
Comparing Krypton to Other Elements in Period 4:
Understanding Krypton's position within Period 4 requires comparing it to its neighboring elements. Unlike the highly reactive alkali metals (like Potassium) at the beginning of Period 4, Krypton shows little reactivity. This contrast highlights the dramatic changes in chemical properties as we move across a period. Compared to the transition metals in the middle of Period 4, Krypton lacks the ability to form a wide range of oxidation states and complex ions. Its inertness stands in stark contrast to the rich chemistry of its transition metal neighbors.
Conclusion: Krypton's Significance in Chemistry
Krypton, the last element in Period 4, is a fascinating example of a noble gas. Its unreactive nature, stemming from its complete valence electron shell, distinguishes it from other elements in the period. While its chemical applications are limited due to its inertness, it finds important applications in lighting, laser technology, and scientific research. Understanding Krypton's properties and its placement within the periodic table provides valuable insights into the fundamental principles of chemical bonding, reactivity, and the organization of matter. The study of Krypton serves as a powerful example of how the periodic table provides a comprehensive framework for understanding the behavior and properties of all known elements. The unique properties of Krypton demonstrate the remarkable diversity found even within a single period of the periodic table. By understanding its properties and its place in the broader context of the periodic table, we deepen our understanding of the fundamental principles governing the behavior of matter.
Latest Posts
Latest Posts
-
Compare Protein Synthesis In Prokaryotes And Eukaryotes
Apr 03, 2025
-
A Closed Figure Made Of Line Segments
Apr 03, 2025
-
During Cell Division What Role Do Centrosomes Play
Apr 03, 2025
-
How Many Millimeters Are In 7 Cm
Apr 03, 2025
-
Good Words To Describe Your Mother
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Last Element In Period 4 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
