What Is The Electron Configuration For Calcium
Juapaving
Apr 02, 2025 · 6 min read
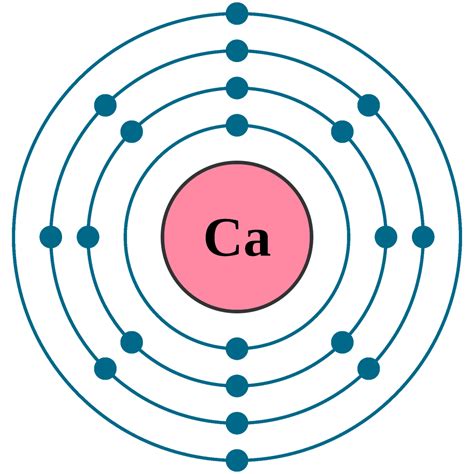
Table of Contents
What is the Electron Configuration for Calcium? A Deep Dive into Atomic Structure
Calcium, a vital element for life, plays a crucial role in various biological processes. Understanding its electronic structure is key to grasping its chemical behavior and biological significance. This comprehensive guide delves into the electron configuration of calcium, explaining the underlying principles and its implications.
Understanding Electron Configuration
Before diving into calcium's specific configuration, let's establish a foundational understanding of what electron configuration represents. Electron configuration describes the arrangement of electrons in the different energy levels and sublevels within an atom. This arrangement dictates how an atom will interact with other atoms, forming chemical bonds and exhibiting specific properties. It's governed by fundamental principles like the Aufbau principle, the Pauli exclusion principle, and Hund's rule.
The Aufbau Principle: Filling Orbitals in Order of Increasing Energy
The Aufbau principle, meaning "building-up" in German, states that electrons fill atomic orbitals in order of increasing energy. This means electrons first occupy the lowest energy levels available before moving to higher energy levels. The order of filling is generally: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, and so on.
The Pauli Exclusion Principle: A Maximum of Two Electrons per Orbital
The Pauli exclusion principle dictates that each orbital can hold a maximum of two electrons, and these two electrons must have opposite spins. This is crucial for understanding the arrangement of electrons within subshells.
Hund's Rule: Maximizing Unpaired Electrons
Hund's rule states that electrons will individually occupy each orbital within a subshell before doubling up in any one orbital. This minimizes electron-electron repulsion and leads to a more stable configuration.
Determining the Electron Configuration of Calcium (Ca)
Calcium (Ca) has an atomic number of 20, meaning it has 20 protons and, in its neutral state, 20 electrons. To determine its electron configuration, we follow the Aufbau principle, filling orbitals in order of increasing energy:
-
1s²: The first energy level (n=1) contains only the s subshell, which can hold up to two electrons. These two electrons fill the 1s orbital.
-
2s²: The second energy level (n=2) starts with the s subshell, again holding two electrons.
-
2p⁶: The second energy level also contains the p subshell, which has three orbitals, each capable of holding two electrons, for a total of six electrons.
-
3s²: The third energy level (n=3) begins with the s subshell, holding another two electrons.
-
3p⁶: The p subshell in the third energy level accommodates another six electrons.
-
4s²: Finally, we reach the fourth energy level (n=4), starting with the s subshell, which takes the last two electrons.
Therefore, the complete electron configuration for calcium is: 1s²2s²2p⁶3s²3p⁶4s².
Simplified Notation and Valence Electrons
While the full electron configuration is informative, a shorthand notation, using the noble gas configuration, is often used for brevity and clarity. Calcium's configuration can be simplified by noting that the electron configuration of Argon (Ar), a noble gas with atomic number 18, is 1s²2s²2p⁶3s²3p⁶. We can represent calcium's configuration as: [Ar]4s².
The valence electrons are the electrons in the outermost shell, which are most involved in chemical bonding. In calcium's case, these are the two electrons in the 4s orbital. This explains calcium's tendency to lose these two electrons to achieve a stable, noble gas configuration (like Argon), resulting in a +2 ion (Ca²⁺).
The Significance of Calcium's Electron Configuration
Calcium's electron configuration is directly linked to its chemical and biological properties. The two valence electrons readily participate in ionic bonding, forming stable compounds with electronegative elements like oxygen, chlorine, and sulfur. This is crucial for the many roles calcium plays in biological systems.
Calcium's Role in Biological Systems
The relatively low ionization energy required to remove calcium's valence electrons makes it readily available to participate in a wide range of biological processes. These include:
-
Bone and Teeth Formation: Calcium phosphate forms the structural basis of bones and teeth, providing crucial strength and support.
-
Muscle Contraction: Calcium ions are essential for muscle contraction, regulating the interaction between actin and myosin filaments.
-
Nerve Impulse Transmission: Calcium ions play a role in the transmission of nerve impulses, influencing the release of neurotransmitters.
-
Blood Clotting: Calcium ions are cofactors in the coagulation cascade, crucial for blood clot formation and preventing excessive bleeding.
-
Enzyme Activation: Calcium ions act as cofactors for many enzymes, influencing their activity and regulatory functions.
-
Cellular Signaling: Calcium ions are secondary messengers in various cellular signaling pathways, regulating various cellular responses.
Implications of Electron Configuration in Calcium's Biological Functions
The ease with which calcium loses its two valence electrons is fundamental to its biological activity. The resulting Ca²⁺ ion readily interacts with various negatively charged molecules and proteins, facilitating its involvement in the diverse biological processes mentioned above. The specific interactions depend on the shape and charge distribution of the molecules it binds to, all dictated by its fundamental electron configuration.
Beyond the Basics: Excited States and Ionization Energies
While the ground state electron configuration describes the most stable arrangement of electrons, calcium can also exist in excited states. In an excited state, one or more electrons occupy higher energy levels than predicted by the Aufbau principle. This occurs when an atom absorbs energy (e.g., from light). The electron can transition back to the ground state, emitting energy in the process. The energy difference between these states is quantized and determines the wavelengths of light absorbed or emitted.
Understanding calcium's ionization energies provides additional insight into its behavior. The first ionization energy is the energy required to remove the first electron, the second ionization energy is required to remove the second, and so on. For calcium, the first and second ionization energies are relatively low, reflecting the ease with which the two 4s electrons are removed, consistent with its tendency to form a +2 ion. The subsequent ionization energies increase dramatically, reflecting the increasing difficulty in removing electrons from inner shells.
Conclusion: A Foundation for Understanding Calcium's Significance
The electron configuration of calcium, [Ar]4s², is not just a theoretical concept; it's the foundation for understanding its chemical reactivity and crucial biological roles. The two valence electrons readily participate in ionic bonding, leading to the formation of essential compounds and facilitating interactions with numerous biological molecules. This comprehensive understanding underscores the intimate relationship between an element's fundamental atomic structure and its macroscopic properties and biological significance, making calcium a prime example of how electron configuration dictates function. Further research into the intricacies of calcium's electron configuration and interactions can lead to advances in various fields, from material science to medicine.
Latest Posts
Latest Posts
-
Laplace Transform Of Piecewise Function Calculator
Apr 03, 2025
-
What Are The Factors For 10
Apr 03, 2025
-
Balaneced Chemical Reaction Between C4h10 Oz Yeliding Co2
Apr 03, 2025
-
Which Is The Longest Phase In Meiosis
Apr 03, 2025
-
How Many Commas In A Billion
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Electron Configuration For Calcium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
