What Is Meant By The Simplest Formula Of A Compound
Juapaving
Apr 02, 2025 · 6 min read
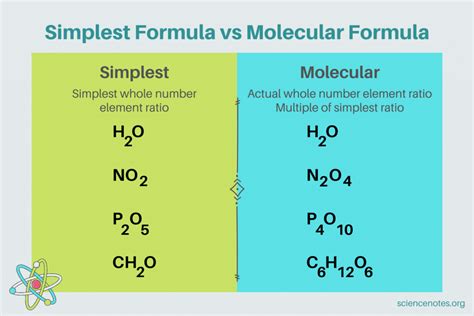
Table of Contents
What is Meant by the Simplest Formula of a Compound?
The simplest formula of a compound, also known as the empirical formula, represents the simplest whole-number ratio of atoms of each element present in a compound. It doesn't necessarily reflect the actual number of atoms in a molecule, but rather the ratio between them. Understanding the difference between the empirical formula and the molecular formula is crucial in chemistry. This article will delve deep into the concept of the simplest formula, exploring its significance, how to determine it, and its applications in various chemical contexts.
Understanding the Difference: Empirical vs. Molecular Formula
Before we dive into the intricacies of determining the simplest formula, let's clearly differentiate it from the molecular formula.
-
Molecular Formula: This formula precisely indicates the actual number of atoms of each element present in a single molecule of a compound. For example, the molecular formula of glucose is C₆H₁₂O₆, showing that one molecule of glucose contains 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms.
-
Empirical Formula: This formula provides the simplest whole-number ratio of atoms in a compound. For glucose (C₆H₁₂O₆), the empirical formula is CH₂O, representing the simplest ratio of 1 carbon atom, 2 hydrogen atoms, and 1 oxygen atom. Notice that the ratio remains consistent: 1:2:1, just expressed in the smallest whole numbers.
Many compounds share the same empirical formula but have different molecular formulas. This is because the molecular formula is a multiple of the empirical formula. For example, both ethene (C₂H₄) and cyclopropane (C₃H₆) have the same empirical formula, CH₂, but different molecular formulas reflecting their distinct structures and molecular weights.
Determining the Simplest Formula: A Step-by-Step Guide
Determining the simplest formula typically involves experimental data, often obtained through techniques like combustion analysis. This process involves several key steps:
1. Obtain the Mass (or Percentage) of Each Element: This is the crucial starting point. Combustion analysis, for instance, can precisely measure the amounts of carbon dioxide and water produced when a compound is burned completely. From this data, the mass of carbon and hydrogen in the original compound can be calculated. Other methods, such as gravimetric analysis, can determine the mass of other elements present. If given percentages, convert them to grams assuming a 100g sample.
2. Convert Mass to Moles: Using the molar mass (atomic weight) of each element, convert the mass of each element into the number of moles. This step is critical as it allows us to compare the amounts of each element in terms of their relative numbers of atoms. The formula for moles is:
Moles = Mass (g) / Molar Mass (g/mol)
3. Determine the Mole Ratio: Divide the number of moles of each element by the smallest number of moles obtained in the previous step. This step reveals the simplest whole-number ratio of atoms in the compound.
4. Express as a Formula: The whole-number ratios calculated in Step 3 become the subscripts in the empirical formula. If the ratios are not whole numbers, you may need to multiply all the ratios by a small integer to obtain whole numbers.
Example: Determining the Empirical Formula of a Compound
Let's walk through an example to solidify our understanding. Suppose a compound is analyzed and found to contain 40.0% carbon, 6.7% hydrogen, and 53.3% oxygen by mass. Let's find its empirical formula:
1. Convert Percentages to Grams: Assuming a 100g sample, we have:
- Carbon: 40.0 g
- Hydrogen: 6.7 g
- Oxygen: 53.3 g
2. Convert Grams to Moles: Using molar masses (C = 12.01 g/mol, H = 1.01 g/mol, O = 16.00 g/mol):
- Moles of C: 40.0 g / 12.01 g/mol = 3.33 mol
- Moles of H: 6.7 g / 1.01 g/mol = 6.63 mol
- Moles of O: 53.3 g / 16.00 g/mol = 3.33 mol
3. Determine the Mole Ratio: Divide by the smallest number of moles (3.33 mol):
- C: 3.33 mol / 3.33 mol = 1.00
- H: 6.63 mol / 3.33 mol = 1.99 ≈ 2.00
- O: 3.33 mol / 3.33 mol = 1.00
4. Express as a Formula: The simplest whole-number ratio is 1:2:1. Therefore, the empirical formula is CH₂O.
Moving Beyond the Empirical Formula: Determining the Molecular Formula
The empirical formula provides a starting point, but to determine the actual molecular formula, we need additional information – the molar mass of the compound. This is because the molecular formula is a whole-number multiple of the empirical formula.
The relationship is expressed as:
Molecular Formula = n x Empirical Formula
where 'n' is a whole number (1, 2, 3, etc.).
To find 'n', we divide the experimentally determined molar mass of the compound by the molar mass of the empirical formula. Let's assume the molar mass of our compound (CH₂O) is experimentally determined to be 180.16 g/mol.
1. Calculate the Empirical Formula Mass:
- Molar mass of CH₂O: (12.01 g/mol) + 2(1.01 g/mol) + (16.00 g/mol) = 30.03 g/mol
2. Determine the Value of 'n':
- n = (Molar mass of compound) / (Molar mass of empirical formula) = 180.16 g/mol / 30.03 g/mol ≈ 6
3. Determine the Molecular Formula:
- Molecular Formula = 6 x CH₂O = C₆H₁₂O₆
Therefore, the molecular formula of the compound is C₆H₁₂O₆, which is glucose.
Applications of the Simplest Formula
The determination of the simplest formula has widespread applications across various fields:
-
Chemical Analysis: It's a fundamental tool in identifying unknown compounds. By analyzing the composition of a substance, chemists can determine its empirical formula and potentially its molecular formula, aiding in its identification.
-
Determining Chemical Formulas from Experimental Data: As demonstrated earlier, the empirical formula is derived directly from experimental data obtained through various analytical methods.
-
Stoichiometric Calculations: In stoichiometry (the quantitative relationships between reactants and products in chemical reactions), the empirical formula is used to calculate the amounts of reactants needed or products produced in a reaction.
-
Polymer Chemistry: In polymer chemistry, the empirical formula helps determine the repeating unit in a polymer chain. This repeating unit forms the basis for understanding the polymer's properties and behavior.
-
Forensic Science: Forensic scientists use the simplest formula in analyzing substances found at crime scenes, helping identify unknown materials and trace evidence.
Conclusion
The simplest formula, or empirical formula, is a fundamental concept in chemistry, providing a simplified representation of the elemental composition of a compound. Its determination, involving several crucial steps, provides essential information for identifying unknown compounds, performing stoichiometric calculations, and understanding various chemical processes. While it doesn't depict the precise molecular structure like the molecular formula, the empirical formula acts as a crucial stepping stone in many chemical analyses and forms the basis for understanding the chemical composition of various substances. Understanding this crucial distinction between empirical and molecular formulas is vital for anyone working in the field of chemistry or related scientific disciplines.
Latest Posts
Latest Posts
-
Kg M 3 To G Cm3
Apr 03, 2025
-
120 Cm Is How Many Inches
Apr 03, 2025
-
The Scattering Of Light By A Colloid Is Called The
Apr 03, 2025
-
Roots Forming A Number In Logo
Apr 03, 2025
-
Alloy Of Mercury With Another Metal
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is Meant By The Simplest Formula Of A Compound . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
