What Colour Does Litmus Paper Turn In Acid
Juapaving
Apr 04, 2025 · 6 min read
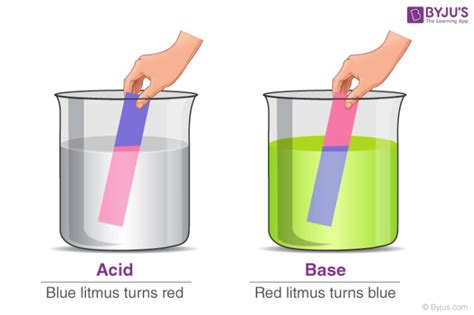
Table of Contents
What Color Does Litmus Paper Turn in Acid? Understanding pH Indicators
Litmus paper, a simple yet powerful tool, plays a crucial role in chemistry and various other scientific disciplines. Its primary function is to act as a pH indicator, revealing whether a solution is acidic or alkaline (basic). But the question remains: what color does litmus paper turn in acid? This comprehensive article will delve deep into the fascinating world of litmus paper, exploring its composition, mechanism of action, applications, and, most importantly, answering the central question in detail. We'll also touch upon the broader concept of pH and its importance.
Understanding pH and the Acid-Base Spectrum
Before diving into the specifics of litmus paper, it's crucial to grasp the concept of pH. The pH scale is a logarithmic scale that measures the acidity or alkalinity of a solution. It ranges from 0 to 14, with 7 being neutral. A pH value below 7 indicates acidity, while a pH value above 7 indicates alkalinity. The lower the pH value, the stronger the acid; the higher the pH value, the stronger the base.
Key pH values and their significance:
- pH 0-3: Strongly acidic (e.g., battery acid, stomach acid)
- pH 4-6: Weakly acidic (e.g., vinegar, orange juice)
- pH 7: Neutral (e.g., pure water)
- pH 8-10: Weakly alkaline (e.g., baking soda solution)
- pH 11-14: Strongly alkaline (e.g., drain cleaner, lye)
Understanding this spectrum is fundamental to interpreting the color changes observed with litmus paper.
What is Litmus Paper?
Litmus paper is a type of indicator paper made from a mixture of different dyes extracted from lichens. These dyes, primarily derived from species of Roccella, Lecanora, and Dendrographa lichens, exhibit a remarkable color change depending on the pH of the solution they are exposed to. The specific combination of dyes used determines the sensitivity and accuracy of the litmus paper's response to different pH levels.
The composition of litmus paper is not completely standardized, with variations existing between different manufacturers. However, the core principle remains consistent: the presence of multiple dyes enables a clear distinction between acidic and alkaline solutions.
Two Types of Litmus Paper: Red and Blue
Litmus paper comes in two forms: red litmus paper and blue litmus paper. These two types differ in their responses to acidic and alkaline solutions, enabling a more precise determination of the solution's pH.
-
Red litmus paper: This type of paper turns blue in the presence of an alkaline (basic) solution. It remains red in acidic solutions.
-
Blue litmus paper: This type of paper turns red in the presence of an acidic solution. It remains blue in alkaline solutions.
What Color Does Litmus Paper Turn in Acid? The Definitive Answer
Now, we arrive at the core question: what color does litmus paper turn in acid?
The answer is nuanced, depending on which type of litmus paper you're using:
-
Blue litmus paper turns red in the presence of an acid. This is the classic indicator of acidity. The intensity of the red color can sometimes provide a rough indication of the strength of the acid, with stronger acids producing a more vivid red.
-
Red litmus paper remains red in the presence of an acid. It will only change color in the presence of a base.
Therefore, observing a color change from blue to red using blue litmus paper is the definitive confirmation of an acidic solution. The lack of color change in red litmus paper when exposed to an acidic solution further supports this conclusion.
The Mechanism Behind the Color Change
The color change observed in litmus paper is due to a chemical reaction involving the dyes extracted from lichens. These dyes act as weak acids and bases, changing their molecular structure and thus their color in response to changes in the surrounding pH.
When blue litmus paper is exposed to an acid, the hydrogen ions (H⁺) from the acid interact with the dye molecules. This interaction leads to a change in the electron distribution within the dye molecules, resulting in a shift in the wavelength of light absorbed, and consequently, the color change to red.
Applications of Litmus Paper
Litmus paper's simplicity and effectiveness make it a valuable tool in various fields:
- Chemistry: Basic pH testing in laboratories and educational settings.
- Environmental monitoring: Assessing the acidity or alkalinity of soil, water samples, and other environmental materials.
- Medicine: Basic pH testing in clinical settings, though more precise methods are generally used for medical diagnostics.
- Food science: Testing the pH of food products during manufacturing and quality control.
- Home testing kits: Simple pH tests for various applications, such as pool water or soil testing.
While not as precise as electronic pH meters, litmus paper provides a quick, affordable, and convenient method for determining the approximate pH of a solution.
Limitations of Litmus Paper
Despite its usefulness, litmus paper has certain limitations:
- Qualitative rather than quantitative: Litmus paper only provides a general indication of whether a solution is acidic or alkaline. It doesn't give precise pH values. For precise measurements, a pH meter is necessary.
- Not suitable for all solutions: Some solutions may interfere with the litmus paper's color change, leading to inaccurate results.
- Sensitivity: Litmus paper is not as sensitive as some other pH indicators, meaning it might not detect weak acids or bases effectively.
Choosing and Using Litmus Paper Effectively
When selecting litmus paper, it's crucial to choose a reputable brand to ensure accuracy and reliability. Look for brands that specify the dye content and testing parameters. Always follow the manufacturer's instructions for proper handling and usage.
For accurate results:
- Use a clean, dry strip of litmus paper: Avoid contamination that could skew the results.
- Dip the paper briefly into the solution: Prolonged immersion can lead to inaccurate readings.
- Compare the color change to a standard color chart: This will help you correctly interpret the results.
- Ensure proper disposal: Follow local guidelines for disposing of used litmus paper.
Conclusion
In conclusion, blue litmus paper turns red in acid, while red litmus paper remains unchanged. This simple color change is a fundamental principle in chemistry and has numerous practical applications. While not a precise tool for determining pH, litmus paper remains a valuable, accessible, and cost-effective method for quickly assessing the acidity or alkalinity of a solution. Understanding its limitations and using it correctly are essential for accurate results. By understanding the broader concepts of pH and the mechanism behind the color change, we can fully appreciate the power and significance of this humble indicator. Further research into other indicators and advanced pH measurement techniques can provide even greater insight into the complexities of acid-base chemistry.
Latest Posts
Latest Posts
-
Coordination Number Of Body Centered Cubic
Apr 04, 2025
-
What Percent Is 3 Of 8
Apr 04, 2025
-
26 Rounded To The Nearest Hundredth
Apr 04, 2025
-
How Long Does It Take For The Sun To Rotate
Apr 04, 2025
-
Which Molecule Is Expected To Have The Smallest Pka
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Colour Does Litmus Paper Turn In Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
