Coordination Number Of Body Centered Cubic
Juapaving
Apr 04, 2025 · 5 min read
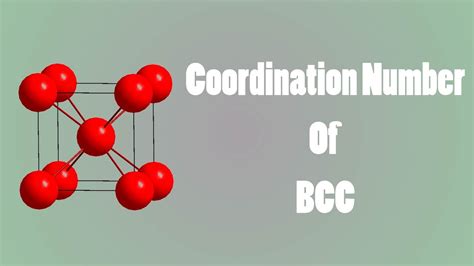
Table of Contents
Coordination Number of Body-Centered Cubic (BCC) Structures: A Deep Dive
The coordination number in crystallography represents the number of nearest neighbors surrounding a central atom in a crystal lattice. Understanding coordination numbers is crucial for predicting material properties like density, hardness, and conductivity. This article delves deep into the coordination number of body-centered cubic (BCC) structures, explaining its determination, implications, and practical applications.
What is a Body-Centered Cubic (BCC) Structure?
A body-centered cubic (BCC) structure is a common type of cubic crystal structure. It's characterized by atoms located at the eight corners of a cube, and one additional atom positioned precisely in the center of the cube. This central atom is equidistant from all eight corner atoms. Many metals, including iron (at room temperature), chromium, tungsten, and vanadium, exhibit a BCC structure. This structure is significantly different from the face-centered cubic (FCC) structure, which has additional atoms at the center of each face of the cube.
Determining the Coordination Number in BCC
To determine the coordination number of a BCC structure, we must visualize the arrangement of atoms and identify the nearest neighbors of a central atom. Let's consider a single atom located at the center of the cube. This central atom is directly surrounded by eight corner atoms. These eight corner atoms are the nearest neighbors to the central atom.
Visualizing Nearest Neighbors: Imagine lines connecting the central atom to each of the eight corner atoms. These lines represent the shortest distances between the central atom and its surrounding atoms. No other atoms are closer to the central atom than these eight.
Therefore, the coordination number for a body-centered cubic structure is 8. Each atom in a BCC structure is directly coordinated to eight other atoms.
Implications of the Coordination Number (8) in BCC Structures
The coordination number of 8 in BCC structures has significant implications for various material properties:
-
Atomic Packing Factor (APF): The APF represents the fraction of volume in a unit cell that is occupied by atoms. In a BCC structure, the APF is approximately 68%, meaning that a considerable amount of space is unoccupied within the unit cell. This relatively lower packing efficiency compared to FCC (74%) influences several material properties.
-
Density: Due to the lower APF, BCC structures generally have a lower density compared to FCC structures of the same element.
-
Mechanical Properties: The specific arrangement of atoms in the BCC structure affects its mechanical properties. The coordination number and packing efficiency influence the material's strength, ductility, and hardness. BCC metals often exhibit higher strength and lower ductility compared to FCC metals. The directional nature of bonding in BCC crystals contributes to their anisotropic properties (different properties in different directions).
-
Thermal Properties: The thermal expansion and heat capacity of a material are also affected by the crystal structure and coordination number. The relatively loose packing in BCC structures affects how effectively heat is conducted.
-
Electrical Conductivity: The electrical conductivity of BCC metals is usually less than that of FCC metals due to the difference in electron band structure that's influenced by the atomic packing.
-
Magnetic Properties: Several BCC metals, like iron, exhibit ferromagnetic properties. The coordination number influences the exchange interactions between atoms and therefore the overall magnetic behavior.
Comparison with Other Crystal Structures
It's essential to compare the BCC coordination number with those of other common crystal structures to understand the differences in their properties:
-
Face-Centered Cubic (FCC): FCC structures have a coordination number of 12. Each atom is surrounded by 12 nearest neighbors. This higher coordination number leads to a higher atomic packing factor (74%) and consequently, higher density and different mechanical properties compared to BCC.
-
Simple Cubic (SC): SC structures have a coordination number of only 6. This is the lowest coordination number among common crystal structures, resulting in a much lower atomic packing factor (52%). Simple cubic structures are rare in metals due to their low packing efficiency.
-
Hexagonal Close-Packed (HCP): HCP structures also exhibit a coordination number of 12, similar to FCC. However, the arrangement of atoms differs significantly, leading to different physical and mechanical properties.
Advanced Concepts and Applications
The coordination number of 8 in BCC structures influences various advanced concepts and applications:
-
Alloying and Phase Transformations: The BCC structure is not static; it can undergo phase transformations upon changes in temperature or pressure. Understanding the coordination number helps predict phase transitions and design alloys with desired properties. Many alloys involve BCC phases or transformations between BCC and other structures.
-
Solid Solutions: The ability of elements to form solid solutions within a BCC lattice is partly determined by the atomic size and the coordination environment. Substitutional solid solutions and interstitial solid solutions in BCC structures exhibit different properties based on the substituted or interstitial atoms.
-
Defects in BCC Crystals: Point defects (vacancies, interstitial atoms) and line defects (dislocations) within the BCC lattice significantly impact its mechanical properties. The coordination environment of atoms near these defects influences the overall behavior of the material.
-
Nanomaterials and Nanoparticles: The coordination number and atomic arrangement in nanoparticles can differ from the bulk material. Nanoparticles with a BCC structure may exhibit unique properties due to the high surface-to-volume ratio and altered coordination environments near the surface.
-
Computational Materials Science: Computational methods are used extensively to model and predict properties of BCC materials. The coordination number is a key parameter in simulations that aim to accurately reproduce the behavior of these materials. Software packages employ sophisticated algorithms to model atomic interactions within BCC lattices.
Conclusion
The coordination number of 8 in a body-centered cubic structure is a fundamental aspect influencing a wide range of material properties. Understanding this coordination number, along with its implications for atomic packing, density, mechanical behavior, thermal properties, electrical conductivity, and magnetic properties, is crucial for materials science and engineering applications. This article has provided a thorough overview of the concept, its ramifications, and its relevance in various contexts, from basic crystallography to advanced computational materials science. The knowledge gained about BCC structures enables material scientists and engineers to design and develop materials with specific and tailored properties for different applications. Further exploration into the intricate relationships between crystal structure, coordination number, and material properties continues to be a significant area of research.
Latest Posts
Latest Posts
-
1 1 4 As A Percent
Apr 05, 2025
-
Where In The Cell Does Krebs Cycle Occur
Apr 05, 2025
-
A Man Standing On The Roof Of A House
Apr 05, 2025
-
What Is Called A Group Of Birds
Apr 05, 2025
-
Difference Between Pnp And Npn Transistor
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Coordination Number Of Body Centered Cubic . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
