What Are The Horizontal Rows Called On The Periodic Table
Juapaving
Apr 05, 2025 · 6 min read
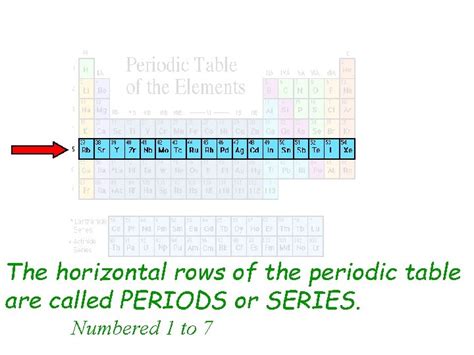
Table of Contents
What are the Horizontal Rows Called on the Periodic Table?
The periodic table, that iconic chart of chemical elements, is organized in a specific way to reflect the properties and behaviors of each element. Understanding its structure is key to understanding chemistry itself. One of the fundamental organizational principles of the periodic table lies in its horizontal and vertical arrangements. This article delves deep into the horizontal rows, exploring what they're called, how they're organized, and the significance of their arrangement in understanding the periodic trends of elements.
Understanding the Periodic Table's Structure
Before we dive into the specifics of the horizontal rows, let's briefly review the overall structure of the periodic table. It's a tabular arrangement of chemical elements organized by their atomic number (number of protons), electron configuration, and recurring chemical properties. The table is structured into:
- Periods (Horizontal Rows): These are the horizontal rows and are the focus of this article. Elements within a period share the same highest principal energy level (or electron shell).
- Groups or Families (Vertical Columns): These are the vertical columns, and elements within a group share similar chemical properties due to having the same number of valence electrons (electrons in the outermost shell).
What are the Horizontal Rows Called? Periods!
The answer is simple: the horizontal rows on the periodic table are called periods. Each period represents a principal energy level, signifying the electron shells that are progressively filled as you move across the row. The number of the period corresponds directly to the highest principal quantum number (n) of the electron orbitals being filled in that period.
Significance of Period Number
The period number provides valuable information about an element. For example:
- Period 1: Contains only hydrogen (H) and helium (He), which have electrons only in the first principal energy level (n=1). This level can accommodate a maximum of two electrons.
- Period 2: Elements in this period (lithium (Li) to neon (Ne)) fill the second principal energy level (n=2), which includes the s and p orbitals.
- Period 3: Similar to Period 2, but with the s and p orbitals of the third principal energy level (n=3) being filled.
- Period 4 and beyond: These periods become increasingly complex, introducing d orbitals (transition metals) and f orbitals (lanthanides and actinides).
Periodic Trends and Their Relationship to Periods
The arrangement of elements in periods allows us to observe and understand crucial periodic trends. These are predictable patterns in the properties of elements as you move across a period or down a group. Some key periodic trends that are significantly affected by period number include:
1. Atomic Radius
Atomic radius generally decreases across a period. As you move from left to right, the number of protons increases, leading to a stronger positive charge in the nucleus. This stronger pull attracts the electrons closer to the nucleus, resulting in a smaller atomic radius.
2. Ionization Energy
Ionization energy, the energy required to remove an electron from a neutral atom, generally increases across a period. The increased nuclear charge, as discussed above, holds the electrons more tightly, making it more difficult to remove an electron.
3. Electron Affinity
Electron affinity, the energy change when an electron is added to a neutral atom, generally increases across a period (with some exceptions). The increasing nuclear charge enhances the attraction for an additional electron.
4. Electronegativity
Electronegativity, the ability of an atom to attract electrons in a chemical bond, generally increases across a period. Similar to ionization energy and electron affinity, the stronger nuclear charge pulls electrons more strongly towards itself.
5. Metallic Character
Metallic character generally decreases across a period. Elements on the left side of a period are typically metals, characterized by their ability to lose electrons easily and form positive ions. As you move to the right, the non-metallic character increases, with elements becoming more likely to gain electrons and form negative ions.
These trends are clearly observable when examining elements within a particular period, showcasing the predictive power of the periodic table's organization.
The Unique Characteristics of Each Period
While the general periodic trends are important, each period also exhibits unique characteristics based on the electron configuration of its elements:
Period 1: The Simplest Period
This shortest period only includes hydrogen and helium. Hydrogen, a reactive non-metal, is unique in its ability to form both positive and negative ions. Helium, a noble gas, is exceptionally unreactive due to its filled electron shell.
Period 2: The Foundation of Organic Chemistry
Period 2 elements play a vital role in organic chemistry. Carbon's unique ability to form four covalent bonds is the basis of organic molecules. Other elements in this period, such as oxygen and nitrogen, are crucial components of biological molecules.
Period 3: Expanding the Possibilities
Period 3 introduces elements that expand on the properties of period 2, but with larger atomic sizes and slightly different chemical behavior. Phosphorus, sulfur, and chlorine are examples of elements with greater reactivity and diverse applications.
Periods 4-7: Increasing Complexity
The longer periods beyond period 3 introduce transition metals, elements with partially filled d orbitals, which give rise to various oxidation states and complex coordination compounds. Periods 6 and 7 further include the lanthanides and actinides, respectively, characterized by the filling of the f orbitals, leading to a substantial increase in the number of elements within these periods. The differences in the physical and chemical properties across these longer periods are vast.
The Importance of Understanding Periods
Understanding the concept of periods and their significance in the periodic table is fundamental to comprehending the behaviour of elements and predicting their chemical reactions. The periodic trends observed across periods provide a framework for understanding the properties of elements and their interactions. This knowledge is not just relevant in academic settings; it also plays a crucial role in various fields, including:
- Materials Science: Designing new materials with specific properties often involves selecting elements from different periods based on their desired attributes.
- Chemical Engineering: Understanding the reactivity of elements within a period is essential for optimizing chemical processes and designing efficient chemical reactions.
- Biochemistry: The elements within periods, especially periods 2 and 3, form the foundation of biological molecules and are crucial for life processes.
Conclusion: Periods – The Horizontal Backbone of Chemistry
The horizontal rows, or periods, on the periodic table are far more than just a convenient way of arranging elements. They represent fundamental principles of atomic structure and directly influence the chemical and physical properties of elements. By understanding the concept of periods, their relationship to electron configurations, and the periodic trends associated with them, we gain a deeper appreciation of the intricate organization and predictable behavior of the elements within the periodic table. The periods are, indeed, the horizontal backbone of our understanding of chemistry, crucial for both theoretical advancement and practical applications in various scientific and technological fields.
Latest Posts
Latest Posts
-
What Is The Scientific Name For A Cat
Apr 06, 2025
-
How Many Valence Electrons In Iodine
Apr 06, 2025
-
How Many Feet In 3 Yard
Apr 06, 2025
-
Find An Eigenvector Corresponding To The Eigenvalue
Apr 06, 2025
-
Which Of The Following Is Correct
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Are The Horizontal Rows Called On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
