Electron Configuration For A Neutral Atom Of Oxygen
Juapaving
Apr 05, 2025 · 6 min read
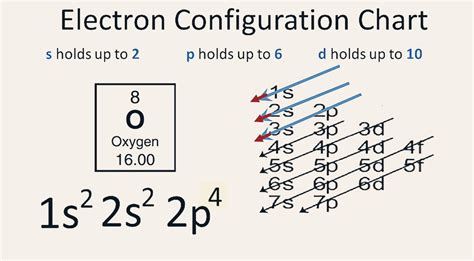
Table of Contents
Electron Configuration for a Neutral Atom of Oxygen: A Deep Dive
Oxygen, the life-giving element, is a fascinating subject for exploring the intricacies of electron configuration. Understanding its electron configuration is key to comprehending its chemical behavior, reactivity, and its crucial role in various biological and chemical processes. This article will provide a comprehensive explanation of oxygen's electron configuration, delving into the underlying principles and exploring its implications.
Understanding Electron Configuration
Before we dive into the specifics of oxygen, let's establish a foundational understanding of electron configuration itself. Electron configuration describes the arrangement of electrons in the various energy levels and sublevels within an atom. These arrangements are governed by specific rules and principles, primarily the Aufbau principle, the Pauli exclusion principle, and Hund's rule.
The Aufbau Principle
The Aufbau principle states that electrons fill the lowest energy levels first before occupying higher energy levels. This is analogous to building a house—you start with the foundation before adding the upper floors. The order of filling is generally: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, and so on.
The Pauli Exclusion Principle
The Pauli exclusion principle dictates that no two electrons within an atom can have the same set of four quantum numbers (principal, azimuthal, magnetic, and spin quantum numbers). This means each orbital can hold a maximum of two electrons, and these electrons must have opposite spins.
Hund's Rule
Hund's rule states that electrons will individually occupy each orbital within a subshell before doubling up in any one orbital. This minimizes electron-electron repulsion and leads to a more stable configuration. Think of it like students choosing seats in a classroom – they'll spread out before pairing up.
Oxygen's Atomic Structure
Oxygen (O) has an atomic number of 8, meaning it has 8 protons in its nucleus and, in a neutral atom, 8 electrons surrounding the nucleus. These electrons are distributed amongst various energy levels and sublevels according to the principles mentioned above.
Determining Oxygen's Electron Configuration
To determine the electron configuration of a neutral oxygen atom, we follow the Aufbau principle and fill the orbitals sequentially:
-
1s²: The first energy level (n=1) contains the 1s subshell, which can hold a maximum of two electrons. These two electrons fill the 1s orbital completely.
-
2s²: The second energy level (n=2) begins with the 2s subshell, which also holds a maximum of two electrons. These two electrons completely fill the 2s orbital.
-
2p⁴: The second energy level also includes the 2p subshell, which consists of three orbitals (2px, 2py, and 2pz), each capable of holding two electrons. This gives the 2p subshell a maximum capacity of six electrons. In oxygen, only four electrons occupy the 2p subshell. According to Hund's rule, these four electrons will fill the three 2p orbitals individually before pairing up. This results in two orbitals with one electron each and one orbital with two electrons.
Therefore, the complete electron configuration of a neutral oxygen atom is 1s²2s²2p⁴.
Orbital Diagrams and Oxygen
Representing the electron configuration using an orbital diagram provides a clearer visual representation. Each orbital is depicted as a box, and electrons are represented by arrows. Up arrows represent spin up (+1/2), and down arrows represent spin down (-1/2). For Oxygen:
- 1s: ↑↓
- 2s: ↑↓
- 2p: ↑ ↑ ↑↓
This diagram visually confirms that each 2p orbital is singly occupied before electron pairing occurs, in accordance with Hund's rule.
Oxygen's Valence Electrons and Reactivity
The valence electrons are the electrons in the outermost energy level of an atom, and they are primarily responsible for an atom's chemical reactivity. In oxygen, the valence electrons are the four electrons in the 2p subshell. This incomplete outermost shell makes oxygen highly reactive, as it readily seeks to gain two more electrons to achieve a stable octet (eight electrons) in its valence shell, thus attaining the electron configuration of a noble gas, Neon.
This drive to achieve a stable octet explains oxygen's tendency to form two covalent bonds, as seen in the oxygen molecule (O₂), or to form ionic bonds by gaining two electrons to form the oxide ion (O²⁻).
Oxygen's Role in Chemical and Biological Processes
Oxygen's unique electron configuration and high reactivity are pivotal to its essential roles in numerous chemical and biological processes. These roles include:
-
Respiration: Oxygen is the final electron acceptor in aerobic respiration, the process by which organisms generate energy from food. The electron configuration of oxygen allows it to readily accept electrons, driving the energy-producing reactions.
-
Combustion: Oxygen is a key reactant in combustion reactions, where it reacts with fuels to release energy in the form of heat and light.
-
Oxidation: Oxygen participates extensively in oxidation reactions, where it acts as an oxidizing agent, accepting electrons from other substances. This is crucial in processes such as rusting and the degradation of organic matter.
Electron Configuration and Spectroscopy
The electron configuration of oxygen also plays a vital role in understanding its spectral lines. When oxygen atoms absorb energy, their electrons transition to higher energy levels. When these excited electrons return to their ground state (the electron configuration described above), they emit photons of light at specific wavelengths. The analysis of these spectral lines provides further confirmation of oxygen's electron configuration and energy levels.
Isotopes and Electron Configuration
While the electron configuration described above is for a neutral oxygen atom with 8 protons and 8 electrons, oxygen also exists as isotopes. Isotopes are atoms of the same element with the same number of protons but differing numbers of neutrons. While the number of neutrons affects the atomic mass, it does not alter the electron configuration of a neutral atom. Therefore, all isotopes of oxygen will have the same electron configuration: 1s²2s²2p⁴. The difference lies solely in their nuclear mass and potentially their stability.
Conclusion: The Significance of Oxygen's Electron Configuration
The electron configuration of oxygen, 1s²2s²2p⁴, is not just a theoretical arrangement of electrons. It's the fundamental basis for understanding oxygen's chemical properties, reactivity, and its indispensable role in life itself. By grasping the underlying principles of electron configuration—the Aufbau principle, the Pauli exclusion principle, and Hund's rule—we gain a deeper appreciation for the elegance and predictability of atomic structure and chemical behavior. From combustion to respiration, the behavior of oxygen is directly linked to its electronic structure, solidifying its importance in both the natural and man-made worlds. The seemingly simple arrangement of eight electrons around the oxygen nucleus is anything but simple; it is a foundation of immense chemical and biological significance.
Latest Posts
Latest Posts
-
What Is The Radius Of Gyration
Apr 05, 2025
-
How Many Inches Are In 18 Cm
Apr 05, 2025
-
Difference Between Starch And Glycogen And Cellulose
Apr 05, 2025
-
300 Inches Is How Many Feet
Apr 05, 2025
-
A Bomb Calorimeter Is Used To Measure
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Electron Configuration For A Neutral Atom Of Oxygen . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
