What Are Rows Called In The Periodic Table
Juapaving
Apr 02, 2025 · 6 min read
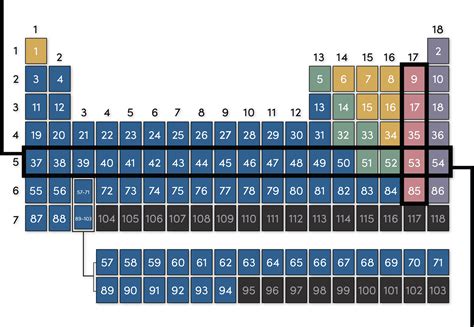
Table of Contents
What are Rows Called in the Periodic Table? Understanding Periods and Their Significance
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic number and recurring chemical properties. While most are familiar with the table's columns, known as groups or families, understanding the rows, or periods, is equally crucial for grasping the underlying principles of chemical behavior. This comprehensive guide will delve into the terminology, structure, and significance of the rows in the periodic table, exploring how they reflect the arrangement of electrons within atoms and consequently influence the elements' properties.
Understanding the Periodic Table's Structure: Rows vs. Columns
The periodic table’s organization isn't arbitrary; it's a carefully constructed system reflecting fundamental atomic structure. The elements are arranged in a grid format, with:
-
Periods (Rows): Horizontal rows, numbered 1 through 7. Each period represents a principal energy level or electron shell. Elements within the same period have their outermost electrons in the same principal energy level.
-
Groups or Families (Columns): Vertical columns, numbered 1 through 18. Elements in the same group share similar chemical properties due to having the same number of valence electrons (electrons in the outermost shell).
The unique arrangement, where periods and groups intersect, allows for predictions about an element's properties based solely on its location. This predictive power is a major reason why the periodic table is such an indispensable tool for chemists and scientists across diverse fields.
The Significance of Periods: Electron Shells and Atomic Radius
The primary significance of periods lies in their direct correlation with electron shells. Each period corresponds to a principal energy level where electrons are located. As you move across a period, electrons are successively added to the same outermost electron shell. This addition subtly alters the atom's size and how strongly it holds onto its electrons, influencing its chemical reactivity.
Atomic Radius Trends Across a Period
As you progress across a period from left to right, the atomic radius generally decreases. This is because the increasing number of protons in the nucleus exerts a stronger pull on the electrons, drawing them closer to the center. This trend is a direct consequence of increased nuclear charge and the relatively unchanging shielding effect of inner electrons within the same principal energy level.
This shrinking atomic radius across a period has important consequences for several chemical properties. For instance, the smaller atomic size often leads to:
-
Increased ionization energy: More energy is required to remove an electron from a smaller atom because of the stronger attraction from the nucleus.
-
Increased electronegativity: Smaller atoms tend to attract electrons more strongly in chemical bonds, leading to higher electronegativity values.
Periodicity of Properties: A Deeper Dive into Period Trends
The periodic table's name itself, “periodic,” highlights the repeating patterns of properties observed across periods and down groups. While atomic radius is a primary example, many other properties exhibit cyclical variations:
Ionization Energy
Ionization energy, the energy required to remove an electron from an atom, generally increases across a period. As mentioned, the increased nuclear charge pulls electrons more tightly, making them harder to remove. However, subtle irregularities appear due to electron configurations and electron-electron repulsions within subshells.
Electronegativity
Electronegativity, an atom's tendency to attract electrons in a chemical bond, also generally increases across a period. Smaller atoms with greater nuclear charge more effectively attract electrons involved in bonding, resulting in higher electronegativity.
Electron Affinity
Electron affinity, the energy change when an atom gains an electron, shows a more complex trend across a period. Although there’s a general tendency toward higher electron affinity across a period, exceptions exist due to electronic shell filling and the stability of half-filled or fully filled subshells.
Periods and the Building-Up Principle
The arrangement of elements within periods is directly linked to the building-up principle, also known as the Aufbau principle. This principle dictates that electrons fill atomic orbitals in order of increasing energy levels, starting with the lowest energy level and working upwards. Each period represents a filling of a principal energy level.
-
Period 1: Fills the 1s orbital (2 electrons maximum). Contains only Hydrogen and Helium.
-
Period 2: Fills the 2s and 2p orbitals (8 electrons maximum). Contains elements like Lithium, Beryllium, Boron, Carbon, Nitrogen, Oxygen, Fluorine, and Neon.
-
Period 3: Fills the 3s and 3p orbitals (8 electrons maximum). Follows a similar pattern to period 2, but with larger atoms.
-
Period 4 onwards: The complexity increases as d and f orbitals are incorporated, resulting in the transition metals and inner transition metals (lanthanides and actinides) and leading to more nuanced trends in properties.
The Significance of Transition Metals and Inner Transition Metals in Periods
Periods 4 through 7 include transition metals, characterized by partially filled d orbitals. These elements exhibit unique properties due to the involvement of d electrons in bonding, often leading to multiple oxidation states and colorful compounds.
Furthermore, periods 6 and 7 accommodate the inner transition metals, also known as the lanthanides and actinides, respectively. These elements, with partially filled f orbitals, have distinct properties and applications in various fields, ranging from lighting (lanthanides) to nuclear technology (actinides).
Predicting Properties Using Periodicity
The periodic table's strength lies in its predictive power. Knowing an element's position in the periodic table (period and group) allows chemists to anticipate many of its properties:
-
Metallic Character: Elements on the left side of a period generally exhibit stronger metallic characteristics (luster, conductivity, malleability), while those on the right tend to be non-metals.
-
Reactivity: Highly reactive metals are found at the beginning of periods (alkali and alkaline earth metals), while highly reactive non-metals are found toward the end (halogens).
-
Oxidation States: The potential oxidation states of an element can often be predicted from its group number.
Applications and Relevance of Periodicity
Understanding periods in the periodic table is vital in numerous scientific and technological applications. These include:
-
Material Science: Designing new materials with specific properties, such as strength, conductivity, or reactivity.
-
Drug Discovery: Understanding the interaction of elements with biological molecules to develop new drugs and therapies.
-
Catalysis: Designing catalysts for chemical reactions, using elements with specific electronic configurations.
-
Environmental Science: Assessing the impact of elements on the environment and developing strategies for remediation.
Conclusion: The Enduring Importance of Understanding Periods
In summary, the rows (periods) of the periodic table represent a crucial aspect of its organization and predictive power. They directly correlate with the filling of principal energy levels within atoms, providing a framework for understanding the systematic trends in various atomic and chemical properties. By understanding the significance of periods and their influence on atomic structure, we gain valuable insights into the behavior of elements, leading to a deeper appreciation of the periodic table’s importance in chemistry and related fields. From predicting the reactivity of an element to designing new materials with tailored properties, the understanding of periods forms the fundamental groundwork for countless scientific and technological advancements. The periodic table’s organization, therefore, remains a cornerstone of chemical knowledge and a constantly evolving tool for discovery and innovation.
Latest Posts
Latest Posts
-
A Nucleotide Is Made Of Three Parts
Apr 03, 2025
-
Who Is The Father Of Sociology
Apr 03, 2025
-
Whats The Square Root Of 30
Apr 03, 2025
-
How Many Feet Is 18 Yards
Apr 03, 2025
-
Facts And Statistics Collected Together For Reference Or Analysis
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Are Rows Called In The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
