Titration Of A Strong Base With A Strong Acid
Juapaving
Apr 03, 2025 · 6 min read
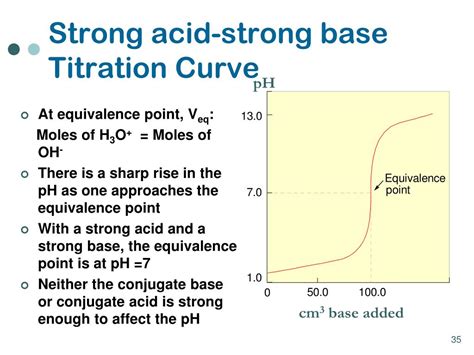
Table of Contents
Titration of a Strong Base with a Strong Acid: A Comprehensive Guide
Titration is a fundamental analytical technique in chemistry used to determine the concentration of an unknown solution, known as the analyte, by reacting it with a solution of known concentration, called the titrant. This article delves into the specifics of titrating a strong base with a strong acid, exploring the underlying chemistry, the procedure, calculations, and applications. Understanding this process is crucial for students and professionals alike in various fields, including chemistry, environmental science, and pharmaceuticals.
Understanding Strong Acids and Strong Bases
Before diving into the titration process, it's crucial to understand the properties of strong acids and strong bases.
Strong acids are acids that completely dissociate in water, releasing all their hydrogen ions (H⁺). Common examples include hydrochloric acid (HCl), sulfuric acid (H₂SO₄), nitric acid (HNO₃), and perchloric acid (HClO₄). Their complete dissociation results in a high concentration of H⁺ ions, leading to a low pH.
Strong bases are bases that completely dissociate in water, releasing all their hydroxide ions (OH⁻). Common strong bases include sodium hydroxide (NaOH), potassium hydroxide (KOH), lithium hydroxide (LiOH), and calcium hydroxide (Ca(OH)₂). Their complete dissociation results in a high concentration of OH⁻ ions, leading to a high pH.
The Neutralization Reaction
The core principle behind the titration of a strong base with a strong acid is the neutralization reaction. This is an acid-base reaction where the hydrogen ions (H⁺) from the acid react with the hydroxide ions (OH⁻) from the base to form water (H₂O). The general equation is:
H⁺(aq) + OH⁻(aq) → H₂O(l)
This reaction is exothermic, meaning it releases heat. The heat released can be used to determine the endpoint of the titration using techniques like thermometric titration.
The Titration Process: A Step-by-Step Guide
The titration of a strong base with a strong acid involves several key steps:
-
Preparation: Accurately prepare a known concentration of the strong acid titrant. This is usually done by dissolving a precisely weighed amount of the acid in a specific volume of distilled water. Precise measurements are crucial for accurate results.
-
Setting up the apparatus: A burette is filled with the standardized strong acid titrant. A precisely measured volume of the strong base analyte is placed in an Erlenmeyer flask. An indicator, such as phenolphthalein, is added to the flask. Phenolphthalein is colorless in acidic solutions and pink in basic solutions.
-
Titration: The strong acid is added dropwise from the burette to the strong base in the flask while constantly swirling the flask to ensure complete mixing. The addition continues until the endpoint is reached.
-
Endpoint Determination: The endpoint is reached when the indicator changes color, signaling that the neutralization reaction is complete. For phenolphthalein, this is the point where the pink color disappears, indicating that the solution has become slightly acidic.
-
Data Recording: The initial and final burette readings are recorded to calculate the volume of strong acid used to neutralize the strong base. This volume is crucial for calculating the concentration of the unknown base.
Choosing the Right Indicator
Indicator selection is critical for accurate titration. The indicator should change color near the equivalence point of the titration. The equivalence point is the point where the moles of acid are equal to the moles of base. For strong acid-strong base titrations, the equivalence point is at pH 7. Phenolphthalein is a suitable indicator for strong acid-strong base titrations because its color change occurs near pH 7. Other indicators, like methyl orange or bromothymol blue, can also be used. However, careful selection considering the pH range of color change is paramount.
Calculations and Data Analysis
Once the titration is complete, the concentration of the unknown strong base can be calculated using the following formula:
M₁V₁ = M₂V₂
Where:
- M₁ = Molarity of the strong acid titrant (known)
- V₁ = Volume of the strong acid titrant used (measured)
- M₂ = Molarity of the strong base analyte (unknown)
- V₂ = Volume of the strong base analyte (known)
This formula is based on the stoichiometry of the neutralization reaction (1:1 mole ratio for strong acid-strong base). Solving for M₂, we can determine the concentration of the unknown strong base.
Sources of Error and Mitigation Strategies
Several factors can influence the accuracy of titration results. These include:
-
Improper standardization of the titrant: Inaccurate preparation of the strong acid titrant will directly impact the results. Using analytical balances and volumetric flasks with appropriate precision is vital.
-
Parallax error: Incorrect reading of the burette meniscus can lead to errors in the volume measurement. Reading at eye level to minimize parallax error is essential.
-
Indicator error: The indicator might not change color precisely at the equivalence point, leading to slight deviations. Choosing a suitable indicator with a sharp color change near the equivalence point minimizes this error.
-
Incomplete mixing: Inadequate mixing during the titration can lead to uneven reaction and inaccurate results. Consistent swirling ensures a homogenous reaction mixture.
-
Contamination: Impurities in the solutions or glassware can interfere with the reaction and affect the accuracy. Using clean glassware and high-purity chemicals is paramount.
By carefully controlling these factors and employing precise techniques, the accuracy of titration results can be significantly improved.
Applications of Strong Acid-Strong Base Titration
Strong acid-strong base titrations have a wide range of applications in various fields:
-
Determining the concentration of unknown solutions: This is the primary application, useful in various chemical analyses and quality control.
-
Acid-base content analysis: Determining the acidity or basicity of samples in various industries such as food processing, pharmaceuticals, and environmental monitoring.
-
Water analysis: Determining the alkalinity or acidity of water samples to assess water quality.
-
Soil analysis: Determining the pH of soil samples to assess its suitability for agriculture.
-
Pharmaceutical analysis: Determining the purity and concentration of active pharmaceutical ingredients.
Advanced Titration Techniques
While the standard titration procedure outlined above is widely applicable, several advanced techniques enhance precision and adaptability:
-
Potentiometric titration: This technique uses a pH meter to monitor the pH changes during the titration, offering a more precise determination of the equivalence point compared to using indicators.
-
Conductometric titration: This method measures the conductivity changes during the titration. The equivalence point is detected by a sharp change in conductivity.
-
Thermometric titration: As previously mentioned, this technique monitors the temperature changes during the titration. The equivalence point is indicated by a change in the rate of temperature change.
These advanced techniques are often preferred for titrations requiring higher accuracy or dealing with complex samples.
Conclusion
The titration of a strong base with a strong acid is a fundamental analytical technique with wide-ranging applications. By understanding the underlying chemistry, mastering the procedure, accurately performing calculations, and utilizing appropriate techniques, one can effectively determine the concentration of unknown strong bases and perform various analytical tasks in a variety of settings. The accuracy and reliability of the results hinge upon careful execution, precision in measurements, and meticulous attention to detail throughout the process. Adherence to best practices, as described above, ensures that the titration yields valuable data, contributing to informed decision-making in diverse scientific and industrial contexts. The understanding of potential errors and the implementation of mitigation strategies are equally crucial to achieve reliable and meaningful results.
Latest Posts
Latest Posts
-
Who Is Credited For Discovering Cells
Apr 03, 2025
-
Is 7 A Prime Or Composite Number
Apr 03, 2025
-
Are Humans Warm Blooded Or Cold Blooded
Apr 03, 2025
-
What Is 0 08 As A Percent
Apr 03, 2025
-
How Many Chambers Does A Fish Heart Have
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Titration Of A Strong Base With A Strong Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
