The Horizontal Rows In The Periodic Table Are Called
Juapaving
Mar 31, 2025 · 6 min read
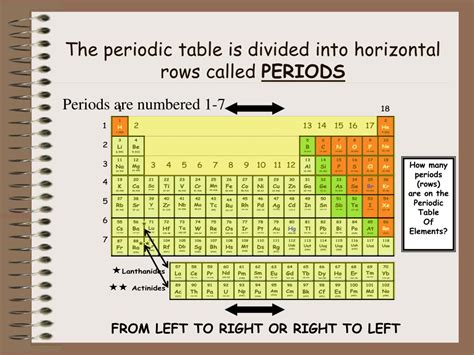
Table of Contents
- The Horizontal Rows In The Periodic Table Are Called
- Table of Contents
- The Horizontal Rows in the Periodic Table are Called: Periods – A Deep Dive into Periodic Trends
- Understanding Periods: A Structural Overview
- Number of Periods and Electron Shells
- Periodic Trends Across Periods: A Detailed Examination
- 1. Atomic Radius: A Decreasing Trend
- 2. Ionization Energy: An Increasing Trend
- 3. Electronegativity: An Increasing Trend
- 4. Electron Affinity: A Generally Increasing Trend (with exceptions)
- 5. Metallic Character: A Decreasing Trend
- 6. Reactivity: A Complex Trend
- Implications of Periodic Trends: Examples in Chemical Reactions
- Beyond the Basics: Exploring the Exceptions
- The Significance of Periods in Chemical Understanding
- Latest Posts
- Latest Posts
- Related Post
The Horizontal Rows in the Periodic Table are Called: Periods – A Deep Dive into Periodic Trends
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. While many focus on the vertical columns (groups), understanding the horizontal rows, known as periods, is equally crucial to grasping the nuances of chemical behavior. This comprehensive guide delves into the significance of periods, explaining their structure, the periodic trends they exhibit, and their implications in various chemical phenomena.
Understanding Periods: A Structural Overview
The horizontal rows in the periodic table are called periods. Each period represents a principal energy level or shell within an atom. As we move across a period from left to right, the atomic number increases by one, signifying the addition of a proton and, typically, an electron. This gradual increase in protons and electrons profoundly influences the properties of the elements within that period.
Number of Periods and Electron Shells
There are currently seven periods in the periodic table. Each period corresponds to a particular electron shell. The first period (containing hydrogen and helium) only has one electron shell, while subsequent periods add more electron shells. The number of elements within each period varies, reflecting the complexity of filling electron subshells (s, p, d, f).
- Period 1: Contains only two elements (hydrogen and helium) and represents the filling of the 1s subshell.
- Period 2: Contains eight elements (lithium to neon) reflecting the filling of the 2s and 2p subshells.
- Period 3: Also contains eight elements (sodium to argon), similarly filling the 3s and 3p subshells.
- Period 4: Contains 18 elements (potassium to krypton), with the addition of the 3d subshell filling after the 4s subshell.
- Period 5: Contains 18 elements (rubidium to xenon), mirroring the structure of period 4.
- Period 6: Contains 32 elements (cesium to radon), including the filling of the 4f subshell (lanthanides) after the 6s subshell.
- Period 7: Is an incomplete period, with elements currently extending to oganesson. The filling of the 5f subshell (actinides) contributes to the extended length of this period. Further elements are predicted but yet to be synthesized and verified.
This increasing complexity in electron shell filling directly correlates with the observed trends in properties across the periods.
Periodic Trends Across Periods: A Detailed Examination
The systematic arrangement of elements in periods allows us to observe predictable trends in their properties. These trends are fundamental to understanding chemical reactivity, bonding, and the overall behavior of elements. Key periodic trends across a period include:
1. Atomic Radius: A Decreasing Trend
Atomic radius refers to the size of an atom. As we move across a period from left to right, the atomic radius generally decreases. This is primarily due to an increase in the effective nuclear charge. With the addition of protons in the nucleus, the positive charge increases, pulling the electrons closer to the nucleus and thus reducing the atomic size. Electrons are added to the same principal energy level, and the shielding effect remains relatively constant, leading to a stronger attraction between the nucleus and the outer electrons.
2. Ionization Energy: An Increasing Trend
Ionization energy is the energy required to remove an electron from a gaseous atom. As we move across a period, the ionization energy generally increases. This is a direct consequence of the increasing effective nuclear charge. The stronger attraction between the nucleus and electrons makes it increasingly difficult to remove an electron, hence the higher ionization energy. The electrons are held more tightly, requiring more energy to overcome the electrostatic attraction.
3. Electronegativity: An Increasing Trend
Electronegativity measures the ability of an atom to attract electrons in a chemical bond. Across a period, electronegativity generally increases. This is again related to the rising effective nuclear charge. Atoms with higher electronegativity have a stronger pull on shared electrons in a covalent bond, resulting in a more polar bond. Fluorine, located in the upper right corner (excluding noble gases), is the most electronegative element.
4. Electron Affinity: A Generally Increasing Trend (with exceptions)
Electron affinity represents the energy change when an electron is added to a neutral gaseous atom. While there are exceptions, across a period, electron affinity generally increases. This is because the addition of an electron to an atom with a higher effective nuclear charge releases more energy. However, exceptions occur due to electron shell configurations and the stability of half-filled or completely filled subshells.
5. Metallic Character: A Decreasing Trend
Metallic character refers to the properties associated with metals, such as conductivity, malleability, and ductility. Across a period, metallic character generally decreases. This is because as we move across a period, the atoms become smaller and have a stronger hold on their electrons. This makes them less likely to lose electrons and exhibit metallic properties. Non-metals dominate the right side of the period, showing characteristics opposite to metals.
6. Reactivity: A Complex Trend
The reactivity of elements varies across a period in a complex manner. For metals (left side), reactivity generally decreases across the period due to the increasing ionization energy. For non-metals (right side), reactivity generally increases towards the halogens (Group 17), which readily gain electrons to achieve a stable octet. Noble gases (Group 18) are generally unreactive due to their stable electron configurations.
Implications of Periodic Trends: Examples in Chemical Reactions
Understanding these periodic trends is vital for predicting the behavior of elements in chemical reactions and forming compounds. For instance:
-
Formation of Ionic Compounds: Highly reactive alkali metals (Group 1) readily lose an electron to form a +1 ion, while highly electronegative halogens (Group 17) readily gain an electron to form a -1 ion. The electrostatic attraction between these oppositely charged ions leads to the formation of ionic compounds like sodium chloride (NaCl).
-
Covalent Bonding: Nonmetals in the same period tend to share electrons to achieve a stable octet, forming covalent bonds. The electronegativity difference between the atoms influences the polarity of the covalent bond.
-
Oxidation States: Periodic trends help predict the common oxidation states exhibited by elements. For example, Group 1 elements typically exhibit a +1 oxidation state, while Group 2 elements exhibit a +2 oxidation state.
-
Catalysis: The electronic structure and properties of elements determined by their position in the periodic table influence their catalytic activity. Transition metals, occupying the d-block, frequently serve as catalysts due to their variable oxidation states and ability to form complexes.
Beyond the Basics: Exploring the Exceptions
While periodic trends are generally predictable, there are exceptions. These exceptions often arise from the complexities of electron-electron interactions, the subtle nuances of electron shielding, and the stability of half-filled or fully filled subshells. Careful consideration of these factors is necessary for a complete understanding of elemental behavior.
The Significance of Periods in Chemical Understanding
Understanding the horizontal rows (periods) in the periodic table is fundamental to comprehending the fundamental principles of chemistry. The periodic trends exhibited by elements across periods provide a powerful framework for predicting their reactivity, bonding behavior, and overall chemical characteristics. This knowledge is invaluable in various fields, including materials science, pharmaceuticals, environmental chemistry, and numerous others, where understanding the properties and reactions of elements is crucial for developing new materials and technologies. The systematic organization of elements in the periodic table, with its emphasis on periods, is a testament to the elegance and predictive power of scientific organization. The intricate relationships between atomic structure and chemical properties highlighted by periodic trends continue to inspire scientific inquiry and advancements across the spectrum of chemical disciplines.
Latest Posts
Latest Posts
-
What Is 12 Percent Of 180
Apr 03, 2025
-
Find The Prime Factorization Of 100
Apr 03, 2025
-
How Many Centimetres In 2 Metres
Apr 03, 2025
-
Is 16 A Prime Number Or A Composite Number
Apr 03, 2025
-
Everyday Examples Of Newtons First Law Of Motion
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about The Horizontal Rows In The Periodic Table Are Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
