The First 10 Elements Of The Periodic Table
Juapaving
Apr 06, 2025 · 8 min read
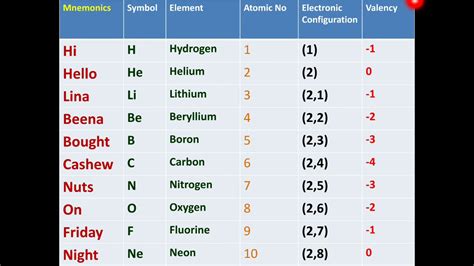
Table of Contents
The First 10 Elements of the Periodic Table: A Deep Dive
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic number and recurring chemical properties. Understanding the fundamental properties of the first ten elements is crucial for grasping the broader principles that govern the behavior of all matter. This article delves deep into the first ten elements – hydrogen, helium, lithium, beryllium, boron, carbon, nitrogen, oxygen, fluorine, and neon – exploring their characteristics, discovery, applications, and significance in the universe.
1. Hydrogen (H): The Most Abundant Element
Hydrogen, with atomic number 1, is the simplest and most abundant element in the universe. Its single proton and electron define its unique properties.
Discovery and Properties:
Henry Cavendish is credited with the discovery of hydrogen in 1766, although he initially mistook it for a type of air. Its lightness, flammability, and reactivity are defining characteristics. Hydrogen exists in three isotopic forms: protium (¹H), deuterium (²H), and tritium (³H), differing in the number of neutrons in their nuclei.
Applications:
Hydrogen's applications are vast and varied. It's a key component in ammonia production (Haber-Bosch process), crucial for fertilizers. It's also used in petroleum refining, the production of methanol, and increasingly, as a clean energy source in fuel cells. Research into hydrogen fuel cell technology is rapidly expanding due to its potential to provide a sustainable energy alternative. The quest for efficient and safe hydrogen storage remains a significant challenge in this area.
2. Helium (He): The Inert Giant
Helium, atomic number 2, is a noble gas, meaning it is exceptionally unreactive. Its two electrons fill its outermost shell, making it exceptionally stable.
Discovery and Properties:
Helium was first discovered in the sun's spectrum in 1868 by Pierre Janssen and Norman Lockyer before being isolated on Earth. Its low density, inertness, and low boiling point are defining features. Helium is much less dense than air, making it useful for balloons and airships.
Applications:
Helium's inertness makes it invaluable in applications requiring an unreactive atmosphere, such as in welding and the production of semiconductors. Its low boiling point makes it crucial for cryogenics, enabling the cooling of superconducting magnets used in MRI machines and scientific research. Helium is also used as a lifting gas in balloons and blimps, although this application faces increasing environmental concerns. The dwindling supply of helium is a growing concern for various industries.
3. Lithium (Li): The Lightweight Metal
Lithium, atomic number 3, is an alkali metal, characterized by its high reactivity and low density.
Discovery and Properties:
Johan August Arfvedson discovered lithium in 1817. It is the lightest metal and has a relatively low melting point. Its reactivity makes it prone to oxidation in air.
Applications:
Lithium's applications are rapidly expanding, particularly in batteries. Lithium-ion batteries power a wide range of portable devices, electric vehicles, and energy storage systems. Lithium is also used in ceramics, glass, and lubricating greases. The demand for lithium is soaring due to the growth of the electric vehicle market and the need for energy storage solutions. Concerns about the environmental impact of lithium mining and processing are increasingly important considerations.
4. Beryllium (Be): A Strong and Toxic Metal
Beryllium, atomic number 4, is an alkaline earth metal known for its strength and toxicity.
Discovery and Properties:
Louis Nicolas Vauquelin identified beryllium in 1798. It is a lightweight yet exceptionally strong metal with high melting and boiling points. However, beryllium is highly toxic; inhaling beryllium dust can cause serious lung disease (berylliosis).
Applications:
Despite its toxicity, beryllium's unique properties make it valuable in specialized applications. It's used in aerospace components, X-ray windows, and nuclear reactors due to its low density and neutron transparency. The handling of beryllium requires strict safety precautions due to its toxicity. Sustainable alternatives to beryllium are being explored to mitigate the health risks associated with its use.
5. Boron (B): A Metalloid with Diverse Uses
Boron, atomic number 5, is a metalloid, exhibiting properties of both metals and nonmetals.
Discovery and Properties:
Sir Humphry Davy, Jöns Jakob Berzelius, and Louis Joseph Gay-Lussac independently isolated boron in the early 19th century. Boron exists in several allotropic forms, differing in their physical properties. It is a relatively hard and brittle element.
Applications:
Boron's compounds are used extensively in various applications. Boric acid is used as an antiseptic and insecticide. Boron fibers are used in advanced composites for their high strength-to-weight ratio. Boron is also a key component in borosilicate glass (Pyrex), known for its resistance to thermal shock. The expanding use of boron in materials science continues to drive research and development in this area.
6. Carbon (C): The Basis of Life
Carbon, atomic number 6, is the cornerstone of organic chemistry and the basis of life as we know it.
Discovery and Properties:
Carbon's discovery dates back to antiquity. It exists in various allotropes, most notably diamond and graphite, exhibiting vastly different properties due to their distinct atomic structures. Carbon’s ability to form four strong covalent bonds makes it uniquely capable of building complex molecules.
Applications:
Carbon's applications are virtually limitless. Diamond is renowned for its hardness and used in cutting tools and jewelry. Graphite is used in pencils, lubricants, and electrodes. Fullerenes (e.g., buckminsterfullerene, or "buckyballs") and nanotubes are exciting areas of nanotechnology research, offering potential applications in materials science and electronics. Carbon is fundamental to all living organisms, forming the backbone of organic molecules such as proteins, carbohydrates, and lipids.
7. Nitrogen (N): An Essential Gas
Nitrogen, atomic number 7, is a diatomic gas that comprises about 78% of the Earth's atmosphere.
Discovery and Properties:
Daniel Rutherford is generally credited with the discovery of nitrogen in 1772. It is relatively unreactive under normal conditions but can form various compounds with other elements.
Applications:
Nitrogen's uses are widespread. It is crucial for the production of ammonia (via the Haber-Bosch process), a vital component of fertilizers. It is also used in the food industry as a preservative and in the electronics industry in the manufacture of semiconductors. Liquid nitrogen is used as a cryogenic refrigerant. The increasing demand for nitrogen-based fertilizers underscores its importance in agriculture and food production.
8. Oxygen (O): Essential for Respiration
Oxygen, atomic number 8, is a diatomic gas essential for respiration in most living organisms.
Discovery and Properties:
Carl Wilhelm Scheele and Joseph Priestley independently discovered oxygen in the late 18th century. It is a highly reactive element, readily combining with many other elements to form oxides. Oxygen is crucial for combustion processes.
Applications:
Oxygen's applications are numerous. It's vital for respiration in humans and animals. It is used in medicine to treat respiratory conditions. It plays a crucial role in combustion processes, powering engines and industrial furnaces. Oxygen is also used in various industrial processes, including welding and metal fabrication. The growing importance of oxygen in medicine and industry makes it a critically important element.
9. Fluorine (F): The Most Reactive Element
Fluorine, atomic number 9, is the most electronegative and reactive element in the periodic table.
Discovery and Properties:
Henri Moissan successfully isolated fluorine in 1886. Its high reactivity makes it extremely difficult to handle and requires specialized techniques for safe containment.
Applications:
Despite its reactivity, fluorine's compounds have found important applications. Fluoride compounds are added to toothpaste and drinking water to prevent tooth decay. Fluorocarbons are used as refrigerants (although their use is declining due to environmental concerns). Fluorine is also used in the production of certain plastics and pharmaceuticals. The balance between fluorine’s usefulness and its potential environmental and health impacts requires careful consideration.
10. Neon (Ne): The Glowing Gas
Neon, atomic number 10, is a noble gas known for its characteristic reddish-orange glow when electrically excited.
Discovery and Properties:
Sir William Ramsay and Morris Travers discovered neon in 1898. It is an inert gas with a low boiling point. Its distinctive glow when subjected to an electric discharge makes it useful in signage.
Applications:
Neon's primary use is in neon signs, where its characteristic glow provides vibrant lighting. It is also used in some lasers and as a cryogenic refrigerant. Its inertness makes it suitable for applications requiring an unreactive atmosphere. While neon signs remain iconic, the use of neon is relatively limited compared to some other elements on the periodic table.
Conclusion: Building Blocks of Matter
The first ten elements of the periodic table, while only a small fraction of the entire table, represent a crucial foundation for understanding the chemical behavior of matter. Their unique properties, applications, and roles in various scientific and technological fields highlight the fundamental importance of these elements in shaping our world. Continued research into these elements, as well as their interactions and applications, will undoubtedly lead to further advancements in diverse fields, further cementing their significance in chemistry and beyond. The careful consideration of the environmental and health implications associated with the use of these elements is crucial for responsible scientific and technological development.
Latest Posts
Latest Posts
-
The Pigment Molecules Responsible For Photosynthesis Are Located In The
Apr 06, 2025
-
How Many Factors Does The Number 121 Have
Apr 06, 2025
-
How Much Time Is 90 Minutes
Apr 06, 2025
-
How Many Quarts Of Dirt In A Cubic Foot
Apr 06, 2025
-
Which Of The Following Is Equal To 5 1 3
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about The First 10 Elements Of The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
