Is Water A Pure Substance Or Mixture
Juapaving
Apr 05, 2025 · 4 min read
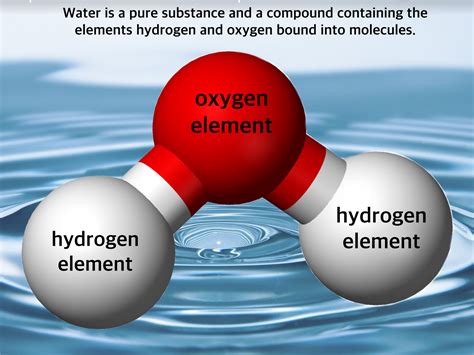
Table of Contents
Is Water a Pure Substance or a Mixture? A Deep Dive into the Chemistry of H₂O
The seemingly simple question, "Is water a pure substance or a mixture?" delves into the fundamental concepts of chemistry and the intricacies of matter. While the answer might seem straightforward at first glance, a deeper exploration reveals a nuanced understanding of purity, substances, and mixtures, especially when considering the real-world context of water. Let's unravel this fascinating topic.
Defining Pure Substances and Mixtures
Before tackling the question about water, let's establish clear definitions:
Pure Substances
A pure substance is a form of matter that has a constant composition and properties throughout the sample. It cannot be separated into components by physical methods. Pure substances can be either elements or compounds.
-
Elements: These are substances that cannot be broken down into simpler substances by chemical means. Examples include oxygen (O), hydrogen (H), and iron (Fe). They are fundamental building blocks of all matter.
-
Compounds: These are substances formed when two or more elements chemically combine in a fixed ratio. Water (H₂O), for instance, is a compound composed of two hydrogen atoms and one oxygen atom, always in this precise 2:1 ratio. The properties of a compound are distinct from the properties of its constituent elements.
Mixtures
A mixture, in contrast, consists of two or more substances physically combined, retaining their individual chemical identities. Mixtures can be separated into their components by physical methods like filtration, distillation, or evaporation. Mixtures can be homogeneous or heterogeneous.
-
Homogeneous Mixtures: These have a uniform composition throughout the sample. For example, saltwater is a homogeneous mixture where salt is evenly distributed in the water. You can't visually distinguish the salt from the water.
-
Heterogeneous Mixtures: These have a non-uniform composition, with different parts having visibly distinct properties. A salad, for example, is a heterogeneous mixture with clearly identifiable ingredients like lettuce, tomatoes, and cucumbers.
The Case of Water: Pure or Impure?
Now, let's examine water. In its purest form, water (H₂O) is indeed a pure substance, a compound formed by the chemical bonding of two hydrogen atoms and one oxygen atom. This pure water is characterized by its specific properties:
- Boiling Point: 100°C (212°F) at standard atmospheric pressure.
- Freezing Point: 0°C (32°F) at standard atmospheric pressure.
- Density: 1 g/mL at 4°C.
- Colorless, Odorless, and Tasteless: Pure water lacks any color, odor, or taste.
However, the water we encounter in everyday life is rarely pure. Natural water sources, like rivers, lakes, and oceans, contain dissolved minerals, gases, and various organic and inorganic substances. This makes them mixtures.
Types of Impurities in Water
The impurities in water can significantly alter its properties. These include:
-
Dissolved Minerals: Calcium, magnesium, sodium, and other minerals are common dissolved ions found in natural water sources, contributing to hardness and influencing taste.
-
Dissolved Gases: Oxygen, carbon dioxide, and nitrogen are gases that can dissolve in water, affecting its pH and biological activity.
-
Organic Matter: Decaying plant and animal matter, along with pollutants from human activities, introduce organic compounds into water bodies.
-
Suspended Solids: Clay, silt, sand, and other particulate matter can be suspended in water, creating turbidity and affecting its clarity.
-
Microbial Contaminants: Bacteria, viruses, and other microorganisms can contaminate water sources, posing health risks.
-
Chemical Pollutants: Industrial and agricultural activities release various chemical pollutants into water bodies, including heavy metals, pesticides, and fertilizers.
The Significance of Water Purity
The purity of water is crucial for various applications. Pure water is essential for:
-
Laboratory Experiments: In scientific research, pure water is vital to avoid interfering with experimental results.
-
Pharmaceutical Industry: Pure water is used in the production of pharmaceuticals to maintain product integrity and safety.
-
Medical Applications: Pure water is essential in many medical procedures and treatments.
-
Industrial Processes: Numerous industrial processes rely on pure water to prevent corrosion, fouling, and other issues.
-
Drinking Water: While complete purity is not always necessary or even desirable for drinking water (some minerals are beneficial), the absence of harmful contaminants is paramount.
Methods for Purifying Water
Several methods are employed to purify water, removing unwanted substances and enhancing its purity:
-
Filtration: This process removes suspended solids and larger particles from water by passing it through a filter medium.
-
Distillation: Distillation involves boiling water and then condensing the steam, leaving behind dissolved solids and other non-volatile impurities.
-
Reverse Osmosis: This membrane-based process forces water through a semipermeable membrane, removing dissolved salts and other impurities.
-
Activated Carbon Treatment: Activated carbon filters effectively adsorb organic contaminants and chlorine from water.
-
UV Disinfection: Ultraviolet (UV) light can be used to kill harmful microorganisms in water.
Conclusion: Context Matters
Therefore, the answer to "Is water a pure substance or a mixture?" depends on the context. Pure water (H₂O) is a pure substance, a compound. However, water found in nature is typically a mixture, containing various impurities. Understanding this distinction is fundamental to appreciating the diverse properties and applications of water, and the importance of its purification for various purposes. The presence of impurities fundamentally shifts the characteristics of the water and impacts its suitability for different uses. Maintaining water purity is vital for public health, environmental protection, and numerous industrial applications.
Latest Posts
Latest Posts
-
A Substance Formed As A Result Of A Chemical Reaction
Apr 05, 2025
-
A Frequency Polygon Is Graphed Using
Apr 05, 2025
-
Is Fungus A Consumer Or Producer
Apr 05, 2025
-
How Long Does The Sun Take To Rotate
Apr 05, 2025
-
Least Common Multiple Of 24 And 40
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Is Water A Pure Substance Or Mixture . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
