Electrons Are Lost Or Gained During
Juapaving
Apr 02, 2025 · 6 min read
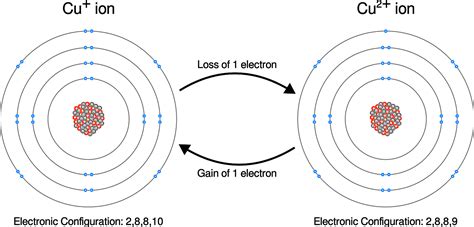
Table of Contents
Electrons Are Lost or Gained During: A Deep Dive into Oxidation and Reduction
The fundamental processes of chemistry hinge on the behavior of electrons. More specifically, the transfer of electrons between atoms and molecules is the driving force behind countless reactions, from the rusting of iron to the processes that power our cells. This transfer is central to understanding oxidation and reduction reactions, collectively known as redox reactions. Understanding when electrons are lost or gained is crucial to predicting and manipulating chemical behavior.
Understanding Electron Transfer: The Heart of Redox Reactions
At the core of all redox reactions lies the movement of electrons. Atoms strive for stability, usually achieved by having a full outer electron shell. This drive for stability dictates whether an atom will readily lose or gain electrons.
-
Oxidation: This describes the loss of electrons by an atom, molecule, or ion. When an atom loses electrons, its oxidation state increases (becomes more positive). Think of oxidation as an atom becoming more "electron-deficient."
-
Reduction: This describes the gain of electrons by an atom, molecule, or ion. When an atom gains electrons, its oxidation state decreases (becomes more negative). Think of reduction as an atom becoming more "electron-rich."
The terms "oxidation" and "reduction" are always coupled. You cannot have one without the other. When one species is oxidized (loses electrons), another must be reduced (gains those electrons). This is why they are called redox reactions: a simultaneous reduction-oxidation process.
Identifying Oxidation and Reduction: Practical Applications
Identifying which species is oxidized and which is reduced in a reaction is paramount. Several methods exist to help determine this:
1. Tracking Changes in Oxidation States:
The oxidation state (or oxidation number) of an atom represents the hypothetical charge an atom would have if all bonds to atoms of different elements were 100% ionic. This is a useful tool, even though few bonds are purely ionic.
- Rules for Assigning Oxidation States: These rules are hierarchical. You should apply them in order.
- The oxidation state of an element in its free (uncombined) state is always 0. (e.g., O₂ , Na, Cl₂)
- The oxidation state of a monatomic ion is equal to its charge. (e.g., Na⁺ = +1, Cl⁻ = -1)
- The sum of the oxidation states of all atoms in a neutral molecule must be 0.
- The sum of the oxidation states of all atoms in a polyatomic ion must equal the charge of the ion.
- The oxidation state of hydrogen is usually +1, except in metal hydrides (e.g., NaH) where it is -1.
- The oxidation state of oxygen is usually -2, except in peroxides (e.g., H₂O₂) where it is -1 and in compounds with fluorine (e.g., OF₂) where it is +2.
- In binary compounds (compounds containing only two elements), the more electronegative element is assigned a negative oxidation state.
Example: Consider the reaction: 2Fe + 3Cl₂ → 2FeCl₃
- Initially, Fe has an oxidation state of 0 (elemental state).
- After the reaction, Fe has an oxidation state of +3 (in FeCl₃).
- Chlorine initially has an oxidation state of 0 (elemental state).
- After the reaction, Chlorine has an oxidation state of -1 (in FeCl₃).
Fe has been oxidized (lost electrons; oxidation state increased from 0 to +3). Cl₂ has been reduced (gained electrons; oxidation state decreased from 0 to -1).
2. Using Half-Reactions:
A powerful method for analyzing redox reactions involves separating the overall reaction into two half-reactions: one for oxidation and one for reduction. This allows a clearer picture of electron transfer.
Example: Consider the reaction: Zn + Cu²⁺ → Zn²⁺ + Cu
- Oxidation Half-Reaction: Zn → Zn²⁺ + 2e⁻ (Zinc loses two electrons)
- Reduction Half-Reaction: Cu²⁺ + 2e⁻ → Cu (Copper gains two electrons)
These half-reactions clearly demonstrate electron transfer. Zn is oxidized (loses electrons), and Cu²⁺ is reduced (gains electrons).
3. Identifying Oxidizing and Reducing Agents:
-
Oxidizing Agent: A substance that causes oxidation in another substance. It itself is reduced in the process. It accepts electrons.
-
Reducing Agent: A substance that causes reduction in another substance. It itself is oxidized in the process. It donates electrons.
In the Zn/Cu reaction above:
- Cu²⁺ is the oxidizing agent (it accepts electrons from Zn).
- Zn is the reducing agent (it donates electrons to Cu²⁺).
Redox Reactions in Everyday Life and Industrial Processes
Redox reactions are ubiquitous, playing crucial roles in numerous everyday phenomena and industrial processes:
1. Combustion:
The burning of fuels (e.g., wood, gasoline) involves the rapid oxidation of the fuel by oxygen. The fuel is oxidized (loses electrons), and oxygen is reduced (gains electrons).
2. Respiration:
Cellular respiration, the process by which living organisms obtain energy, involves a series of redox reactions. Glucose is oxidized, and oxygen is reduced.
3. Corrosion:
Rusting, the corrosion of iron, is a redox reaction. Iron is oxidized (loses electrons to oxygen), forming iron oxide (rust).
4. Batteries:
Batteries utilize redox reactions to generate electricity. A spontaneous redox reaction drives the flow of electrons, creating an electric current.
5. Electroplating:
Electroplating, a process used to coat a metal object with another metal, involves redox reactions. The metal to be plated is reduced (gains electrons), and the metal being used as the plating source is oxidized (loses electrons).
6. Photography:
The development of photographic film involves redox reactions. Silver halide crystals are reduced to metallic silver, forming the image.
Factors Influencing Electron Transfer
Several factors influence the tendency of an atom or molecule to lose or gain electrons:
1. Electronegativity:
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. Atoms with high electronegativity tend to gain electrons (be reduced), while atoms with low electronegativity tend to lose electrons (be oxidized).
2. Ionization Energy:
Ionization energy is the energy required to remove an electron from an atom or ion. Atoms with low ionization energies tend to lose electrons more readily.
3. Electron Affinity:
Electron affinity is the energy change that occurs when an atom gains an electron. Atoms with high electron affinities tend to gain electrons more readily.
Balancing Redox Reactions
Balancing redox reactions can be more complex than balancing other types of chemical reactions. The key is to ensure that the number of electrons lost in the oxidation half-reaction equals the number of electrons gained in the reduction half-reaction. Several methods exist, including the half-reaction method and the oxidation number method. These methods involve systematically adjusting coefficients to achieve electron balance.
Conclusion: The Significance of Electron Transfer
The transfer of electrons, central to redox reactions, underpins a vast array of chemical processes. From the energy production in our bodies to the manufacturing of various materials, understanding when and why electrons are lost or gained is essential for comprehending the world around us and developing new technologies. By carefully analyzing changes in oxidation states, using half-reactions, and identifying oxidizing and reducing agents, we can gain a deeper appreciation of the intricate dance of electrons and their profound impact on the chemical universe. Further exploration into the nuances of redox reactions will only continue to reveal more of their importance and applications in various scientific fields.
Latest Posts
Latest Posts
-
Is The Electromagnetic Spectrum Transverse Or Longitudinal
Apr 03, 2025
-
Compare And Contrast Pulmonary And Systemic Circulation
Apr 03, 2025
-
Icse Tuitions Classes In Jail Road Nashik
Apr 03, 2025
-
What Is A Free Variable In A Matrix
Apr 03, 2025
-
Compare Protein Synthesis In Prokaryotes And Eukaryotes
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Electrons Are Lost Or Gained During . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
